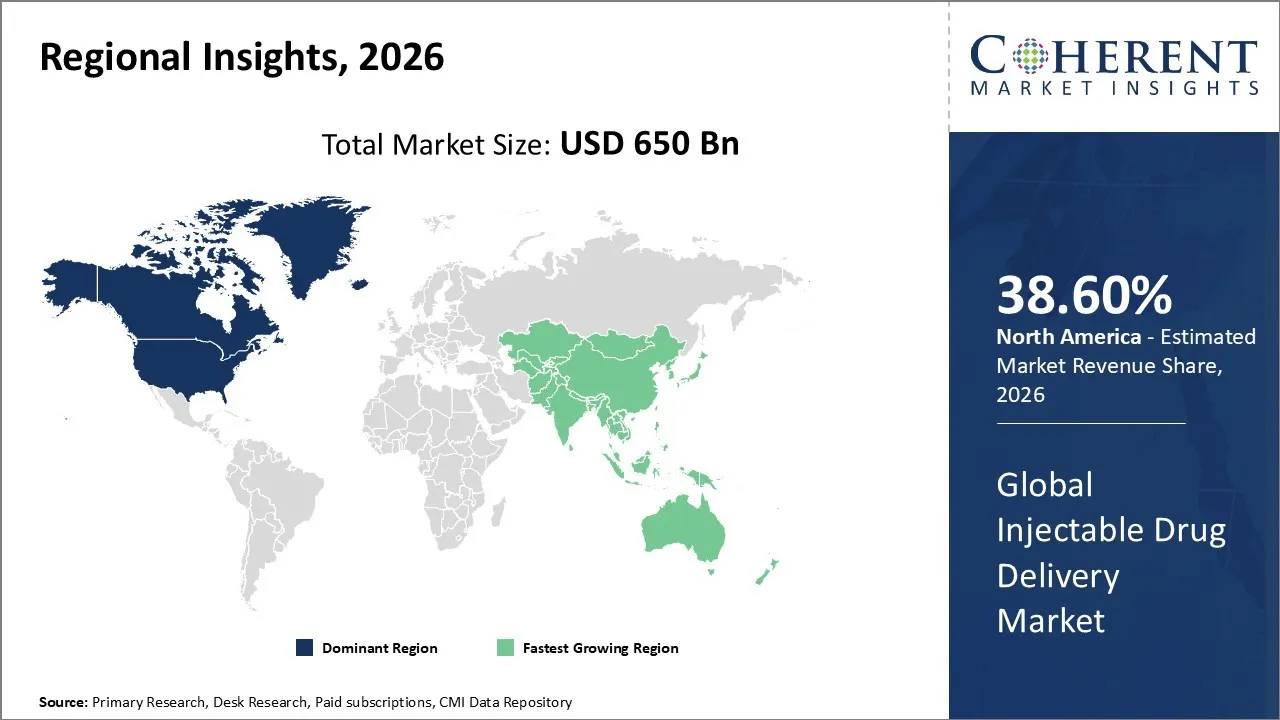
The Injectable Drug Delivery Market is estimated to be valued at USD 650 Bn in 2026 and is expected to reach USD 900 Bn by 2033, growing at a compound annual growth The rate (CAGR) of 7% from 2026 to 2033.
The global injectable drug delivery market is experiencing growth primarily due to the rising prevalence of chronic conditions such as diabetes, cancer, and autoimmune diseases, along with an increasing reliance on biologics and biosimilars. In addition, continuous technological innovation serves as a main driving force, focusing on enhancing patient convenience and adherence. A notable trend in the industry is the movement toward patient self-administration and home healthcare. The innovations particularly emphasize patient-centered solutions, the integration of technology via smart, connected devices for better monitoring, and the growing pipeline of new biologic drugs.
|
Current Event |
Description and the Impact |
|
Technological Innovations and R&D Advances |
|
|
Macroeconomic Factors and Market Dynamics |
|
|
Healthcare Infrastructure and Policy Shifts |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
In terms of device type, the prefilled syringes segment contributes the highest share of 28.50% in the market in 2026 by transforming how healthcare providers and patients administer medication. These devices eliminate the manual step of drawing a drug from a vial, which directly reduces the likelihood of dosing errors and accidental contamination. The segment is growing as pharmaceutical companies are launching additional vaccines and specialized biologics that necessitate accurate premeasured volumes. Prefilled syringes also assist in high-volume clinical operations while empowering patients to manage their treatments at home with greater confidence. The integrated design of these syringes ensures that life-saving medications remain accessible and easy to use for everyone involved in the care cycle.
For instance, in October 2025, West Pharmaceutical Services, Inc. introduced the West Synchrony™ Prefillable Syringe (PFS) System at the CPHI Worldwide in Frankfurt, Germany. This system provides a fully certified platform from a single source and will be available for purchase in January 2026. It marks a significant advancement in drug delivery options.
In terms of material type, the glass segment contributes the highest share of 54.90% in 2026 of the market. Manufacturers favor high-purity borosilicate glass because its chemical inertness prevents unwanted reactions between the container and the medication. This stability is essential for preserving the potency of sensitive biopharmaceuticals over longer periods. Glass provides an exceptional barrier against oxygen and moisture, thus ensuring that the internal environment remains sterile and consistent. Although specialized polymers provide alternative solutions for specific needs, glass continues to lead the market due to its well-established regulatory history and strong global production infrastructure. It continues to be the gold standard for safeguarding a diverse range of liquid formulations and lyophilized powders.
For instance, in October 2025, SCHOTT Pharma, a leader in medication containment solutions and delivery systems, has released the first 5.5 ml prefillable staked-needle glass syringe. This new large-volume syringe makes the transition to large-volume drug delivery systems for at-home biologic therapy easier.
In terms of usability, the disposable segment contributes the highest share of 82.70% in 2026 of the market as healthcare systems prioritize infection control and user safety above all else. Single-use devices ensure sterility with every administration, thereby reducing the spread of bloodborne infections. The capability is especially critical during mass vaccination campaigns, where high patient volumes elevate infection control challenges, as well as for individuals managing chronic conditions in home care settings. The simple use-and-toss nature of these devices removes the complex need for sterilization or maintenance. The disposable devices use advanced shielding technologies to protect users from accidental needlestick injuries. These safety features firmly secure their position as the primary choice for modern, safe, and convenient drug administration.
To learn more about this report, Request Free Sample
North America has remained the dominant region with 38.60% in 2026 of the global Injectable Drug Delivery Market over the past decade. The growth is attributed to the increasing prevalence of chronic conditions like cancer, diabetes, and autoimmune disorders, as well as the growing preference for convenient self-administration and home-based healthcare options. The large share can be attributed to the advanced healthcare infrastructure and considerable healthcare spending. One of the trend is the increasing use of advanced and user-friendly devices such as prefilled syringes, auto-injectors, and wearable injectors.
For instance, in November 2025, the global pharmaceutical giant Lupin Limited has announced the launch of Risperidone in single-dose vials, available in 25 mg, 37.5 mg, and 50 mg formulations of the extended-release injectable suspension. These vials will have a 180-day CGT exclusivity period in the US.
The Asia Pacific region is experiencing the fastest growth in the Injectable Drug Delivery Market. This is owing to the increasing burden of chronic diseases like cancer, diabetes, and autoimmune disorders, alongside a rapidly aging population in the region. The incorporation of biologics, biosimilars, and demand for patient-centric self-administration devices like auto-injectors and pen injectors are key drivers for the market’s growth. China is likely to be the fastest growing country. This is due to its large patient population and healthcare investments.
For instance, in October 2025, the global pharmaceutical giant Lupin Limited has announced the initiation of a strategic collaboration program aimed at expanding the application of PrecisionSphereTM, a long-acting injectable (LAI) platform developed by Nanomi B.V., a subsidiary of Lupin. Recently, PrecisionSphereTM received approval from the U.S. Food and Drug Administration (FDA), confirming its safety and efficacy for drug delivery and readiness for commercial use.
The US is witnessing growth owing to the increasing prevalence of chronic conditions like diabetes, cancer, and autoimmune disorders, which depend on injectable treatments for effective management. A key factor contributing to this growth is the advancement of technology. There is a notable shift towards patient-friendly, self-administration devices like prefilled syringes, auto-injectors, and wearable injectors, which improve convenience and adherence. The market is also seeing a trend towards connected, smart injectors that facilitate remote monitoring and improve treatment outcomes in a patient-centric care model.
For instance, in December 2024, Baxter International Inc., a global leader in injectables, anesthesia, and drug compounding, has announced the release of five new injectable drugs products in the US.
China is experiencing rapid growth due to the rising prevalence of chronic diseases such as diabetes and cancer. The growing demand for biologics and biosimilars and substantial government investment in modernizing healthcare infrastructure are also contributing to the growth. The market trends include a shift toward patient-centric, self-administration devices such as prefilled syringes, auto-injectors, and wearable injectors, which assist in enhancing convenience and treatment adherence. Major international and domestic firms are strategically collaborating and innovating to meet the evolving market demands.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 650 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7% | 2033 Value Projection: | USD 900 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Becton, Dickinson and Company, Pfizer Inc., Novo Nordisk A/S, Gerresheimer AG, Carl-Zeiss-Stiftung, Terumo Corporation, Vetter Pharma-Fertigung GmbH & Co. KG, SHL Group, Owen Mumford Limited, and Injex Pharma GmbH |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The injectable drug delivery market is experiencing sustained growth mainly fueled by the expanding global patient population affected by chronic conditions like diabetes, cancer, and autoimmune disorders. Many modern therapeutic breakthroughs, particularly biologics and monoclonal antibodies, possess complex molecular structures that fail to survive the digestive tract, which renders injection the essential route for administration. As these long-term illnesses require frequent and precise dosing, patients increasingly adopt self-administration practices. The growing demand is propelling innovation in patient-centric delivery services, thereby allowing patients to manage their therapies at home rather than in clinical settings. The injectable drug delivery devices are central to chronic disease management, as they provide consistent pharmacokinetic profiles, rapid bioavailability, and greater uniformity in patient adherence.
The injectable drug delivery market is a key segment within pharmaceutical delivery, driven by growing demand for chronic disease management and biologic therapies. Advanced delivery systems such as prefilled syringes, autoinjectors, and wearable injectors are increasingly adopted to improve dosing accuracy, patient comfort, and self-administration. Prefilled syringes account for a significant portion of the device market, while wearable injectors represent one of the fastest-growing segments.
Therapeutic demand is concentrated in chronic conditions, with diabetes, oncology, and autoimmune disorders leading adoption due to biologics and targeted therapies. Hospitals and ambulatory surgical centers remain the primary end-use settings, though home care is rapidly expanding as user-friendly systems facilitate self-administration.
Regionally, North America continues to be the largest market, supported by high healthcare infrastructure and early adoption of advanced devices, while Asia Pacific is emerging as the fastest-growing region due to expanding healthcare access and investment.
Product trends indicate formulations maintain a leading share, driven by therapeutic volume, while device innovations such as smart and connected injectors are gaining traction through adherence monitoring and real-world usage data collection. Subcutaneous formulations of major biologics further enhance patient convenience and influence prescribing patterns. Overall, the market growth reflects a balance between established clinical delivery and innovative, patient-centric technologies enhancing safety, access, and therapeutic outcomes.
Definition: The Injectable Drug Delivery Market focuses on the development and production of advanced delivery platforms designed for the precise administration of liquid pharmaceuticals. This sector integrates high-precision engineering with digitally enabled smart software to ensure the accurate, consistent, and controlled delivery of complex biologics. The modern injectable delivery solutions emphasize patient-centric design, thus allowing for safe and reliable self-administration. These systems play a critical role in large-scale healthcare facilities and home care settings, where speed, dosage precision, and sterility are important. The market demand is due to the growing global burden of chronic diseases. This is prompting pharmaceutical manufacturers and healthcare providers to use advanced delivery technologies that meet increasingly stringent safety and regulatory requirements.
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients