Materiovigilance is the coordinated system of identification, collection, reporting, and analysis of any untoward occurrences associated with the use of medical devices and protection of patient's health by preventing its recurrences. The purpose of materiovigilance is to study and follow incidents that might result from using medical devices. It enables dangerous devices to be withdrawn from the market and to eliminate faults in medical devices with the intention of constantly improving the quality of devices and providing patients and users with increased safety.
The global materiovigilance market is estimated to be valued at US$ 66.3 billion in 2021 and is expected to exhibit a CAGR of 8.0% during the forecast period (2021-2028).
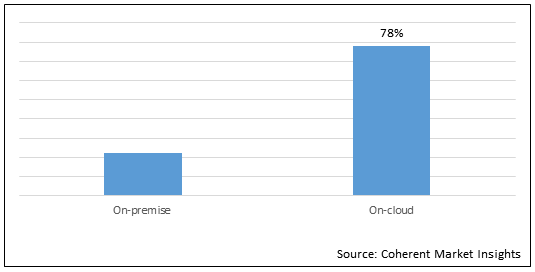
Figure 1.Global Materiovigilance Market Share (%) in Terms of Value, By Delivery Mode, 2021
To learn more about this report, Request Free Sample
Increase in the number of medical device recalls is expected to drive the market growth during the forecast period
The increase in the number of medical device recalls due to technical defects is expected to drive the global materiovigilance market growth over the forecast period. For instance, according to data published by Medical Device and Diagnostic Industry (MD+DI), is a resource exclusively for original equipment manufacturers of medical devices and in vitro diagnostic products, in June 2021, the number of medical device recalls issued during the first half of 2021 is higher than the number of recalls issued during the first half of each of the previous five years. First half of 2021, US Food and Drug Administration (US FDA) had reported 29 medical device recalls.
Materiovigilance Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2020 | Market Size in 2021: | US$ 66.3 Bn |
| Historical Data for: | 2017 to 2020 | Forecast Period: | 2021 to 2028 |
| Forecast Period 2021 to 2028 CAGR: | 8.0% | 2028 Value Projection: | US$ 113.5 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
AssurX, Sparta Systems, Oracle Corporation, Xybion Corporation, Sarjen Systems Pvt. Ltd., MDI Consultants, AB-Cube, QVigilance, Qserve, and ZEINCRO |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
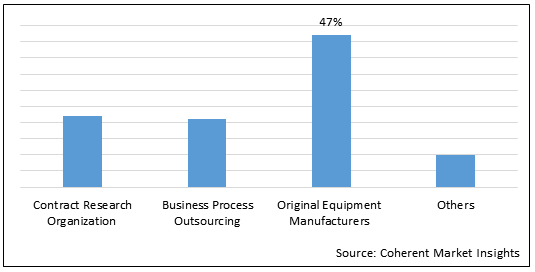
Figure 2.Global Materiovigilance Market Share (%), By End Users, 2021
To learn more about this report, Request Free Sample
Increase in the number of post-market surveillance programs is expected to drive the market growth during the forecast period
Increase in the number of post-market surveillance programs for medical device is expected to drive the global materiovigilance market growth during the forecast period. For instance, in April 2020, Veeva Systems Inc., a leader in cloud-based software for the global life sciences industry, announced Veeva Vault Product Surveillance, a new cloud application for medical device and diagnostics to simplify and standardize the postmarket surveillance process.
Global Materiovigilance Market– Impact of Coronavirus (COVID-19) Pandemic
The rapid global spread of COVID-19 is challenging healthcare systems worldwide in terms of healthcare planning, infrastructure, and spending. Healthcare professionals are forced to treat severely ill patients with limited access to ventilation equipment, qualified intensive care personnel or protective patient monitoring devices. Since December 2019, a novel coronavirus spread throughout China and across the world, causing a continuous increase in confirmed cases within a short period of time. The disease has spread to more than 200 countries across the globe with the World Health Organization declaring it as a public health emergency. For instance, according to the World Health Organization Coronavirus Disease (COVID-19) Dashboard (WHO) report, the manifestation of the coronavirus disease (COVID-19) has resulted in more than 118 million infected individuals worldwide as of March 11, 2021. The coronavirus (COVID 19) pandemic and lockdowns in various countries across the globe have impacted the financial status of businesses across all sectors. The private healthcare sector is one the sectors which has been majorly impacted by the COVID-19 pandemic. The lockdown in various countries has created an economic burden on the private healthcare sector. Healthcare providers were facing challenges with regards to manpower, equipment, and other resources to ensure safety in the treatment of patients with other diseases, and has resulted in declining outpatients visits of patients to the hospitals and clinics during the COVID-19. Moreover, the coronavirus pandemic has negatively impacted the development and supply of services, and affected growth of the healthcare businesses of various companies across the globe. This has led to closure of industrial establishments, except manufacturing of essential commodities and disruption in supply chain of products and services. Thus, COVID-19 pandemic has affected the economy in three main ways; 1) by directly affecting the production and demand; 2) by creating disruptions in distribution channels; and 3) through its financial impact on companies and financial markets.
Global Materiovigilance Market: Restraint
The major factors that hinder growth of the global materiovigilance market include increasing complexity of safety regulations and lack of skilled professionals in underdeveloped and emerging economies. For instance, according to data published by Organization for Economic Co-operation and Development (OECD) in 2016, around 50% of formal firms do not find the workforce with the skills they need in Latin America compared to 36% of firms in OECD countries.
Key Players
Key players operating in the market include AssurX, Sparta Systems, Oracle Corporation, Xybion Corporation, Sarjen Systems Pvt. Ltd., MDI Consultants, AB-Cube, QVigilance, Qserve, and ZEINCRO
Share
Share
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients