The mometasone furoate market is projected to grow from about USD 1,582.6 Mn in 2026 to around USD 2,608.0 Mn by 2033, expanding at a CAGR of 7.4%, due to the need for respiratory treatments, dermatology, new ideas, and the growth of healthcare around the world.
Mometasone, also referred to as mometasone furoate, is a corticosteroid used to treat asthma, hay fever, and several skin problems. Instead of managing asthma attacks, mometasone furoate is used primarily to prevent them. By inhibiting infection-fighting white blood cells (WBCs) from reaching the affected area, it reduces inflammation, itching, and redness. Moreover, it blocks histamine, the chemical messenger that causes the body's allergic reactions. Mometasone furoate is effective in treating symptoms of various skin allergies, as well as other allergic conditions like hay fever and allergic rhinitis.
|
Current Event |
Description and its Impact |
|
Regulatory and Patent Landscape Changes |
|
|
Digital Health and Telemedicine Integration |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
|
Attribute |
Classification / Value |
One‑line Explanation |
|
Potency ranking |
Medium‑to‑high potency |
~2–3× stronger receptor binding than hydrocortisone, enabling effective inflammation control. |
|
Bioavailability |
<1% (nasal spray) |
Minimal systemic absorption, reducing risk of steroid side effects. |
|
Half‑life |
~5.8 hours |
Supports convenient once‑daily dosing in asthma and rhinitis. |
|
Onset of action |
12–24 hours |
Provides rapid symptom relief with full effect in a few days. |
|
Topical use |
Creams, ointments, lotions |
Treats psoriasis, eczema, and dermatitis with balanced potency and safety. |
|
Implant delivery |
3–6 months sustained release |
Long‑acting sinus implants deliver continuous local therapy post‑surgery. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
In terms of dosage form, the nasal spray segment is expected to hold 37% share of the market in 2026, due to effectiveness in treating allergic rhinitis and other sinus problems. They work in one area, quickly relieve symptoms, and have few side effects on the whole body. Nasal sprays will be the most common way to take medicine in 2026 as doctors prefer to prescribe them and patients find them easy to use.
For instance, in November 2025, China's NMPA approved Glenmark's Ryaltris, a nasal spray that combines olopatadine and mometasone furoate, for allergic rhinitis. Grand Pharma sells it, which makes the nasal spray market stronger around the world. Ryaltris is already on the market in a number of countries. It shows how well mometasone furoate nasal sprays work for treating seasonal and perennial allergies.
In terms of indication, the allergic rhinitis segment is projected to account for 42% share of the market in 2026, propelled by increasing instances of seasonal allergies, pollution, and urban lifestyle influences. Many doctors prescribe mometasone furoate nasal sprays as the first treatment. Allergic rhinitis is the biggest cause of market demand in 2026 because it works, is safe, and can lower inflammation.
In terms of route of administration, the nasal segment is expected to lead the market with 47% share in 2026, because it goes straight to the affected area, providing quick relief and less exposure to the whole body. Patients prefer this way because it is easy and non-invasive nature. Doctors suggest it for allergic rhinitis and sinusitis, which supports the idea that nasal administration is the best and most common way to take it in 2026.
For instance, in June 2025, Sanofi's EVEREST Phase 4 trial compared Dupixent and Xolair in people with chronic rhinosinusitis and nasal polyps, both of which were given with mometasone furoate nasal spray. The results showed that Dupixent worked better than mometasone furoate spray in reducing polyp size, congestion, and smell and asthma control. This makes mometasone furoate spray the standard background therapy for nasal administration treatments.
in terms of distribution channel, the retail pharmacies segment is projected to capture the 53% share in 2026, since mometasone furoate products are accessible, inexpensive, and available right away. Patients prefer retail stores because they are convenient, and sales go up in areas where OTC is available. Strong pharmacy networks, especially in North America and Asia-Pacific, make sure that retail pharmacies stay the most important way to get drugs in 2026.
To learn more about this report, Request Free Sample
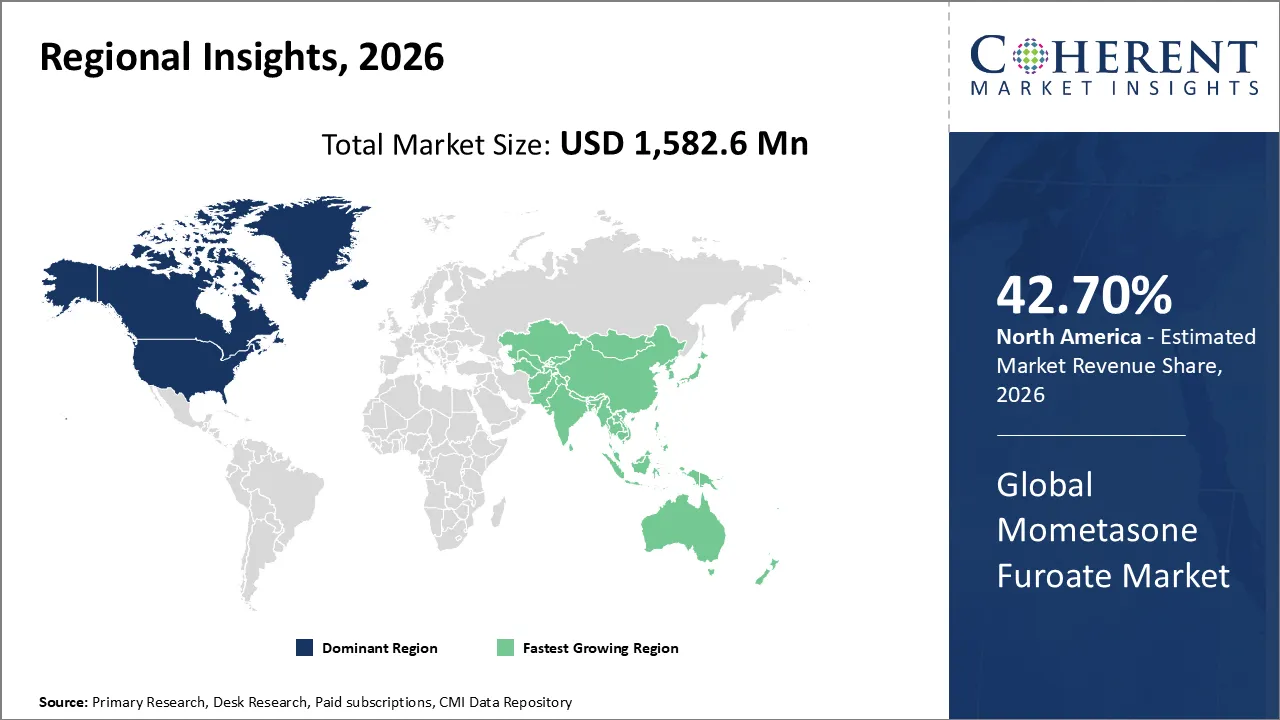
North America is expected to dominate the mometasone furoate market with 42.70% share in 2026, driven by a high number of people with asthma, allergic rhinitis, and skin problems, as well as advanced healthcare infrastructure. The region is the biggest market in the world because it has strong regulatory approvals, big investments in research and development, and quick adoption of new formulations.
For instance, in May 2025, The FDA provided MSD Animal Health's Mometamax Single its approval to treat otitis externa in dogs in the US. This veterinary ear drop has mometasone furoate, gentamicin, and clotrimazole in it. Even though it's not nasal, it shows that mometasone furoate can be used to treat more than just allergies in people. It can also be used to treat animals.
Asia Pacific is expected to exhibit the fastest growth, because more people are getting asthma, allergic rhinitis, eczema, and psoriasis. More accessible healthcare is being made possible by building more healthcare facilities, running more awareness campaigns, and making treatment more affordable. The region is the fastest-growing market in the world due to quick product approvals and health problems caused by urbanization.
In 2026, the U.S. Mometasone Furoate Market will be in high demand because of the high number of people with asthma, allergic rhinitis, and long-term skin problems. Advanced healthcare infrastructure, strong regulatory approvals, and ongoing research and development investments all help new formulations spread quickly. The market's dominant position is strengthened by both patient awareness and physician preference.
For instance, in June 2025, Lyra Therapeutics announced favorable U.S. Phase 3 ENLIGHTEN-2 outcomes for LYR-210, a bioabsorbable nasal implant that administers mometasone furoate for chronic rhinosinusitis. The trial showed big improvements in both symptoms and quality of life, with relief starting as early as week four. The FDA is going to approve LYR-210, which position it against other sinus implants like SINUVA and PROPEL.
In 2026, China's Mometasone Furoate Market demand will be driven by the rising number of asthmas, allergic rhinitis, eczema, and psoriasis cases. This is mostly due to pollution and urbanization. Accessibility is getting better because healthcare infrastructure is growing, the government is working to make treatments more affordable, and more products are getting approved. China's position as a fast-growing market is even stronger because more people are becoming aware of it.
For instance, in January 2026, CSPC Pharmaceutical Group obtained approval from the Chinese NMPA to start trials of an asthma treatment that combines indacaterol acetate and mometasone furoate and is taken once a day by inhalation. This new treatment targets patients whose current treatments fail to function well enough. It improves adherence and strengthens CSPC's respiratory pipeline, which is a good sign for asthma care in China.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1,582.6 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7.4% | 2033 Value Projection: | USD 2,608.0 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Cipla, Teva Pharmaceuticals, Glenmark Pharmaceuticals, AdvaCare Pharma, Medlab Pharmaceuticals Private Limited, Dermocare Laboratories, Iva Healthcare Private Limited, Unilab Chemicals & Pharmaceuticals Pvt Ltd, Organon group of companies, Cosette Pharmaceuticals, Taro Pharmaceutical Industries Ltd., and Hikma Pharmaceuticals |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
A greater amount is being invested in research and development, which is leading to new developments in corticosteroids, especially mometasone furoate. To improve patient outcomes, drug companies are working on better formulations, delivery methods, and therapeutic uses. This increase in R&D not only makes the drug more effective in the clinic, but it also expands potential uses for it, which drives the mometasone furoate market growth. As more advanced solutions become available, the industry is expected to see faster adoption and a stronger competitive position in the global healthcare market.
Regulatory approvals are crucial in shaping the pharmaceutical industry. The fact that an increasing number major companies are getting their products approved for mometasone furoate means that treatments will be available and accessible. These approvals show that the treatments are safe and effective, which makes doctors and patients more confident in them. As a result, the growing number of approved products is directly driving up the mometasone furoate market demand, as healthcare providers are becoming ever more inclined to prescribe these products for skin and respiratory problems, making it easier to get into the market and offer edge over competitors.
Healthcare organizations, NGOs, and drug companies are running awareness campaigns that are having a big impact on patient education and treatment adoption. These programs make it highlights the benefits of mometasone furoate, usage guidelines, and safety aspects, which helps people understand it better and follow the rules. Campaigns that provide patients with information can significantly aid in early diagnosis and treatment compliance. This proactive strategy enhances the visibility of therapeutic options, subsequently expanding market research opportunities for mometasone furoate. Stakeholders are keenly interested in patient behavior, preferences, and the evolving landscape of treatment trends.
The mometasone furoate market forecast indicates robust growth in respiratory applications, particularly for asthma and allergic rhinitis. Urban pollution, shifts in lifestyle, and the increasing prevalence of respiratory conditions are all contributing to the rising popularity of nasal sprays and inhalation powders. Mometasone furoate formulations are favored because they target specific issues and offer prolonged relief, thereby encouraging patient adherence to the prescribed treatment.
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients