The Knee Surgeries and Arthroplasty Pain Management Therapeutics Market is estimated to be valued at USD 4,876.4 Mn in 2026 and is expected to reach USD 7,107.3 Mn by 2033, exhibiting a compound annual growth rate (CAGR) of 5.2% from 2026 to 2033.
The Knee Surgeries and Arthroplasty Pain Management Therapeutics Market deliver effective solutions for managing postoperative pain after knee surgeries, including total and partial knee arthroplasty. It offers a variety of therapeutics, such as NSAIDs, opioids, local anesthetics, and multimodal analgesic regimens, to improve patient comfort and accelerate recovery. Rising knee disorder cases, increasing surgical procedures, advanced healthcare infrastructure, and the implementation of enhanced recovery protocols actively drive market growth, with hospital pharmacies and specialty clinics distributing these therapies.
|
Current Events |
Description and its impact |
|
Global Aging Population & Increasing Osteoarthritis Prevalence |
|
|
Technological Innovations in Surgical Procedures and Pain Management |
|
|
Geopolitical Tensions and Supply Chain Disruptions |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Hospital Pharmacies expected to hold largest market share of 43.3% in 2026. Hospital pharmacies actively drive the Knee Surgeries and Arthroplasty Pain Management Therapeutics Market by distributing pain management drugs efficiently and under strict control. They provide access to prescription-only medications, implement multimodal analgesia protocols, and handle high patient volumes from surgeries. By enhancing safety, optimizing dosing, and collaborating closely with clinical teams, they enable personalized patient care. Increasing surgical procedures, advanced hospital infrastructure, and the demand for regulated drug dispensing continue to strengthen hospital pharmacies’ central role in postoperative pain management. For instance, in November 2025, Chief Commercial Officer Duncan McKechnie reported that the launch of the non-opioid pain reliever Journavx is showing positive trends.
Non-steroidal Anti-inflammatories hold the largest market share of 36.8% in 2026. Non-Steroidal Anti-Inflammatories (NSAIDs) drive growth in the Knee Surgeries and Arthroplasty Pain Management Therapeutics Market by effectively controlling postoperative pain and inflammation. Healthcare providers actively integrate NSAIDs into multimodal pain management strategies to minimize opioid use and accelerate patient recovery. Increasing knee surgery procedures, growing awareness of non-opioid pain relief, the availability of advanced NSAID formulations, and their incorporation into hospital protocols all boost adoption, establishing NSAIDs as a central element of postoperative care. For instance, in February 2026, Pharma major Lupin Limited launched the authorized generic of Horizon Therapeutics’ Vimovo (naproxen/esomeprazole magnesium) delayed-release tablets, 375 mg/20 mg and 500 mg/20 mg, in the U.S. The combination of naproxen, an NSAID, and esomeprazole magnesium, a PPI, treats arthritis pain in patients aged 12 and above while reducing the risk of naproxen-induced gastric ulcers.
To learn more about this report, Request Free Sample
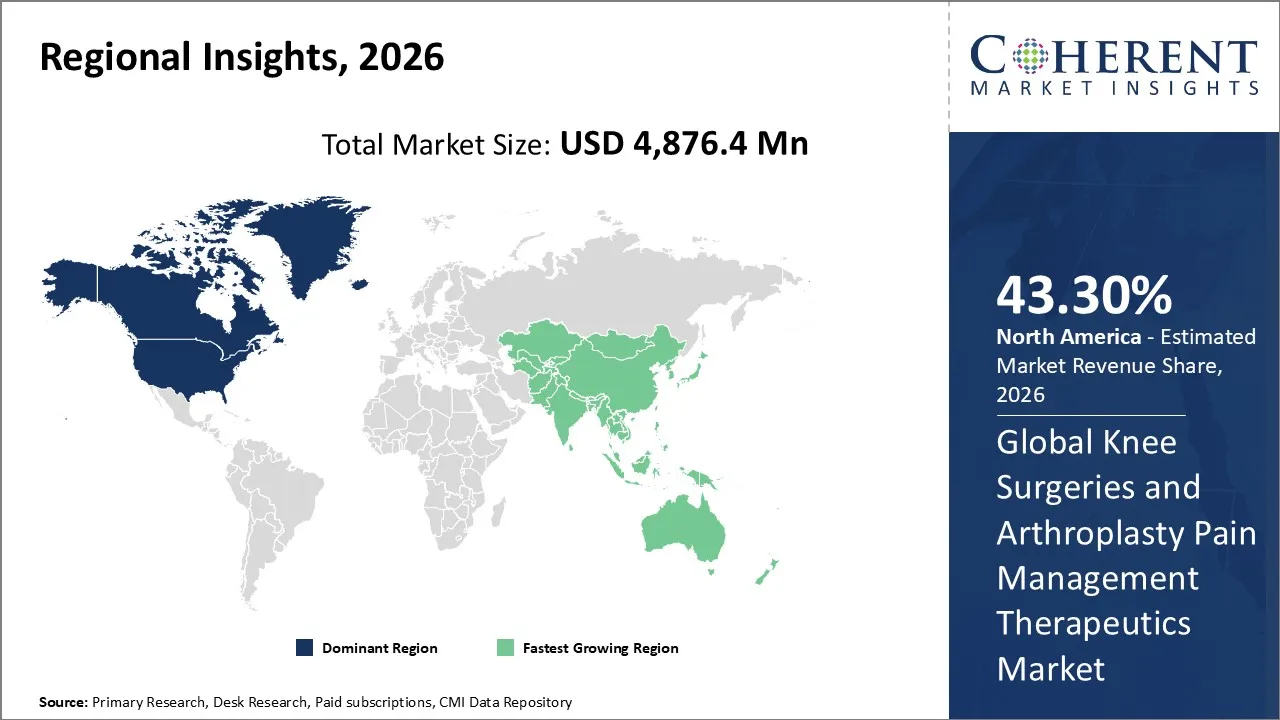
North America dominates the overall market with an estimated share of 43.3% in 2026. The North American Knee Surgeries and Arthroplasty Pain Management Therapeutics Market actively embraces advanced pain management strategies, such as multimodal analgesia and non-opioid therapies. Healthcare providers implement enhanced recovery protocols to improve postoperative outcomes and shorten hospital stays. Well-established healthcare infrastructure, high surgical volumes, and robust insurance coverage enable widespread access to innovative therapeutics. Increasing knee disorder prevalence, ongoing clinical research, and the rapid introduction of new drug formulations and delivery methods continue to drive and shape market trends across the region.
The Asia Pacific Knee Surgeries and Arthroplasty Pain Management Therapeutics Market is rapidly developing as healthcare systems modernize and surgical infrastructure strengthens across the region. Rising knee disorder cases and increased awareness of effective postoperative pain management drive the use of NSAIDs, opioids, and multimodal analgesics. Hospitals actively implement standardized pain control protocols and enhanced recovery practices. Expanding hospital networks, improved access to advanced therapeutics, and continued investments in orthopedic care actively shape the region’s market, establishing it as a key growth segment in global postoperative pain management. For instance, in March 2025, Medicover Hospital in Navi Mumbai introduced the MISSO high-tech robotic system for knee and hip replacements, enhancing surgical precision while reducing patient discomfort and enabling faster recovery.
The United States Knee Surgeries and Arthroplasty Pain Management Therapeutics Market actively advances as hospitals and surgical centers adopt cutting-edge pain management strategies, including multimodal analgesia and non-opioid therapies. Rising cases of knee disorders and increasing demand for total and partial knee arthroplasty drive widespread use of NSAIDs, opioids, and local anesthetics. Healthcare providers implement enhanced recovery protocols to boost patient outcomes and reduce hospital stays, while robust insurance coverage, established healthcare infrastructure, and ongoing clinical research accelerate the adoption of innovative pain management solutions.
The Japan Knee Surgeries and Arthroplasty Pain Management Therapeutics Market actively grows as hospitals adopt advanced postoperative pain management strategies, including multimodal analgesia and non-opioid therapies. An aging population and rising knee disorder cases drive higher demand for total and partial knee arthroplasty, increasing the use of NSAIDs, opioids, and local anesthetics. Healthcare providers implement enhanced recovery protocols to boost patient outcomes and shorten hospital stays, while investments in orthopedic infrastructure, expanded access to innovative therapeutics, and growing awareness of effective pain control actively shape market development.
Robotics is playing an increasingly important role in the arthroplasty pain management therapeutics market by improving surgical precision and patient outcomes. Robotic-assisted joint replacement systems enable surgeons to perform procedures with greater accuracy, minimizing tissue damage and reducing postoperative pain. This leads to faster recovery, shorter hospital stays, and lower reliance on pain medications, including opioids. Additionally, robotics supports personalized surgical planning and better implant alignment, which can enhance long-term joint function and reduce complications.
In July 2025, Sarvodaya Hospital in Gaur City 2, Greater Noida West introduced a fully operational Joint Replacement Robot (RJR) for joint replacement surgeries. The system enhances surgical precision and helps reduce patient recovery time, reflecting the hospital’s commitment to integrating advanced technology into clinical care.
Hospitals and surgical centers increasingly adopt multimodal analgesia strategies, combining NSAIDs, opioids, local anesthetics, and adjunct therapies. This approach enhances pain control while minimizing opioid-related side effects, supporting faster mobilization and recovery. Clinicians favor integrating multiple drug classes and regional anesthesia techniques to tailor therapy to patient needs, improve outcomes, and reduce hospital stays. The trend reflects a broader shift toward personalized, protocol-driven postoperative pain management in orthopedic procedures.
Growing awareness of opioid risks drives healthcare providers to emphasize non-opioid analgesics, including NSAIDs, COX-2 inhibitors, and acetaminophen. These alternatives reduce dependency potential and improve safety profiles, particularly for elderly or comorbid patients. Non-opioid therapies are increasingly incorporated into standardized recovery protocols, aligning with enhanced recovery after surgery (ERAS) guidelines, and hospitals actively invest in training staff on proper implementation to maximize patient comfort and optimize postoperative outcomes.
The growing adoption of minimally invasive knee surgeries presents opportunities for tailored pain management solutions. These procedures require precise postoperative analgesia to support early mobilization and rapid recovery. Manufacturers can focus on developing fast-acting, targeted therapeutics, including local anesthetics and controlled-release NSAIDs, that align with shorter hospital stays. Integrating these solutions into surgical protocols offers hospitals and providers a chance to enhance patient satisfaction and improve rehabilitation outcomes while reducing overall healthcare costs.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 4,876.4 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 5.2% | 2033 Value Projection: | USD 7,107.3 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Pfizer Inc, Allay Therapeutics, Mallinckrodt, Johnson & Johnson, Zydus Cadila Healthcare Ltd., Novartis AG, Purdue Pharma, Delphis Pharmaceutical, Taj Pharmaceuticals Limited, GlaxoSmithKline plc, AbbVie Inc., and Aurobindo Pharma |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients