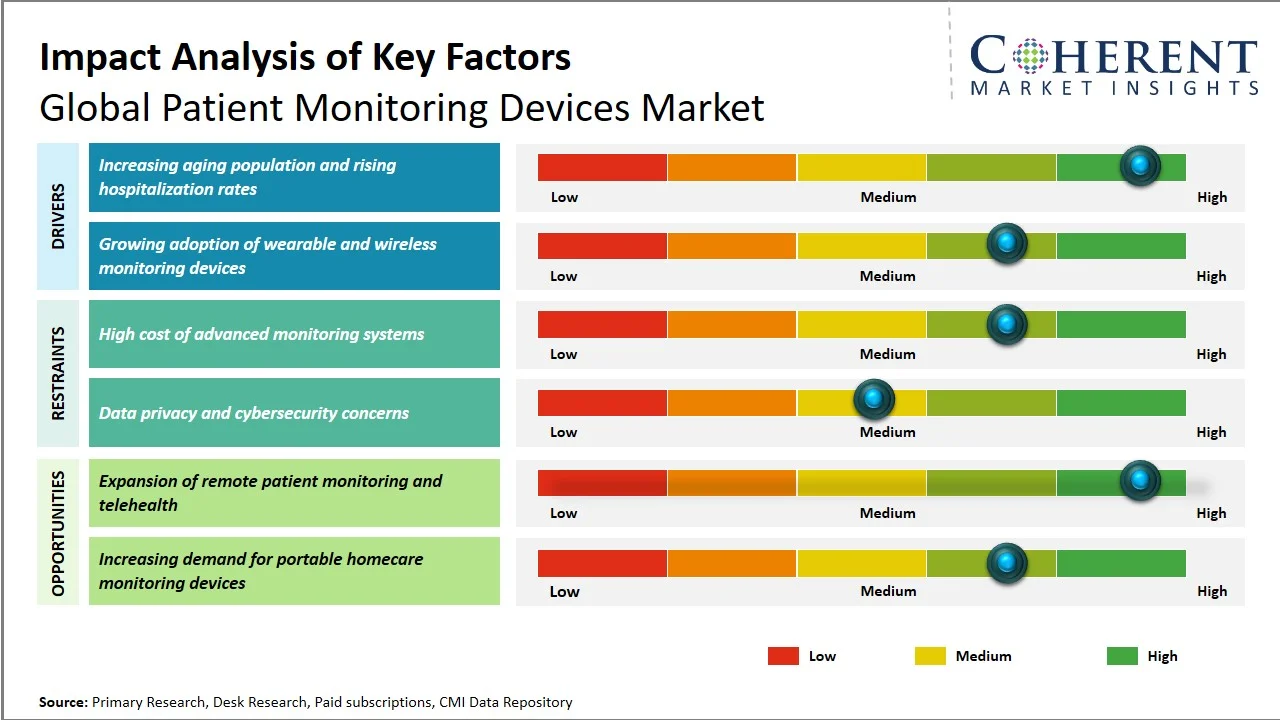
The global patient monitoring devices market is expected to grow from USD 55,400.2 Mn in 2026 to USD 96,080.3 Mn by 2033, registering a compound annual growth rate (CAGR) of 8.2% from 2026 to 2033. The patient monitoring devices market is poised for significant expansion, fueled by the soaring burden of chronic diseases such as cardiovascular disorders, diabetes, obesity, and chronic respiratory disorders, which demand continuous monitoring and sustained management.
According to the World Health Organization, non-communicable diseases (NCDs) have been responsible for over 43 million deaths worldwide, representing 75% of all deaths unrelated to pandemics. The mortality rate due to NCDs was about 18 million deaths per year before the age of 70 years, with about 82% of such early deaths occurring in developing countries.
(Source: World Health Organization)
Additionally, according to the Centers for Disease Control and Prevention, three in four American adults have at least one chronic condition, and over half have two or more chronic conditions. Additionally, over 90% of adults aged 65 and above are affected by at least one chronic disease, reinforcing the need for long-term wearable healthcare devices in the U.S.
(Source: Centers for Disease Control and Prevention)
Respiratory and anesthesia monitors are projected to hold a market share of 24.0% in 2026, driven by the expanding requirement of consistent monitoring of ventilation and real-time tracking of anesthesia and critical care patients during surgeries. The rising prevalence of chronic obstructive pulmonary disease (COPD), sleep apnea, and acute respiratory infections has enhanced the implementation of respiratory monitoring systems in ICUs and emergency departments. Besides, the increasing number of surgeries performed globally coupled with escalating concerns over perioperative patient safety has been catalyzing the uptake of anesthesia monitors to monitor oxygen saturation, carbon dioxide levels and respiration data at enhanced accuracy levels. For instance, according to the World Health Organization, chronic obstructive pulmonary disease (COPD) is the fourth leading cause of death worldwide, causing 3.5 million deaths in 2021, approximately 5% of all global deaths. While, nearly 90% of COPD deaths in those under 70 years of age occur in low- and middle-income countries. (Source: World Health Organization)
To learn more about this report, Request Free Sample
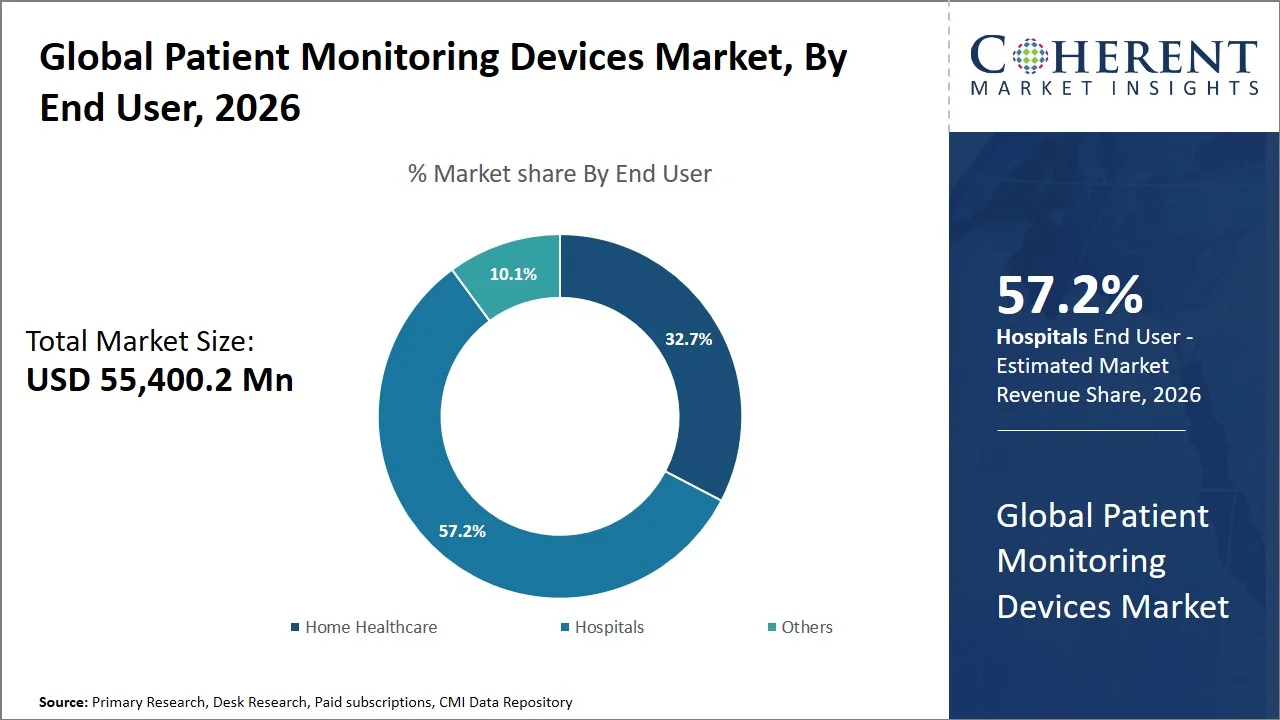
Hospitals are projected to hold a market share of 57.2% in 2026, owing to the critical need for ongoing patient monitoring at the intensive care unit, operation room, emergency department and post-surgery recovery units. The rising number of hospitalizations due to various chronic diseases, trauma cases and advanced surgical processes leads to large-scale installation of multiparameter monitors in hospitals. Furthermore, adoption of centralized monitoring systems linked with electronic medical records in hospitals is fueling workflow and clinical decision-making. Furthermore, the enhanced investment on the smart hospital infrastructure coupled with the adoption of wireless monitoring technologies is expected to facilitate the real-time access of patient data and the speed of medical intervention in the hospital.
For instance, according to the Organization for Economic Co-operation and Development (OECD), health spending from public sources is projected to be twice the average growth in government revenues (2.6% and 1.3% respectively on average across OECD countries). When combined with the need to invest in health systems for future resilience, this could lead to a potential high of 11.8% of GDP being dedicated to health spending by 2040. (Source: Organisation for Economic Co-operation and Development)
|
Current Events |
Description and its Impact |
|
FDA Strengthens Cybersecurity Oversight for Connected Patient Monitoring Devices (July 2025) |
|
|
FDA Expands Digital Health Regulatory Framework for Remote Monitoring Technologies (April 2026) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
(Source: Food and Drug Administration, Food and Drug Administration)
To learn more about this report, Request Free Sample
To learn more about this report, Request Free Sample
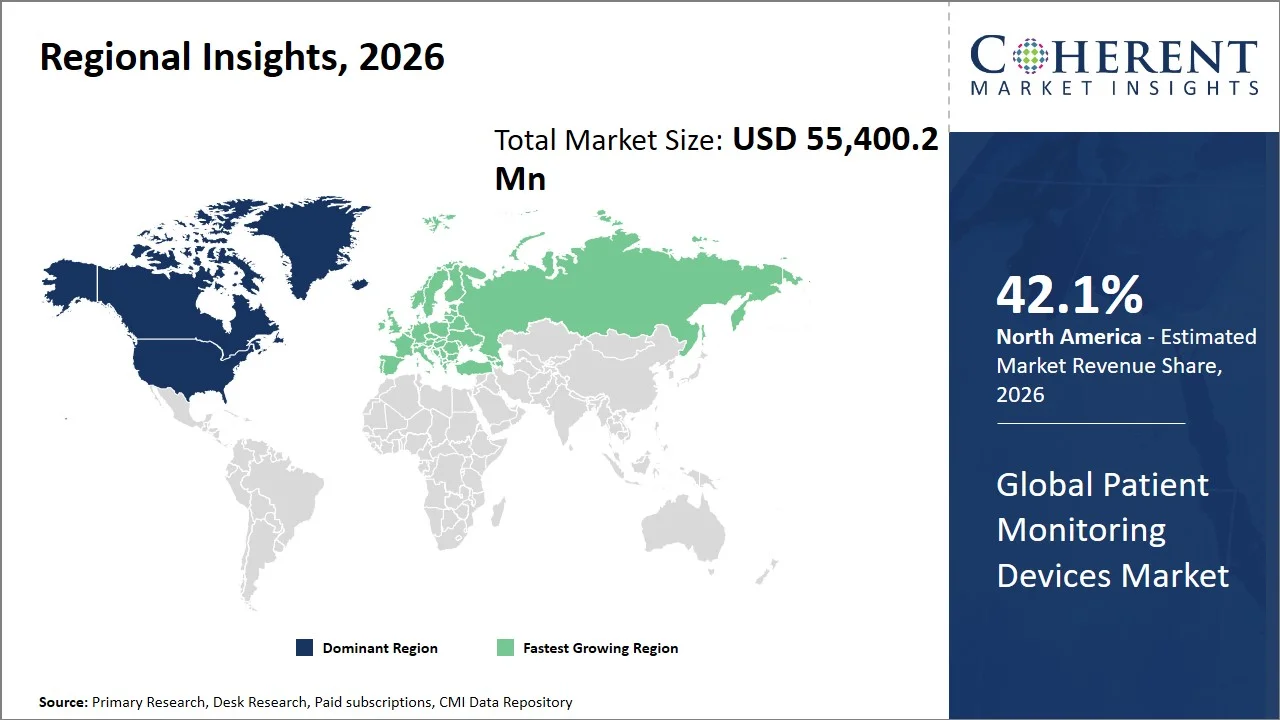
North America leads the global patient monitoring devices market, accounting for an estimated 42.1% share in 2026, owing to the rapid adoption of digital care delivery models and the growing reimbursement coverage for remote physiological monitoring services. The healthcare providers in the U.S. are deploying remote monitoring technologies increasingly to reduce the burden on emergency care, manage patients more efficiently on an outpatient basis and promote value-based care delivery. The region is also witnessing heightened adoption of integrated monitoring systems that communicate with hospital IT infrastructure and telehealth platforms.
Moreover, the region is experiencing a positive reimbursement environment that is leading to widespread adoption of wearable and home-based monitoring devices. For instance, Centers for Medicare & Medicaid Services (CMS) is rapidly increasing reimbursement for remote patient monitoring services across Medicare to foster wider clinical utilization of connected patient monitoring solutions throughout the U.S. Healthcare system. (Source: National Library of Medicine)
The Europe patient monitoring devices market is expected to exhibit the fastest growth with an estimated contribution of 23.2% share to the market in 2026, owing to the expanding usage of digitally connected healthcare facilities and the rising spending on hospital modernization initiatives across the region. Countries like Germany, France, and the U.K. are focusing more on remote patient monitoring and monitoring devices for managing chronic diseases and providing aged care services to ease the burden on the healthcare system and achieve better healthcare results.
Moreover, the increasing emphasis on interoperable healthcare setup and clinical management using evidence-based data is further propelling the adoption of more sophisticated patient monitoring systems throughout hospitals and clinics in Europe. For instance, the European Commission has dedicated USD 6.16 billion (EUR 5.3 billion) from its EU4Health initiative to boosting digital health capacity, resilience of healthcare and the use of technology among EU countries, aiming to further promote the introduction of connected patient monitoring systems throughout the EU region. (Source: European Commission)
The U.S. has become the most innovative and advanced nation for customer adoption in the global patient monitoring devices market, driven by a highly active commercialization climate for digital therapeutics, AI-enabled diagnostic devices, and next-generation biosensor technologies. The country hosts a highly active medical technology ecosystem, driven by close collaborative relationships between healthcare providers, research and academic institutions and device manufacturers through the clinical data management and real-time patient analytics processes.
Moreover, the hospitals in the region are adopting command-center-based patient monitoring models, wherein centralized surveillance mechanisms are used for monitoring and responding to patient needs with greater efficiency. Further, the rising number of U.S. FDA clearances for software-based medical devices and wearable patient monitoring devices is pushing product innovation and faster technology adoption in the U.S.
The U.K. is an attractive market for patient monitoring devices on account of the broadening integration of virtual ward initiatives and digitally enabled community-care systems into the National Health Service. The country is experiencing widespread clinical and administrative deployment of remote monitoring systems to accommodate patient overflow, alleviate inpatient occupancy pressures and enable more efficient post-discharge patient management. Rising adoption of centralized digitally managed care pathways along with AI enabled patient triaging solutions is further augmenting demand for interconnected monitoring systems within hospital and community healthcare environments.
Furthermore, the country's unrelenting emphasis on reducing treatment delays and expanding long-term management of high-risk patients will continue to lead to more investment in scalable patient monitoring frameworks within hospital and outpatient care systems.
China is becoming a significant growth center for the patient monitoring devices market on account of rapid growth in national medical device manufacturing capabilities and widespread adoption of smart healthcare concepts in urban hospital networks. The proliferation of advanced AI-enabled patient monitoring systems, cloud-connected hospital networks and intelligent ward management platforms in Chinese hospitals are supporting improving overall quality and patient throughput.
Furthermore, local production of medical devices and healthcare technologies is reducing prices and improving competitiveness of domestic product suppliers, which is in turn encouraging broader adoption of affordable patient monitoring devices in secondary and tertiary level hospitals. The country's growing emphasis on hospital digitalization and an expanding adoption of intelligent healthcare systems will further strengthen the growth potential of the market in China.
Germany leads the Europe market for patient monitoring devices, benefiting from its pioneering hospital engineering infrastructure and extensive, early adoption of digitally interconnected clinical delivery systems at public and private care providers. The German medical technology manufacturing network has quickly adopted the latest bedside monitoring and telemetry technologies, as well as advanced intensive care monitoring systems. Moreover, hospital operators have moved to adopt data-supported clinical operational systems and interoperable monitoring networks to gain more efficiency in high-acuity care provider settings.
The Japan patient monitoring devices market is gradually evolving in response to the widespread adoption of miniaturized and automated monitoring devices that cater to the dynamic requirements of the high-efficiency clinical settings. The country's healthcare providers are increasingly installing smart bedside monitoring systems, automated vital signs measuring devices and integrated nursing support technology to provide better patient management under soaring clinical workload pressures. The ongoing technological advances in robotic and sensor-based medical devices systems, especially in the sphere of healthcare in Japan is spurring the development of future lines of patient monitoring products. Wide scale acceptance of sophisticated monitoring systems in rehabilitation clinics, specialist medical centers and long-term care facilities are also promoting the market in Japan.
|
Region |
Authority & Classification |
Approval Pathway |
Key Regulatory Insight |
|
North America |
U.S. FDA – Class II/III medical devices; Health Canada Medical Devices Regulations |
FDA 510(k), PMA, Health Canada MDL approval |
Strong emphasis on cybersecurity, interoperability, and real-time remote patient monitoring compliance. |
|
Europe |
European Medicines Agency (EMA) & EU MDR 2017/745; CE Classification (Class IIa/IIb) |
CE Marking through Notified Bodies under MDR |
EU MDR mandates stringent clinical evaluation, post-market surveillance, and data safety requirements. |
|
Asia Pacific |
China NMPA, Japan PMDA, India CDSCO, Australia TGA |
Country-specific registration and clinical review approvals |
Regulatory frameworks are rapidly evolving with increased focus on digital health and imported device quality standards. |
|
Latin America |
Brazil ANVISA, Mexico COFEPRIS |
Local registration, GMP certification, and technical dossier review |
Brazil remains the key regulatory hub with stringent documentation and local compliance requirements. |
|
Middle East & Africa |
Saudi FDA (SFDA), UAE MOHAP, South Africa SAHPRA |
Import licensing, device registration, and conformity assessment |
Growing harmonization with international standards such as FDA and CE certifications to accelerate device adoption. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The expansion of telehealth and remote patient monitoring services provides lucrative opportunities for the patient monitoring devices market, by improving access and accelerating adoption of connected and home-based monitoring devices for providing virtual care. Nowadays, hospitals are widely deploying wireless ECG monitors, pulse oximeters, blood pressure monitors, and cloud-enabled remote monitoring platforms to enhance chronic disease management and avoid costly hospital admissions. Increased adoption of telehealth programs within conventional healthcare framework is thus expected to promote the demand for advanced, integrated patient monitoring systems that facilitate real-time clinical care, and promote ongoing patient engagement.
For instance, data collected by the National Center for Health Statistics (NCHS), shows that in 2021, 80.5% of physicians in office-based settings used telemedicine, underscoring the accelerated growth in digital health infrastructure and the sustained global need for remote health monitoring solutions. (Source: National Center for Health Statistics)

To learn more about this report, Request Free Sample
The global patient monitoring devices market is highly competitive and companies are focusing on advancements in technology, improvements in connectivity and increased capabilities for remote healthcare as ways of maintaining or increasing market share. Market players are looking at a variety of ways to develop new monitor offerings, including investing in systems that are able to monitor patients using AI-enabled, cloud-enabled surveillance platforms, and portable wireless monitors that can deliver real-time information for clinical decision support. Furthermore, devices that are able to integrate seamlessly with hospital information systems are becoming more prevalent as healthcare institutions seek to improve workflow processes. This is all complemented by collaborations with healthcare providers and digital health companies that will allow for the continued creation of new and improved patient monitors for use in hospitals, ambulatory care centers and home health settings. Key focus areas include:
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 55,400.2 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 8.2% | 2033 Value Projection: | USD 96,080.3 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Koninklijke Philips N.V., Medtronic plc, GE HealthCare Technologies Inc., Abbott Laboratories, Siemens Healthineers AG, Nihon Kohden Corporation, Masimo Corporation, Mindray Medical International Limited, Drägerwerk AG & Co. KGaA, and Baxter International Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients