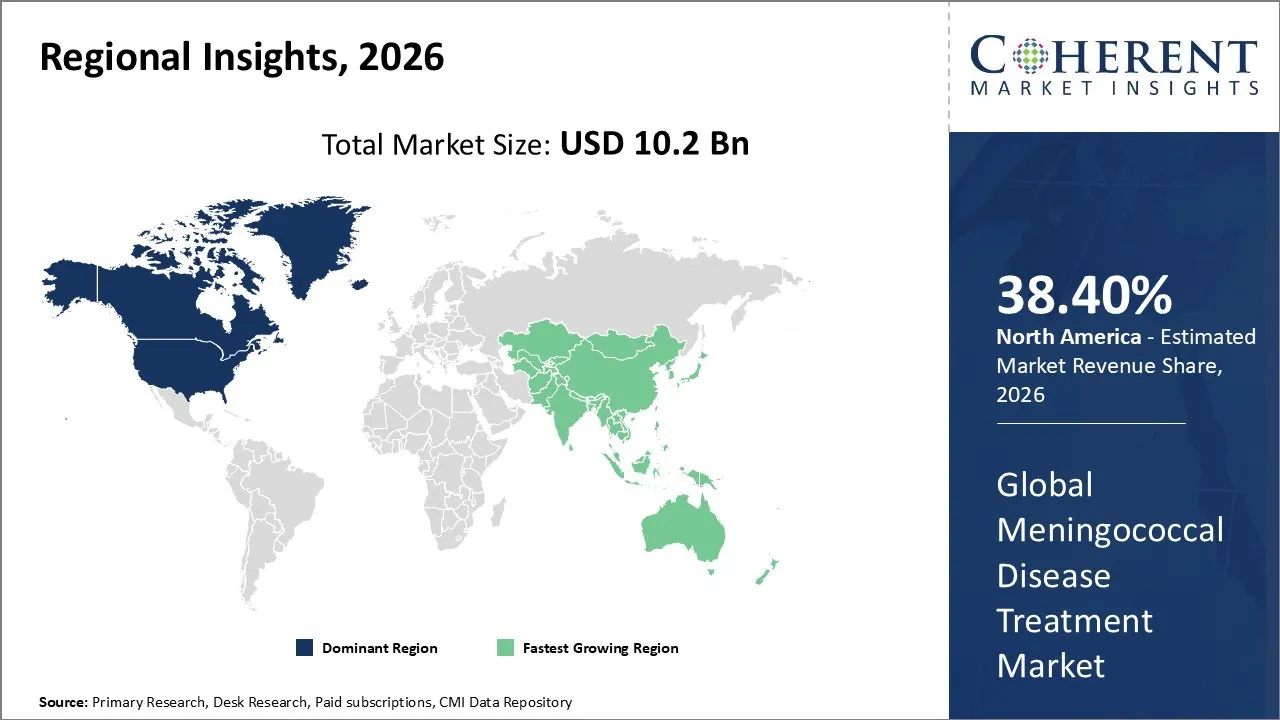
The Meningococcal Disease Treatment Market is estimated to be valued at USD 10.2 Bn in 2026 and is expected to reach USD 28.3 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 13.2% from 2026 to 2033.
The Meningococcal Disease Treatment Market delivers therapies targeting infections caused by Neisseria meningitidis, including antibiotics and vaccines. Growing awareness of meningococcal disease, expanding vaccination programs, and the urgent need to treat severe cases are driving demand. Hospital pharmacies and injectable formulations lead distribution, meeting acute care requirements. Public health initiatives, advanced healthcare infrastructure, and ongoing research across North America and other regions actively support market growth, ensuring healthcare providers can promptly deliver preventive and therapeutic solutions to diverse populations.
|
Current Events |
Description and its impact |
|
Global Vaccine Development and Technological Advancements |
|
|
Rising Meningococcal Disease Incidence in Sub-Saharan Africa |
|
|
Impact of Global Supply Chain Disruptions and Raw Material Shortages |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Injectable expected to hold largest market share of 51.3% in 2026. The injectable segment in the Meningococcal Disease Treatment Market grows as clinicians urgently deliver therapies for severe meningococcal infections. Hospitals and emergency care units rely on injectable antibiotics and vaccines for their rapid systemic action and accurate dosing. Advanced diagnostic tools enable healthcare providers to start targeted parenteral treatments quickly, while outbreak control and prophylactic measures further increase usage. Ready-to-use formulations, robust cold-chain systems, and adherence to clinical protocols enhance the role of injectables in managing meningococcal disease effectively. For instance, in January 2026, SK bioscience collaborated with Sanofi Korea and introduced the quadrivalent meningococcal conjugate vaccine “MenQuadfi” in Korea. The vaccine protects individuals aged 6 weeks to 55 years against invasive meningococcal disease caused by serogroups A, C, W, and Y, with SK bioscience managing domestic distribution and supply.
Antibiotics hold the largest market share of 52.2% in 2026. The Meningococcal Disease Treatment Market relies on antibiotics to rapidly and effectively manage severe infections caused by Neisseria meningitidis. Widespread outbreaks, hospital protocols, and early intervention strategies drive clinicians to prioritize antibiotic therapy and prevent complications. Advancements in diagnostics allow healthcare providers to deliver targeted treatment, while prophylactic use among close contacts increases utilization. Growing awareness among medical professionals, together with established clinical guidelines and emergency care practices, positions antibiotics as a central tool in treating and preventing meningococcal disease.
Hospital Pharmacy acquired the prominent market share of 38.6% in 2026. The hospital pharmacy segment in the Meningococcal Disease Treatment Market expands as hospitals act as central hubs for managing severe meningococcal infections. Hospital pharmacies provide immediate access to essential antibiotics and injectable vaccines, enabling rapid treatment in emergency and inpatient care. By integrating with clinical protocols, antimicrobial stewardship programs, and specialized storage systems, they enhance efficiency and safety. They also support outbreak response and prophylactic measures, while close collaboration with healthcare professionals ensures timely and effective delivery of therapies to patients.
To learn more about this report, Download Free Sample
North America dominates the overall market with an estimated share of 38.4% in 2026. Strong vaccination initiatives and proactive public health strategies drive the North America Meningococcal Disease Treatment Market, focusing on adolescents, high-risk adults, and outbreak-prone communities. Advanced healthcare infrastructure and established hospital networks enable clinicians to diagnose and treat patients quickly with antibiotics and injectable vaccines. Continuous research and development fuels innovation in next-generation vaccines and therapies. Growing awareness among healthcare professionals and patients, along with efficient hospital pharmacy distribution, allows the region to respond effectively to both sporadic cases and disease outbreaks. For instance, the National Foundation for Infectious Diseases (NFID) and DoSomething.org launched the “Complete What’s Missing” program to provide young people with reliable information about meningococcal disease and promote prevention through vaccination.
Governments and healthcare authorities in the Asia Pacific actively expand the Meningococcal Disease Treatment Market by intensifying vaccination campaigns and implementing outbreak control measures, especially in high-risk areas. Clinicians and communities increasingly recognize the importance of timely diagnosis and treatment with antibiotics and injectable vaccines. Expanding healthcare infrastructure, strengthened hospital networks, and the use of advanced diagnostic tools enable better patient management. Public health initiatives, paired with strategic hospital pharmacy distribution, allow the region to respond efficiently to sporadic infections and sudden outbreaks, enhancing disease control and prevention.
Comprehensive immunization programs targeting adolescents, college students, and high-risk groups actively drive the United States Meningococcal Disease Treatment Market. Hospitals and emergency care centers rapidly administer antibiotics and injectable vaccines to manage severe infections effectively. Clinicians use advanced diagnostic technologies to deliver timely and precise treatments, while ongoing research promotes innovation in next-generation vaccines and therapies. Strong public health policies, awareness campaigns, and efficient hospital pharmacy distribution enable the healthcare system to respond quickly to both sporadic cases and outbreak situations nationwide.
Healthcare authorities in Japan actively drive the Meningococcal Disease Treatment Market by strengthening preventive strategies and vaccination programs, especially for high-risk and travel-exposed populations. Hospitals and clinics rapidly administer antibiotics and injectable vaccines to manage severe infections effectively. Clinicians use advanced diagnostic tools to make timely and accurate treatment decisions, while ongoing research advances innovative therapies and next-generation vaccines. Coordinated public health campaigns, hospital pharmacy distribution, and adherence to clinical guidelines enable Japan to control sporadic cases and respond efficiently to potential outbreaks.
Healthcare systems globally are emphasizing vaccination programs targeting adolescents, high-risk adults, and travelers. Governments and public health authorities are expanding campaigns to increase immunization coverage and reduce disease incidence. End-users increasingly prefer preventive strategies over reactive treatment, driving demand for conjugate and polysaccharide vaccines. Ongoing research in next-generation vaccines aims to cover multiple serogroups, simplify dosing schedules, and enhance long-term immunity, reflecting a proactive shift toward disease prevention rather than solely relying on therapeutic interventions.
Injectable antibiotics and vaccines dominate the market due to their rapid systemic action, precise dosing, and suitability for emergency and inpatient care. Hospitals and clinicians favor parenteral administration for severe meningococcal infections to ensure timely intervention. The trend includes ready-to-use formulations, improved cold-chain storage, and integration with clinical protocols, which enhance safety and efficiency. Increasing awareness among healthcare professionals about early antibiotic administration further reinforces the role of injectables as a first-line approach in acute disease management.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 10.2 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 13.2% | 2033 Value Projection: | USD 28.3 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
GlaxoSmithKline Plc., Novartis AG, Sanofi Pasteur Inc., Wockhardt Ltd., Kent Pharmaceuticals, Stravencon Limited., F. Hoffmann-La Roche Ltd., Athlone Laboratories, and Pfizer Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients