Intravenous Stopcock Market is estimated to be valued at USD 537.5 Mn in 2026 and is expected to reach USD 939.2 Mn in 2033, exhibiting a compound annual growth rate (CAGR) of 8.3% from 2026 to 2033.
Analysts’ views on Global Intravenous Stopcock Market:
A stopcock is a type of medical device and ball valve used for intravenous administration. The flow of fluid is directed into the blood vessels with an intravenous cap. There are different types of shut-off valves such as burette shut-off valves, vacuum valves and needle valves. These medical devices are primarily designed to control gas or liquid flow with an on/off controller. A closure clip is used in intravenous extensions to facilitate control of the fluid pathway during fluid or drug administration. However, access to a patient's intravenous line through an open faucet or multiple hubs can increase the risk of bacterial contamination and infection. Stopcocks have been used in infusion therapy for decades, but studies have confirmed concerns about the risk of intraluminal contamination and fluid flow dynamics associated with stopcocks.
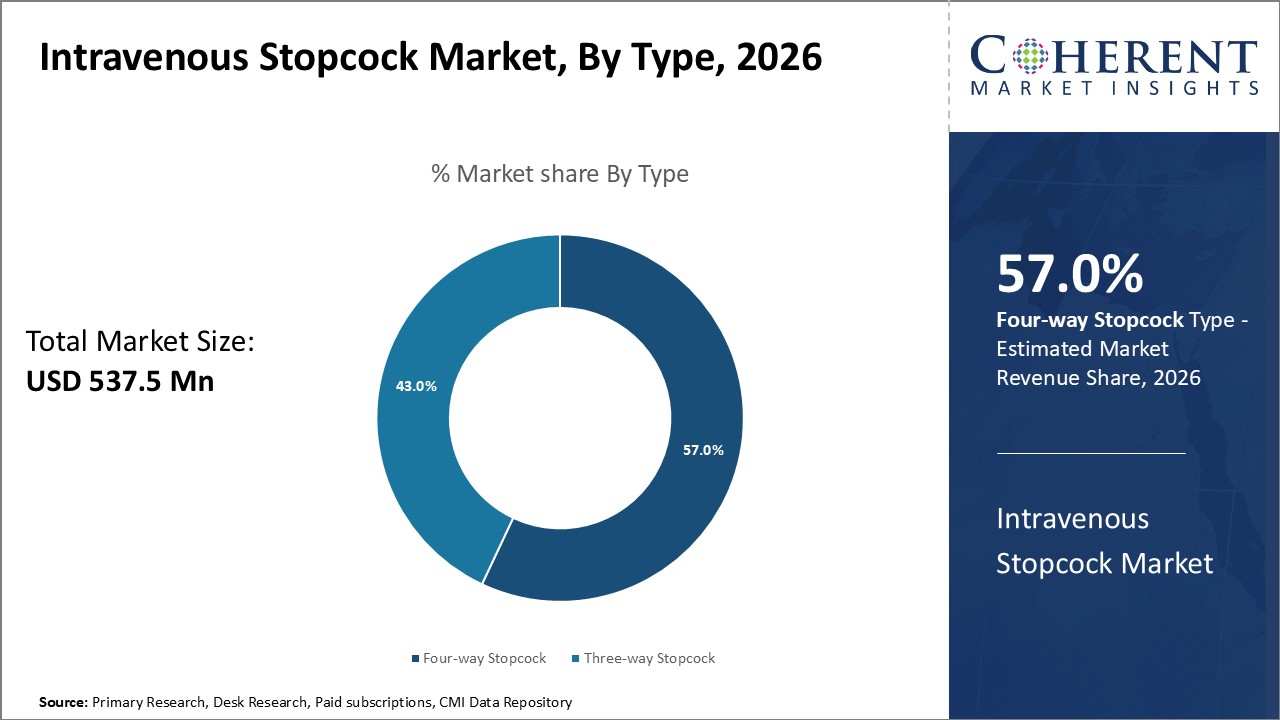
Figure 1. Global Intravenous Stopcock Market Share (%), By Type, 2026
To learn more about this report, Request Free Sample
Global Intravenous Stopcock Market– Drivers
Increasing Research and Development Activities in Intravenous stopcock
Increasing research and development in the field of stopcocks is expected to drive the global intravenous stopcock market growth over the forecast period. For instance, B. Braun SE, explained Discofix C with Safeflow is a 3-way stopcock with integrated swab able valve and combines the advantages of Discofix C and Safe flow. Resistant against pharmaceuticals with previously unattained degree of transparency, resistance against stress cracking resulting from exposure to infusion solutions, drugs and alcohol-based disinfectants, protection against the danger of unnoticed cracks, air infusion and leakage, fast connection with rotating adapter.
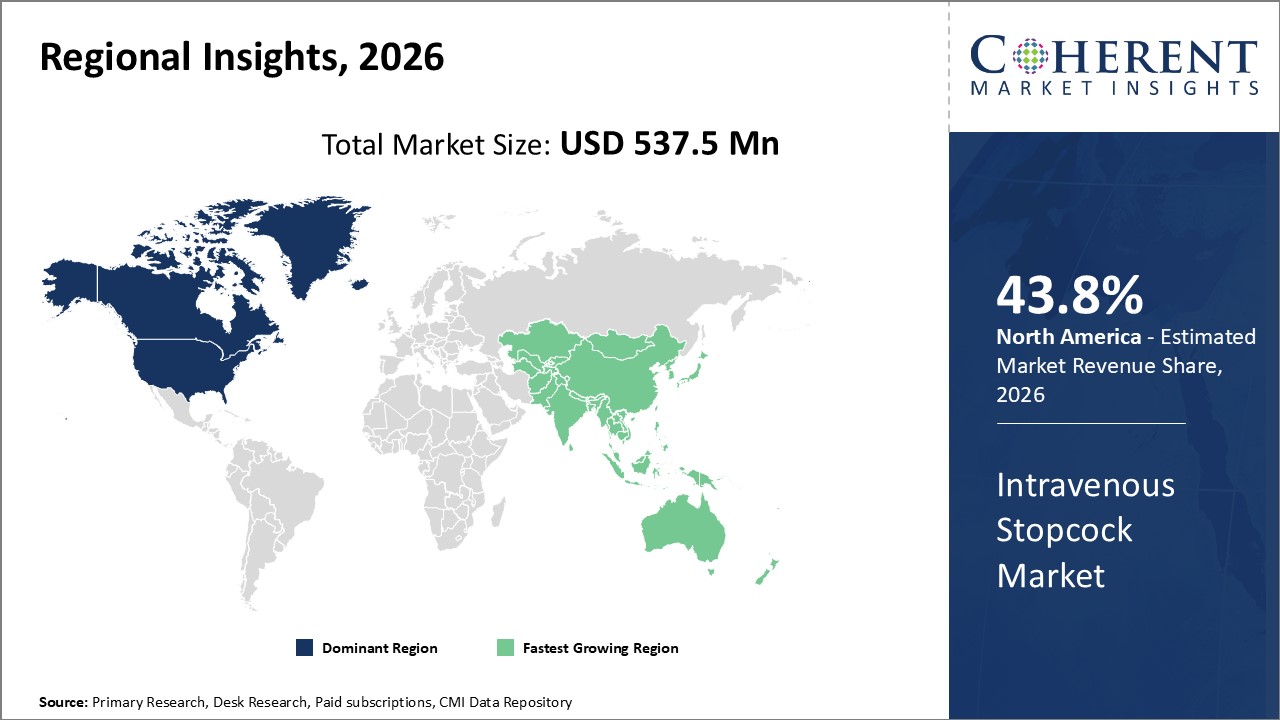
Figure 2. Global Intravenous Stopcock Market Value (US$ Million), By Region, 2026
To learn more about this report, Request Free Sample
Global Intravenous Stopcock Market - Regional Analysis
Among region, North America is estimated to hold a dominant position in the global intravenous stopcock market over the forecast period, owing to increasing treatments of products. For instance, on May 19, 2021, an article published by the National Center for Biotechnology Information, explained Treatment of Phlebitis Secondary to the Insertion of a Peripheral Venous Catheter, hygienic hand washing, and usage of clean gloves for both punctures and equipment, hubs, stopcocks, and bio-connectors handling. Nursing interventions to prevent phlebitis and ensure a proper catheter use included those related to the maintenance of intravenous therapy, asepsis, and choosing the dressing. With regard to the nursing interventions to treat phlebitis, these were focused on vigilance and caring and also on the use of medical treatment protocols. It is not necessary to wear sterile gloves if the previously disinfected area is not touched again during the technique. Using disposable tourniquets can help reduce Peripheral Venous Catheters contamination rates.
Global Intravenous Stopcock Market
Since the virus outbreak in December 2019, the disease has spread to over 100 countries across the globe, and the World Health Organization declared it a public health emergency on January 30, 2020.
For instance, according to an article published by National Center for Biotechnology Information, on April 26, 2022 explained, in the four-chamber view, a right-to-left shunt was sought by injecting 9.5 mL of gelatin solution aerated with 0.5 mL of room air through two syringes connected to a three-way stopcock. Up to three successful contrast studies were performed per patient. The shunt was quantified according to the number of bypassed bubbles as follows: small, moderate and large.
Intravenous Stopcock Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 537.5 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 8.3% | 2033 Value Projection: | USD 939.2 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Becton, Dickinson and Company, Global Medikit Limited, SCW Medicath Ltd., Fresenius Kabi AG, Smith’s Medical, Braun Medical Inc. , Terumo Medical Corporation, ARGON MEDICAL, Baxter, Bound Tree Medical, Mediprim, Neotec Medical Industries, ICU Medical. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Intravenous Stopcock Market Segmentation:
Global Intravenous Stopcock Market report is segmented into type, application, end user and region.
Based on Type, the global intravenous stopcock market is segmented into Four-way Stopcock, Three-way Stopcock. Out of which, three way stopcock segment is expected to dominate the market due to increasing research and development activities.
Based on Application, the global intravenous stopcock market is segmented into Pressure Monitoring, and Infusion Therapy. Among these, infusion therapy segment is expected to dominate the market over the forecast period due to increasing usage of techniques in surgeries.
Based on End User, the global intravenous stopcock market is segmented Hospitals, Specialty Clinics, Ambulatory Surgical Centers, and Others. Out of which, hospitals segment is expected to dominate the market over the forecast period due to advanced treatments.
Based on Region, the global intravenous stopcock market is segmented into North America, Latin America, Europe, Asia Pacific, and Middle East & Africa. Among these, North America is expected to dominate the market over forecast period due to increased research and development activities.
Global Intravenous Stopcock Market: Key Developments
On April 13, 2021, Mais India Medical devices, a medical disposable equipment manufacturer company, India explained for the simultaneous administration of two or more fluids or medicines to patients, the concept of Stopcock was introduced to provide proper control over the flow of direction. A stopcock is a kind of valve used to control the flow of direction of liquids or gases. They are widely available in different designs or forms among which 3-way stopcock is most commonly used for synchronous and continuous infusion of two fluids.
Global Intravenous Stopcock Market: Key Trends
Introduction of newer instruments for intravenous stopcock
Introduction of newer and more efficient instruments to boost up techniques can drive growth of market. On March 15, 2023, Niir Project Consultancy services, Indian based company which provide technical consulting services announced start a Manufacturing Business of 3-way Stop Cocks. The basic design of a three-way stop click is simple, but the science behind it is more complex. The valve inside the shut-off valve is made of a ball or cap that turns with a handle. There is a hole in the center of the ball or cap through which liquid can pass if it is directed into one of the ports. The faucet valve is also designed to create a tight seal when closed, preventing liquid leakage. This is important because many liquids that flow through a stopcock can be dangerous or infectious.
Global Intravenous Stopcock Market: Restraints
Contamination risk with intravenous stopcock
The sterility is expected to hamper the global intravenous stopcock market growth. For instance, according to an article published by National Center for Biotechnology Information, on February 3, 2023, explained the use of an antimicrobial dressing reduces skin colonization at the peripheral intravenous catheter insertion site and may provide an additional benefit by reducing the risk of local infection and/or catheter-related bloodstream infection. Protection of PIVC cells is important to form a barrier to infection and prevent transmission. Strong ligamentous adhesion is an important part of catheter fixation. To reduce the risk of contamination, protection should be done in a standardized way. Extensions increase safety by minimizing movement at the installation site.
To counterbalance this restraint, a robust protocol should be controlled by sterility in intravenous stopcock.
Global Intravenous Stopcock Market - Key Players
Major players operating in the global intravenous stopcock market include Becton, Dickinson and Company, Global Medikit Limited, SCW Medicath Ltd., Fresenius Kabi AG, Smith’s Medical, Braun Medical Inc. , Terumo Medical Corporation, ARGON MEDICAL, Baxter, Bound Tree Medical, Mediprim, Neotec Medical Industries, and ICU Medical.
*Definition: An intravenous ball valve and type of medical equipment is called a stopcock. An intravenous cap directs the fluid flow into the blood arteries. Shut-off valves come in a variety of designs, including burette, vacuum and needle valves. With an on/off controller, this medical gadget is primarily made to regulate the flow of gas or liquid. In intravenous extensions, a closure clip is used to help manage the fluid channel during the administration of fluids or medications.
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients