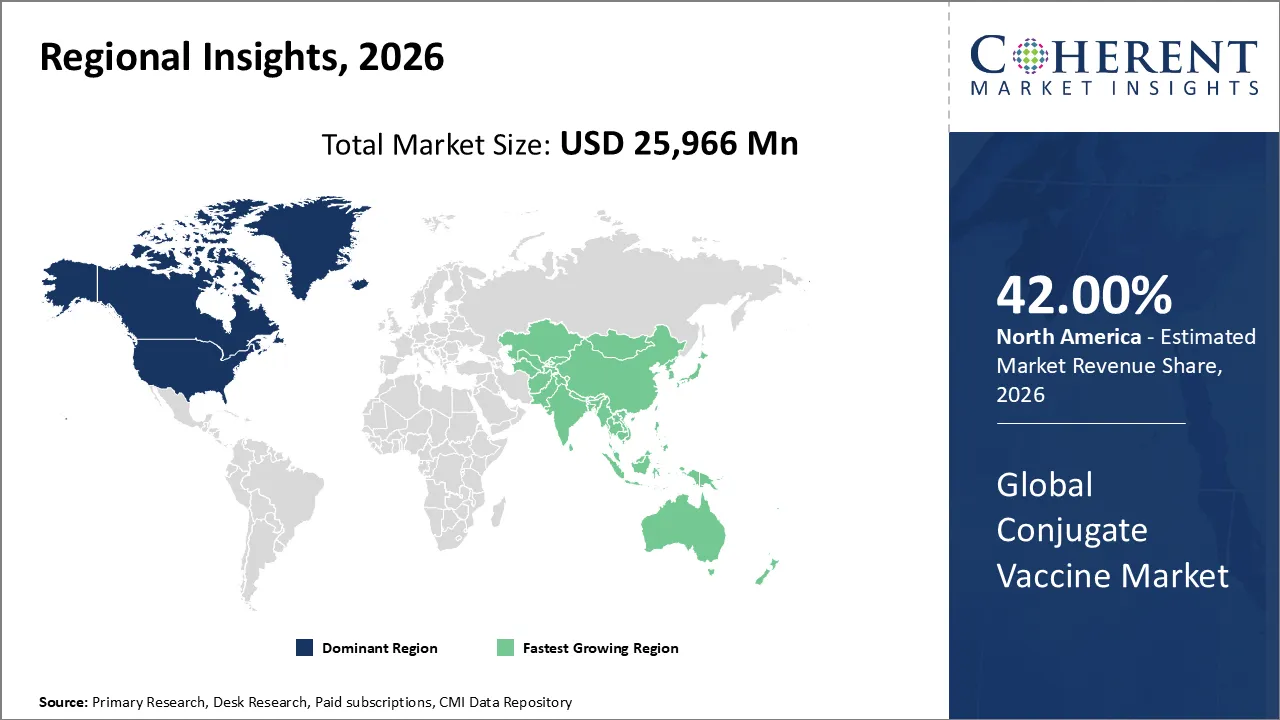
Conjugate vaccine market is estimated to be valued at USD 25,966 Mn in 2026 and is expected to reach USD 49,220 Mn by 2033, exhibiting a compound annual growth rate (CAGR) of 9.6% from 2026 to 2033.
Increasing awareness of vaccine-preventable diseases and the development of immunization programs by the government are expected to boost the market for conjugate vaccines worldwide. Conjugate vaccines, which help boost the immune system to fight infections like bacteria and viruses, are gaining popularity as vaccine candidates, particularly for protecting against diseases like pneumococcal disease, meningococcal disease, influenza, and typhoid fever. Pharmaceuticals are now developing multivalent conjugate vaccines to combat infectious diseases and engage parents and adults, thereby increasing market growth in the conjugate vaccine industry.
|
Current Events |
Description and its impact |
|
China's Vaccine Manufacturing Expansion |
|
|
Supply Chain Vulnerabilities and Raw Material Constraints |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
By the type of product, Multivalent Conjugate Vaccine segment will capture a major market share of 61% by 2026. Multivalent vaccines protect against several strains within one dose, reducing the numbers of injections and helping to increase patient compliance. The breadth of their coverage makes them favored options in national immunization programs, especially for pneumococcal and meningococcal diseases.
For instance, in May 2025, Vietnam Vaccine Joint Stock Company (VNVC) introduced and started using the highly advanced 20 valent pneumococcal vaccine in Vietnam, which is a major breakthrough in the development of broad coverage conjugate vaccines against 20 serotypes of pneumococcal diseases.
Based on disease indication, the Pneumococcal segment holds the highest market share of 54% in the year 2026. Pneumococcal diseases are a serious health hazard, especially in children below the age of 5 and the elderly population. The market share of conjugate vaccines for pneumococcal diseases has been growing because of widespread vaccination campaigns in parts of North America, Europe, and Asia.
For instance, in August 2025, Pfizer introduced its 20 valent pneumococcal conjugate vaccine (PCV20), extending vaccine protection against 20 serotypes of invasive and non-invasive pneumococcal disease to adults in India with its single dose formulation.
Based on pathogen type, the Bacterial segment is projected to account for 68% of the market share in 2026. Bacterial conjugate vaccines such as pneumococcal, meningococcal, and typhoid fever diseases comprise the core of routine immunization across the globe. The established efficacy of bacterial vaccines, along with their regulatory approval and government immunization programs, ensure the dominance of the bacterial segment.
For instance, in October 2025, the Government of Bangladesh, with support from UNICEF, Gavi, and WHO, launched a nationwide Typhoid Conjugate Vaccine (TCV) campaign aimed at immunizing around 50 million children from 9 months up to 15 years of age against typhoid fever, which is caused by bacteria.
By patient type, the Pediatric Segment is expected to lead, recording a share of 69% by 2026. Conjugate vaccines are used in the early stages of life for protection against life-threatening bacterial and viral diseases. National vaccination schedules for children are the major reason for the dominance of the pediatric segment.
For instance, in November 2024, Abbott introduced its PneumoShield-14 (14 valent pneumococcal conjugate vaccine) vaccine in the Indian market, targeted at children starting from the age of 6 weeks, by providing broader protection against pneumococcal bacterial diseases than previous PCV vaccines.
To learn more about this report, Request Free Sample
North America is anticipated to lead the conjugate vaccine industry in 2026, holding a significant share of 45%. The growth of the conjugate vaccine market in North America is attributed to factors like high healthcare spending, immunization activities, awareness of vaccine-preventable diseases, and presence of key pharmaceutical companies. Immunization campaigns targeting children and adults are propelling conjugate vaccines.
For instance, in May 2025, Pfizer expanded distribution of its 20‑valent pneumococcal conjugate vaccine (PCV20) in the U.S. and Canada, targeting infants, children, and older adults. The vaccine offers broad protection against 20 pneumococcal strains and has been integrated into national immunization schedules, supporting strong pediatric and adult vaccine coverage in North America.
The Asia Pacific region is expected to be the fastest-growing market in conjugate vaccines due to rising awareness of healthcare, increasing immunization programs by government authorities, expanding pediatric vaccination coverage, and improving multivalent conjugate vaccine access. Countries like India, China, and Japan are investing in large-scale vaccine rollouts to reduce childhood mortality and infectious diseases.
For instance, in August 2025, Pfizer launched PCV20 in India to extend the scope of pneumococcal vaccine protection for infants, children, and adults. This is a move in the broader direction of strengthening vaccine infrastructure through public-private partnerships.
The U.S. conjugate vaccine market is expanding rapidly, based on strong national immunization programs, high healthcare spending, and wide awareness of vaccine-preventable diseases. Conjugate vaccines such as pneumococcal, meningococcal, and typhoid are indicated for infants, children, teens, and high-risk adults.
For instance, in June 2025, the U.S. Food and Drug Administration (FDA) approved Merck’s CAPVAXIVE™ 21‑valent pneumococcal conjugate vaccine for adults, broadening protection against invasive pneumococcal disease and reinforcing adult vaccination strategies in the U.S. market.
China's healthcare reform initiatives and growing emphasis on preventive medicine are driving increased adoption of conjugate vaccines within the country. National policies encouraging wider immunization coverage, including for pneumococcal and typhoid vaccines, extend access to both children and adults.
For instance, in September 2025, CanSino Biologics launched its domestically developed iPneucia® (13‑valent pneumococcal conjugate vaccine) in the China market and shipped it for use, marking a major milestone in China’s self‑sufficient vaccine R&D.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 25,966 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 9.6% | 2033 Value Projection: | USD 49,220 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Sanofi S.A, Pfizer, Inc., Merck & Co., Inc., GlaxoSmithKline plc., Bharat Biotech, Serum institute of India Pvt. Ltd., Biological E. Limited, Bio-Med, Bavarian Nordic, CSL Limited, Novartis AG, Vaxcyte, GSBPL, Taj Pharmaceuticals Limited, and Bavarian Nordic. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The rise in pneumococcal, meningococcal, and typhoid infections has fueled the need for conjugate vaccines. In addition, a higher awareness level among parents and medical professionals of the need to vaccinate children and vulnerable adults against these potentially life-threatening diseases has propelled the market. Immunization programs and campaigns run by national governments and schools have provided a significant boost to this market segment.
The market also has big growth opportunities in the development and launch of multivalent vaccines that cover more bacterial strains in a single dose. Next-generation vaccines, including 20 valent and 21 valent pneumococcal conjugates, which offer much broader protection with simplification of immunization schedules. Besides this, the expansion of pediatric and adult vaccination programs in emerging economies and strategic collaborations between global and local manufacturers can accelerate market expansion more rapidly. Innovations in delivery systems, formulations, and combination vaccines also remain untapped potential.
The market for conjugate vaccines is undergoing a revolutionary overhaul, with recent breakthroughs in vaccine development and emerging health priorities playing a central role. The recent FDA approval of advanced vaccines for pneumococcal diseases in both pediatric and adult populations bears testimony to a definitive shift in the paradigm towards broad-spectrum protection against multiple bacterial pathogens.
The trend of pediatric vaccination remains dominant, marked by the expansion of national programs for vaccination and improved access to multivalent conjugate vaccines. Companies based domestically, such as those based in China and India, are also advancing at a rapid pace and are launching vaccines that suit regional healthcare requirements while also supporting global pharmaceutical firms. This is indicative of an expanding focus on self-reliance for vaccine development.
Another prominent area for growth in vaccines is adult vaccination, with the related theme gaining much attention and importance since governments are making every attempt to provide preventive healthcare for adults on a priority basis, resulting in a reduced disease burden for adults.
Share
Share
Nikhilesh Ravindra Patel is a Senior Consultant with over 8 years of consulting experience. He excels in market estimations, market insights, and identifying trends and opportunities. His deep understanding of the market dynamics and ability to pinpoint growth areas make him an invaluable asset in guiding clients toward informed business decisions. He plays a instrumental role in providing market intelligence, business intelligence, and competitive intelligence services through the reports.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients