Microneedle Flu Vaccine Market is estimated to be valued at USD 1,862.9 Mn in 2026 and is expected to reach USD 2,894.9 Mn in 2033, exhibiting a compound annual growth rate (CAGR) of 6.5% from 2026 to 2033.
Analysts’ Views on Global Microneedle Flu Vaccine Market:
Rising demand for microneedle flu vaccines for the treatment of influenza is expected to drive the market growth over the forecast period. For instance, in July 2020, according to a report published by ScienceDirect, an open-source journal, a new product VaxiPatch, which is a novel vaccination system comprised of subunit antigens, adjuvants, and microneedle skin delivery for the treatment of influenza was developed by Verndari, Inc., a vaccine development company.
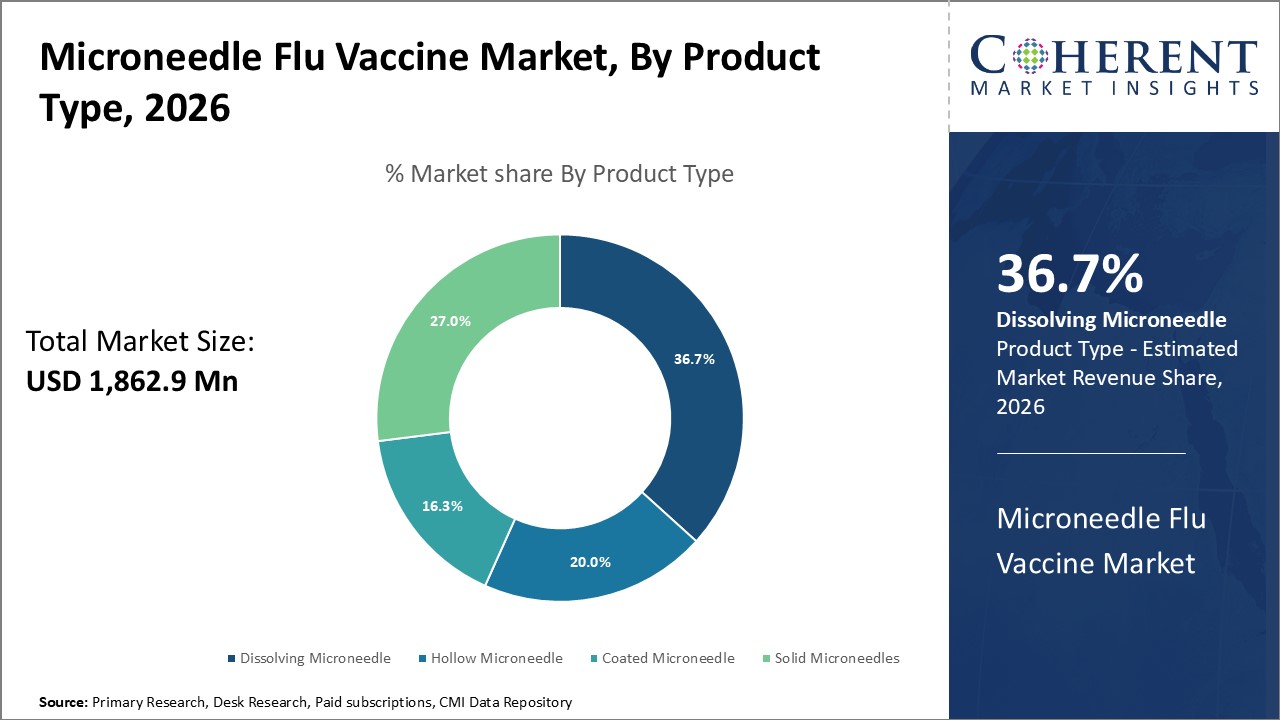
Figure 1. Global Microneedle Flu Vaccine Market Share (%), by Product Type, 2026
To learn more about this report, Request Free Sample
Global Microneedle Flu Vaccine Market- Driver
Increasing prevalence of influenza
The increasing prevalence of influenza which requires microneedle flu vaccines for its treatment is expected to drive the market growth over the forecast period. For instance, in October 2022, a report published on WebMD LLC., a full-service Internet healthcare portal, stated that an estimated 5,000 to 14,000 deaths have been attributed to influenza since October 2021.
Microneedle Flu Vaccine Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1,862.9 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 6.5% | 2033 Value Projection: | USD 2,894.9 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Sanofi, GC Pharma, Harro Höfliger, CosMED Pharmaceutical Co.Ltd., Micron Biomedical, Inc., LTS Lohmann Therapie-Systeme AG, Micralyne, Inc., Sorrento Therapeutics, Inc., TSRL, Inc., QUADMEDICINE, Raphas Co., Ltd., Cutanos GmbH, 3M, NanoPass, Corium, Inc., BD, FluGen, Inc., MICRODERMICS INC |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Increasing launch of new flu prevention campaigns and initiatives by healthcare organizations
The launch of new flu prevention campaigns and initiatives by healthcare organizations is expected to drive demand for influenza vaccines and develop effective vaccination delivery systems. For instance, in October 2021, The Centers for Disease Control and Prevention (CDC) and the National Foundation for Infectious Diseases (NFID), along with other public health and medical organizations, jointly announced the opening of the 2020-2021 flu vaccination season, highlighting the importance of vaccination and recommending that everyone six months and older get vaccinated against flu once a year.
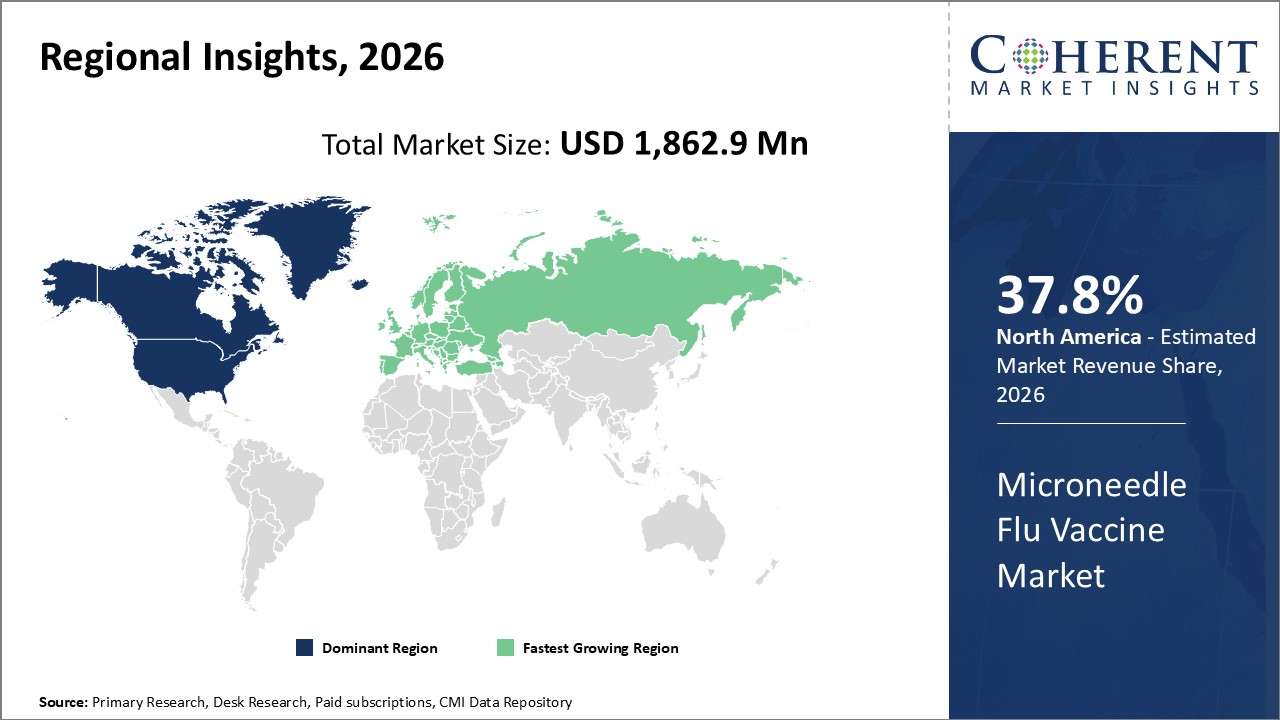
Figure 2. Global Microneedle Flu Vaccine Market Share (%), by Region, 2026
To learn more about this report, Request Free Sample
Global Microneedle Flu Vaccine Market- Regional Analysis
Among region, North America is expected to dominate the market over the forecast period. This is attributed to the increasing adoption of organic growth strategies such as expansion by key players in the region to expand their product portfolio which is expected to drive the market growth over the forecast period. For instance, in October 2021, Vaxess Technologies Inc., engaged in developing biomedical innovations with a focus on improving access to vaccines, announced the grand opening of a Good Manufacturing Practice (GMP) manufacturing facility, a GMP Facility is a production facility or a clinical trial materials pilot plant for the manufacture of pharmaceutical products. Vaxess Technologies Inc. is focused on developing a pipeline of MIMIX therapies.
The Europe region is expected to be the second largest region over the forecast period, owing to increasing research and development activities by key players in the region to expand product portfolio, which is expected to drive the market growth over the forecast period. For instance, in October 2021, LTS Lohmann Therapie-Systeme AG, a developer and manufacturer of transdermal systems and oral thin films, announced that it has expanded the research and development area for microneedles.
According to the World Health Organization (WHO), 40 million people were affected globally till October 10, 2022.
Global Microneedle Flu Vaccine Market Segmentation:
The global microneedle flu vaccine market report is segmented into product type, vaccine type, flu type, and region
Based on Product Type, the market is segmented into solid microneedles, hollow microneedles, coated microneedles, and dissolving microneedles. Out of which, the dissolving microneedles segment is expected to dominate the global microneedle flu vaccine market during the forecast period and this is attributed to an increase in the product launches by key players in the market.
Based on Vaccine Type, the market is segmented into trivalent flu vaccine, and quadrivalent flu vaccine. Out of which, the trivalent flu vaccine segment is expected to dominate the market over the forecast period and this is due to an increase in the use of trivalent flu vaccines for the treatment of influenza flu.
Based on Flu Type, the microneedle flu vaccine market is segmented into influenza A (H1N1, H3N1), and influenza B. Influenza A segment is expected to dominate in the market over the forecast period and this is attributed to the increase in the usage of microneedle flu vaccine to treat influenza.
Based on Region, the microneedle flu vaccine market is segmented into North America, Latin America, Europe, Middle East, Asia Pacific, and Africa. Of which, North America segment is expected to dominate the market over the forecast period and this is attributed to the increasing inorganic growth strategies such as collaborations by key players in the region
Among all segmentation, product type segment has the highest potential due to the increasing adoption of research and development activities. For instance, in April 2021, a report published by National Center for Biotechnology Information (NCBI), stated that according to research, microneedles are a new generation of vaccine delivery systems.
Global Microneedle Flu Vaccine Market Cross Sectional Analysis:
In flu type segment, influenza A segment held a dominant position in North America region due to the increasing prevalence of influenza in the North America region which will expand the use of microneedle flu vaccines for treatment. For instance, on January 12, 2023, an article published by World Health Organization (WHO), stated that research estimates that 100% of deaths in children under 5 years of age with influenza-related lower respiratory tract infections are found in developing countries.
Global Microneedle Flu Vaccine Market: Key Developments
Global Microneedle Flu Vaccine Market: Restraint
The disadvantages of the microneedle flu vaccine
The major factors that can hamper the growth of the global microneedle flu vaccine market over the forecast period include disadvantages like it is time-consuming, it may result in scarring of the skin, etc. For instance, in December 2022, The American Society for Microbiology, an open-access journal, published an article stating that the amount of time it takes to dissolve a microneedle may range from minutes to several hours, which is a major disadvantage of using microneedles.
Therefore, overcoming the disadvantages associated with the use of microneedle flu vaccines may help in increasing the wider application of microneedle flu vaccines and can tackle these restraints.
Global Microneedle Flu Vaccine Market: Key Players
Major players operating in the global microneedle flu vaccine market include Sanofi, GC Pharma, Harro Höfliger, CosMED Pharmaceutical Co.Ltd., Micron Biomedical, Inc., LTS Lohmann Therapie-Systeme AG, Micralyne, Inc., Sorrento Therapeutics, Inc., TSRL, Inc., QUADMEDICINE, Raphas Co., Ltd., Cutanos GmbH, 3M, NanoPass, Corium, Inc., BD, FluGen, Inc., MICRODERMICS INC
Definition: A microneedle flu vaccine makes use of different types of microneedles for the delivery of drugs and vaccines and is mainly used in the treatment of influenza
Share
Share
Abhijeet Kale is a results-driven management consultant with five years of specialized experience in the biotech and clinical diagnostics sectors. With a strong background in scientific research and business strategy, Abhijeet helps organizations identify potential revenue pockets, and in turn helping clients with market entry strategies. He assists clients in developing robust strategies for navigating FDA and EMA requirements.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients