Intra Abdominal Pressure Measurement Devices Market is estimated to be valued at USD 167.7 Mn in 2026 and is expected to reach USD 482.6 Mn in 2033, exhibiting a compound annual growth rate (CAGR) of 16.3% from 2026 to 2033.
Analysts’ Views on Intra Abdominal Pressure Measurement Devices Market:
The purpose of technology is to review devices designed to measure intra-abdominal pressure (IAP). These devices are used to detect intra-abdominal hypertension (IAH) and abdominal compartment syndrome (ACS). Safety information about IAP measuring devices comes from the manufacturer and user device Experience (MAUDE) database. For instance, elevated intra-abdominal pressure (IAP) is a known cause of increased morbidity and mortality in critically ill patients. Intra-abdominal hypertension (IAH) and abdominal syndrome can lead to rapid deterioration of organ function and the development of multiple organ failure.
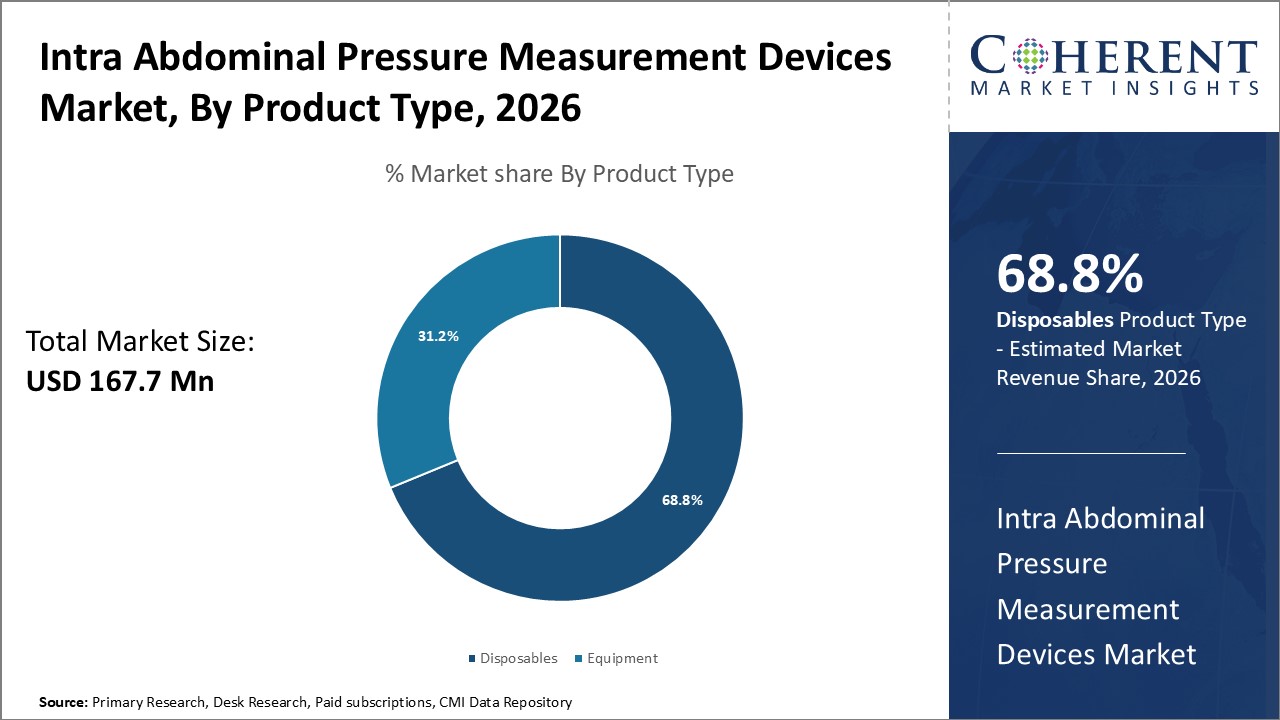
Figure 1. Global Intra-abdominal Pressure Measurement Devices Market Share (%), by Product Type, 2026
To learn more about this report, Request Free Sample
Global Intra Abdominal Pressure Measurement Devices Market – Driver
Development of di-(2-ethylhexyl) phthalate (DEHP) free products is expected to drive the market
The development of di-(2-ethylhexyl) phthalate (DEHP)-free products is expected to boost the growth of the global intra abdominal pressure monitoring devices market during the forecast period. DEHP is often used in the medical device industry as a plasticizer for various plastic materials to make them plastic flexible. The use of these phthalates in the device or disposable manufacturing process can suppress testosterone production in the adult testicles and affect reproductive health. Therefore, market players are focusing on providing alternatives to DHEP, such as DEHT (di-(2-ethylhexyl) terephthalate), which is not genotoxic and does not cause reproductive toxicity. For instance, On Febuary 26, 020, Sun Pharmaceutical Industries Ltd., a Pharmaceutical company has launched Riomet ER in the U.S. as a dietary supplement and exercise supplement to improve glucose control in patients 10 years of age and older with type 2 diabetes.
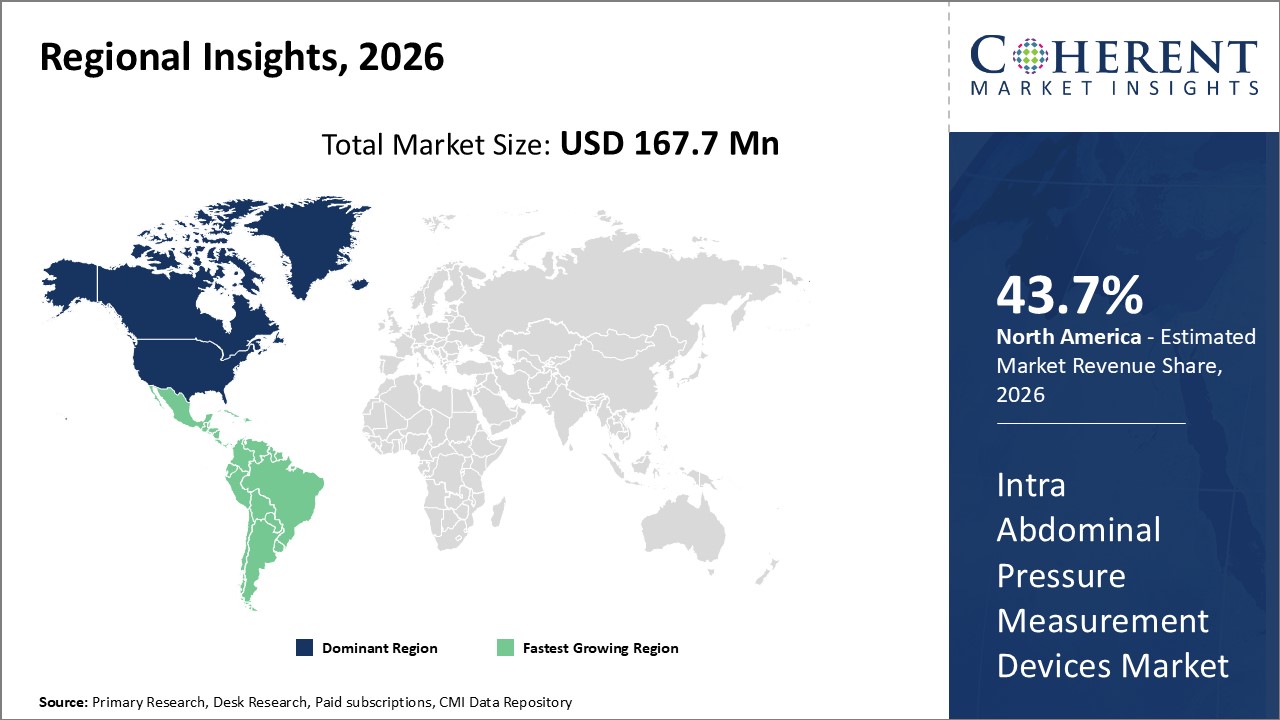
Figure 2. Global Intra Abdominal Pressure Measurement Devices Market (US$ Million), by Region, 2026
To learn more about this report, Request Free Sample
Global Intra Abdominal Pressure Measurement Devices Market- Regional Analysis
The North American region is expected to dominate the global intra-abdominal pressure measurement devices market due to increasing funding for intra-abdominal pressure measurement devices from regulatory agencies. Potrero Medical has raised a total funding of US$ 72.7 Mn over 6 rounds. Their latest funding round was a Series C round on Apr 07, 2026 for US$ 17.7 Mn.
During COVID, the disease has spread to over 100 countries across the globe and the World Health Organization has declared it a public health emergency.
Moreover, various healthcare regulatory authorities have issued guidelines for patients who require critical care during the
Due to ever-evolving travel restrictions, face-to-face conferences had to be rescheduled and replaced with online meetings. The WSACS conference scheduled for May 2021 in Snowbird, Utah, USA was not spared from these attempts and had to be cancelled.
Intra Abdominal Pressure Measurement Devices Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 167.7 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 16.3% | 2033 Value Projection: | USD 482.6 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
CR. Bard, Inc. (Becton, Dickinson, and Company), ConvaTec Group PLC, Stryker Corporation, Biometrix Ltd. (Degania Silicone, Ltd.), Centurion Medical Products (Medline Industries, Inc.), and Potrero Medical |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Intra Abdominal Pressure Measurement Devices Market Segmentation:
The Global Intra Abdominal Pressure Measurement Devices Market is segmented into product type, application, and end user.
Based on product type, disposables, and equipment. Out of which, the disposables is expected to dominate the global Intra Abdominal Pressure Measurement Devices Market over the forecast period, and this is attributed to demand of device in the device market.
Based on Application, Intra-abdominal hypertension, and intra-compartment pressure. Out of which, intra-compartment pressure is expected to dominate the global Intra Abdominal Pressure Measurement Devices Market over the forecast period, and as ACS is a sustained condition that is associated with a new organ dysfunction or failure.
Based on End user, Hospitals, Trauma Centers, Ambulatory Surgical Centers (ASCs), Clinics. Out of which, the Hospital segment is expected to dominate the market over the forecast period and this is attributed to the growing burden of devices.
Global Intra Abdominal Pressure Measurement Devices Market: Key Developments
On Feb 9, 2022, Urimetrix, a medical device company, launched by Biometrix as a comprehensive product line for urine drainage management that includes urine meters, catheters with temperature sensors, and intra abdominal pressure sets with accurate pressure transducer monitoring systems.
On April 2022, article published in PubMed, IAH is a significant and independent predictor of short-term and long-term mortality, with severe organ failure and long-term hospitalization.
Global Intra Abdominal Pressure Measurement Devices Market - Cross sectional analysis
As product base segment, disposables and equipment. Out of which, the disposables is expected to dominate the global Intra Abdominal Pressure Measurement Devices Market. Disposables are more often used as they can be cheaper and cannot be re-used. Disposable means intended to be thrown away after use.
Global Intra Abdominal Pressure Measurement Devices Market - Key Trends
Strategies such as funding and product launch
The key market players are engaged in adopting inorganic strategies such as funding and product launches. For instance, Israeli Sereno Medical on January 20, 2022 raised funding of US$ 1.5 Mn for its kidney function device. Elcam Medical has invested in the company to manufacture Sentinel, Sereno’s urinary output, and IAP Monitoring devices for the detection of acute kidney injury.
Global Intra Abdominal Pressure Measurement Devices Market: Restraint
Complications associated with pressure measurement devices
Procedural complications are expected to hinder the growth of the global intra-abdominal pressure monitoring devices market. Endodominal pressure is usually measured through the patient's bladder. Some of the most serious problems associated with pressure control devices include bladder infections and needlestick injuries, which can cause patient discomfort and increase the risk of compliance. Although several publications have been published in the recent past, for instance, the data published in the Journal of Wound, Ostomy and Continence Nursing in June 2022, IAP measurement techniques and clinical applicability are not yet fully established. Intravesical IAP measurement is consensual. However, there are issues related to the zero point, infusion volume, measurement frequency, and indications. Such a scenario is also expected to hinder the growth of the market.
Global Intra Abdominal Pressure Measurement Devices Market - Key Players
Major players operating in the global ambulatory surgical center market are CR. Bard, Inc. (Becton, Dickinson, and Company), ConvaTec Group PLC, Stryker Corporation, Biometrix Ltd. (Degania Silicone, Ltd.), Centurion Medical Products (Medline Industries, Inc.), and Potrero Medical.
Definition: Intra-abdominal pressure measurement devices are medical devices increasingly used to evaluate the blood pressure in the abdominal compartment. Intra-abdominal pressure occurs in numerous healthcare settings, including acute decompensated heart failure, resuscitation with large volume, hepatorenal syndrome, sepsis, and acute pancreatitis.
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients