The Global Mercury-Free Dental Implants Materials Market is anticipated to grow at a CAGR of 12.8% with USD 2.01 Bn share in 2026 and is expected to reach USD 4.68 Bn in 2033. This growth in the market is driven by the growing shift toward biocompatible, toxin-free dental materials amid stricter regulations on mercury usage and rising demand for aesthetic dental solutions.
In 2026, titanium held the leading segment in this market because of its superior osseointegration, corrosion resistance, and mechanical reliability over time, as well as its extensive clinical validation. According to an article published by Nuclear Resistance of titanium, titanium has excellent resistance to corrosion.
The two key motivators driving the growth of the titanium segment are as follows: demonstrable biocompatibility of titanium and predictable bone integration are reducing clinical risks and making them the preferred materials for routine implantology, and titanium and titanium-alloy manufacturers are developing optimum-performing hydrophilic surface systems with smaller diameters and digitally integrated workflows to facilitate the use of such systems for immediate-placement cases and those with reduced-bone in a less complicated manner.
At IDS 2025, Straumann introduced the iEXCEL system, a four-implant design based on Roxolid, an alloy that is 85% titanium and 15% zirconium. On February 18, 2026, Reuters reported that Straumann had CHF 2.61 billion in sales in 2025, an 8.9% increase in organic revenue, indicating the solid growth of advanced titanium-based implant systems.
To learn more about this report, Request Free Sample
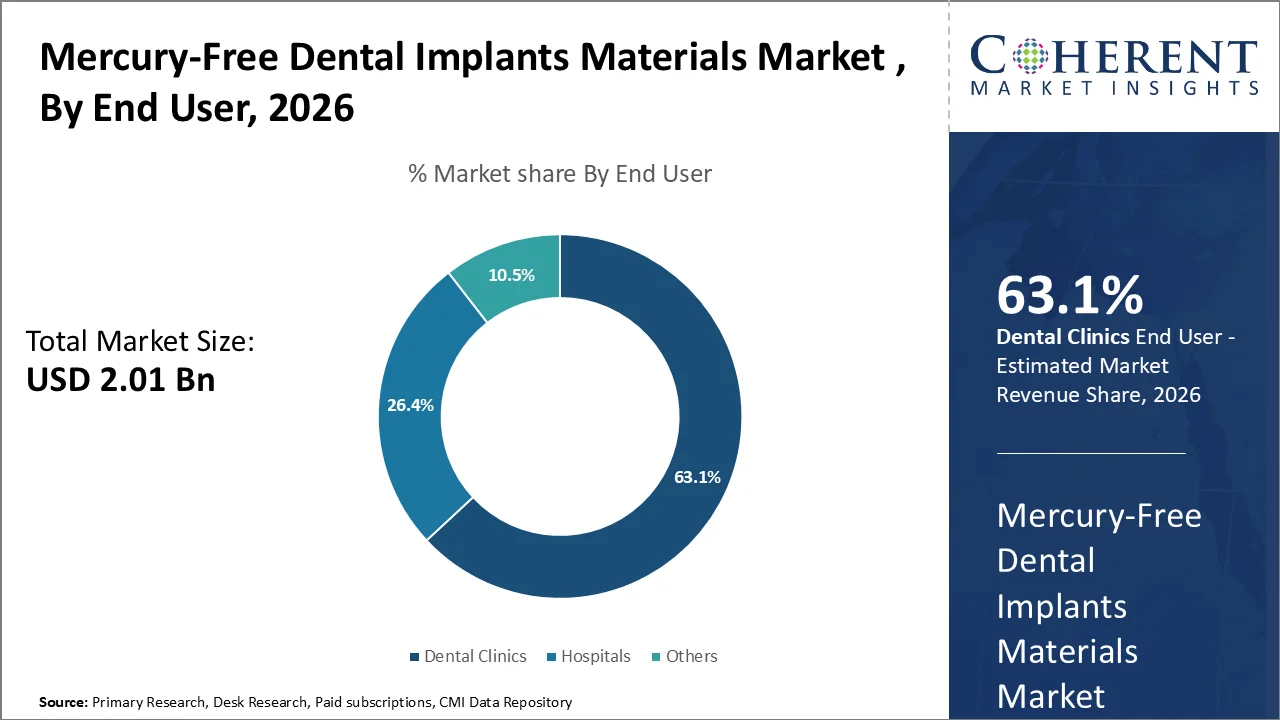
Mercury-Free Dental Implants Materials Market is dominated by Dental Clinics in 2026 with 63.1% share. Most consultations for implants, placement, loading of the prosthesis and follow-up for surgery will be done in the office. According to the dental economy page on the ADA's website regarding Q1 2026, year over year, there is a 4 percent growth in consumer spending on dental care through January 2026, with a total US national dental spending at US$189 Billion in 2024. There has also been an estimated 45 percent of the US population having received dental care in the last 12 months, further indicating the significance and size of the scope of clinic-based care. (ADA)
Outpatient dental clinics have become preferred by patients due to their ability to provide faster implant consultations, coordinated implant treatment planning and implant restorations in one location. In addition, dental clinics are investing in technology such as digital imaging, guided workflows, and specialist-led implant programs, resulting in increased efficiency for these procedures, an expanded candidate pool for implant surgery, and increased chairside conversions from consultation to implant surgery.
In March 2026, a press release from the Aspen Group reported more than 5.2 million patients have been treated by Aspen Dental; the company also added 21 locations to its practice by the end of 2025. Another clinic that reported 27,500 implant patients- ClearChoice Dental Implant Centers- expanded its practice into three additional markets. Aspen Dental has completed a rollout of VideaAI across its 1,100+ practice locations, demonstrating the scale of opportunities in clinic-based care for dental implants. (AspenDental)
The dental implant market continues to innovate by moving from merely replacing existing materials to designing biomaterials that outperform existing options. Zirconia implants continue to increase their commercial importance following a systematic review to the PubMed index that showed cumulative survival rates of 95.1% at 10 years. A 5-year survival/success study on narrow-diameter titanium-zirconium dental implant materials achieved 100% survivorship and success, indicating improved opportunities for less traumatic implants. Simultaneously, the World Health Organization's March 2026 update of the International Classification of Diseases formally accepts mercury-free dental materials and less invasive approaches to oral healthcare, providing further momentum to promote innovation through research and development of ceramic implant materials, advanced titanium-zirconium alloys, and clinical platforms that promote sustainability.
|
Current Events |
Description and Its Impact |
|
WHO issues global guideline endorsing mercury-free dental materials and less invasive oral care |
|
|
EU MDR transition framework continues to tighten compliance expectations for implantable devices in 2026 |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
To learn more about this report, Request Free Sample
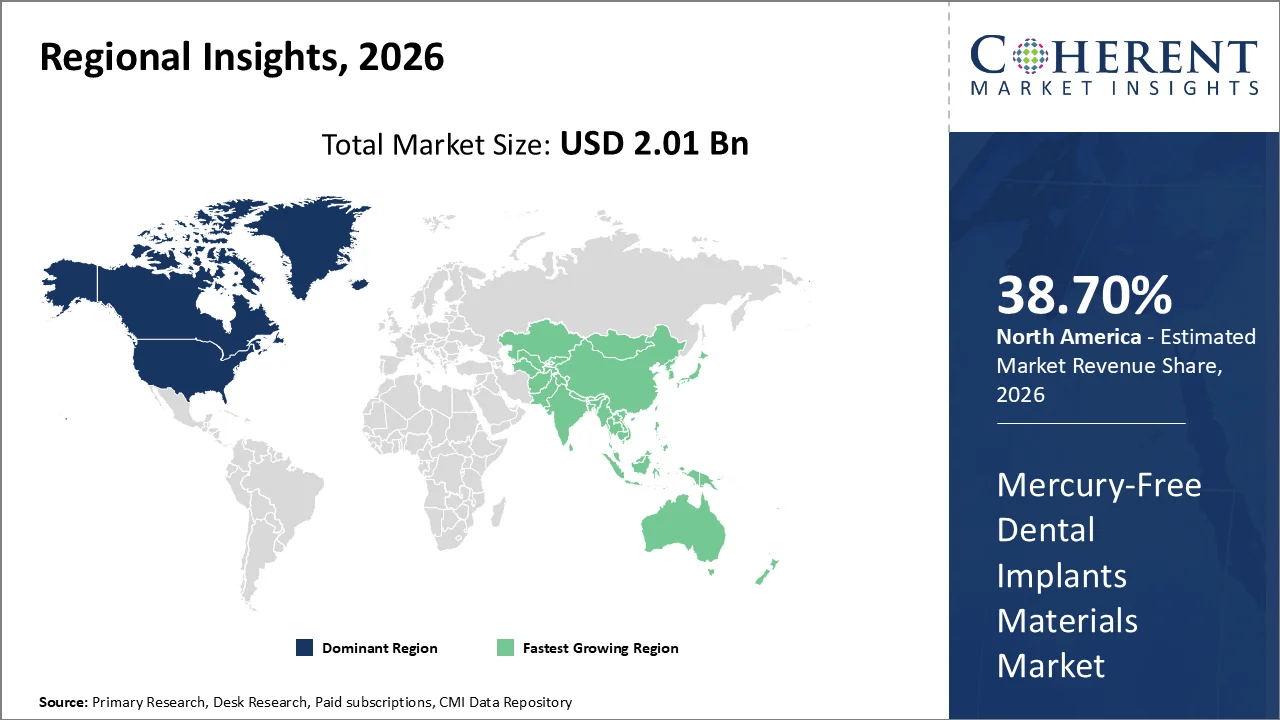
North America is anticipated to have the largest share (38.70%) of the total global market, with a proportionate share of purchases driven primarily by an increased awareness of dental implant procedures combined with a high density of specialists and patients with higher disposable incomes for that type of service. The U.S. remains the key geographic driver; as stated by the American Academy of Implant Dentistry, more than three million individuals have dental implants within the U.S., which is increasing by approximately 500,000 individuals per year, and according to the American Dental Association, the total annual dental expenditure in the U.S. will be US$189 billion by 2024.
Asia Pacific represents the region with the most rapid growth in market share and is projected to account for 14.8% of the total global market by 2026 based on the high number of older individuals, the increasing incidence of tooth-loss, and improving access to restorative dental care within this region. According to ESCAP, the number of people aged 60 years and older in the Asia Pacific region is projected to reach almost 1.3 billion (25 per cent of the total population) by2040. In addition, W.H.O. reports that back in 2019, there were about 92 million adult cases of edentulism in the Western Pacific alone, and almost 960 million are currently living with untreated oral disease within the Western Pacific region.
In North America, the U.S. is the biggest opportunity at a country level due to established patient, practitioner and implant technologies, specifically with respect to patient use (over 3 million people in the U.S. have an implant), have a specialist network of 50,000+ specialists and market expenditures ($189 billion) which indicates established procedural and commercial support for high-end, mercury-free implant products.
The largest opportunity in the Asia Pacific region is India due to long-term growth potential linked to an existing high oral disease burden coupled with an increasing clinical and oral health treatment infrastructure. For instance, as shown in the WHO's India Oral Health Country Profile (2019), 28.8% of Indians suffer from untreated caries on their permanent teeth, 21.8% of Indian adults aged 20+ have severe periodontal disease, 4.0% of Indian adults aged 20+ are edentulous, there were 271,744 dentists in India as of 2019; therefore, the combination of these factors represents a large population requiring treatment and an expanding base for adoption of titanium and zirconia implants within both private and public sector practice.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 2.01 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 12.8% | 2033 Value Projection: | USD 4.68 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Dentsply Sirona, Institut Straumann AG, Coltene Group, VITA Zahnfabrik, Upcera Dental Co. Ltd., Aidite Technology Co. Ltd., Huge Dental Material Co., Ltd., Kuraray Noritake Dental Inc., Zirkonzahn GmBH, 3M, VOCO GmBH, Danaher Corporation, Ivoclar Vivadent AG, Mitsui Chemicals, Inc., GC Corporation, Ultradent Products Inc., and SHOFU Dental Corporation. |
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Major companies are Dentsply Sirona, Institut Straumann AG, Coltene Group, VITA Zahnfabrik, Upcera Dental Co. Ltd., Aidite Technology Co. Ltd., Huge Dental Material Co., Ltd., Kuraray Noritake Dental Inc., Zirkonzahn GmBH, 3M, VOCO GmBH, Danaher Corporation, Ivoclar Vivadent AG, Mitsui Chemicals, Inc., GC Corporation, Ultradent Products Inc., and SHOFU Dental Corporation.
The IDEXCEL Implant System by Straumann has been introduced at the IDS 2025 event in Cologne on March 25th, 2025. This next-generation implant platform will consist of four different implant types, one surgical kit and one prosthetic connection. Additionally, Roxolid and SLActive will be used to differentiate between premium materials and efficiency in workflow. (Straumann)
Share
Share
Manisha Vibhute is a consultant with over 5 years of experience in market research and consulting. With a strong understanding of market dynamics, Manisha assists clients in developing effective market access strategies. She helps medical device companies navigate pricing, reimbursement, and regulatory pathways to ensure successful product launches.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients