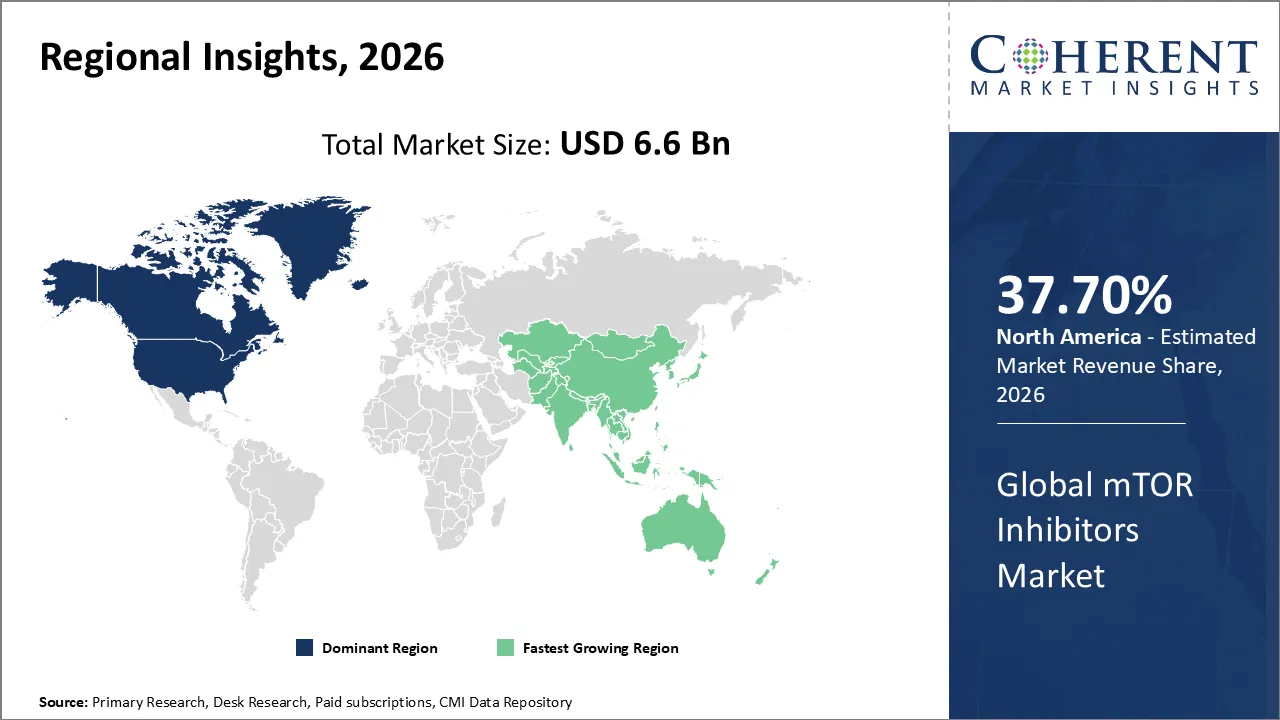
The mTOR Inhibitors Market is estimated to be valued at USD 6.6 Bn in 2026 and is expected to reach USD 8.3 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 5.8% from 2026 to 2033.
Researchers and healthcare providers are driving steady growth in the mTOR inhibitors market as cancer prevalence rises and the use of targeted therapies increases. Physicians widely prescribe mTOR inhibitors such as Everolimus, Temsirolimus, and Sirolimus in oncology and organ transplantation to regulate cell growth and control immune responses. Scientists continue to expand research in molecular biology and explore new clinical applications, while the healthcare sector increasingly adopts precision medicine. At the same time, pharmaceutical companies actively develop new drugs and healthcare systems improve infrastructure, which further accelerates the global adoption of mTOR inhibitor therapies.
|
Current Events |
Description and its impact |
|
Advances in Precision Medicine and Biomarker Development |
|
|
Regulatory and Reimbursement Policy Changes in Key Regions |
|
|
Emerging Competition from Next-Generation Therapeutics and Combination Regimens |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Oral acquired the prominent market share of 52.2% in 2026. Patients and clinicians are driving growth in the oral segment of the mTOR inhibitors market by favoring convenient, self-administered therapies. Medications such as Everolimus and Sirolimus enable patients to continue treatment at home, minimizing hospital visits and simplifying long-term therapy management. Healthcare providers actively recommend oral formulations for chronic conditions and post-transplant care, while pharmaceutical companies develop highly bioavailable, patient-friendly options. Together, these factors—convenience, improved adherence, and continuous innovation—are accelerating the adoption of oral mTOR inhibitor therapies. For instance, in March 2025, the phase 1/2 trial of oral zelenirstat (PCLX-001), a first-in-class N-myristoyltransferase inhibitor, has dosed its first patient with relapsed/refractory acute myeloid leukemia (AML) at The University of Texas MD Anderson Cancer Center, building on prior phase 1 trials in solid tumors and non-Hodgkin lymphoma and preclinical anti-AML data.
Everomilus hold the largest market share of 42.2% in 2026. Clinicians are increasing the use of Everolimus, which is driving its demand in the mTOR inhibitors market. Oncologists commonly prescribe the drug to treat conditions such as renal cell carcinoma and hormone-receptor-positive breast cancer, strengthening its role in targeted therapy. Hospitals also administer it after organ transplantation to regulate immune responses and help prevent rejection. Researchers continue to conduct clinical trials while healthcare providers expand patient access to targeted treatments, and this growing awareness of pathway-based cancer therapy is accelerating the adoption of Everolimus. For instance, in October 2025, Natco Pharma has launched 1 mg Everolimus tablets, a generic version of Novartis’ Zortress, in the immunosuppressant therapeutic class.
Oncology expected to hold largest market share of 36.6% in 2026. Healthcare providers are actively driving the growth of the oncology segment in the mTOR inhibitors market by increasingly adopting pathway-targeted treatments for complex cancers. Doctors commonly prescribe mTOR inhibitors like Everolimus and Temsirolimus to treat conditions such as renal cell carcinoma and neuroendocrine tumors. Clinicians rely on biomarker-based diagnostics to identify patients who will benefit most from these therapies. Meanwhile, pharmaceutical companies develop combination regimens and new oncology indications, and hospitals expand specialized cancer programs, collectively reinforcing the importance of mTOR inhibitors in contemporary oncology care. For instance, in January 2025, Kazia Therapeutics Limited, an oncology-focused drug development company, received regulatory approval and initiated a clinical trial to evaluate the combination of paxalisib and immunotherapy in patients with advanced breast cancer.
Hospital Pharmacies captures the largest market share of 38.8% in 2026. Hospital pharmacies actively drive growth in the mTOR inhibitors market by functioning as the main distribution channel for specialized therapies. Clinicians depend on these pharmacies to supply drugs such as Everolimus and Temsirolimus for oncology and post-transplant treatments, ensuring accurate dosing and careful monitoring. Hospitals provide the necessary infrastructure for safe storage, preparation, and administration of these medications. At the same time, the rising number of cancer treatments and organ transplants, supported by skilled pharmacy staff, enhances the availability and use of mTOR inhibitors through hospital pharmacies.
To learn more about this report, Request Free Sample
North America dominates the overall market with an estimated share of 37.70% in 2026. Healthcare providers in North America are actively driving the mTOR inhibitors market by increasingly adopting targeted therapies for cancer and transplant care. Pharmaceutical companies are developing new formulations, including oral and combination therapies, and conducting research to meet growing clinical demand. Hospitals and specialized treatment centers are expanding patient access, leveraging advanced healthcare infrastructure and skilled professionals. Meanwhile, rising awareness of precision medicine and proactive regulatory support for innovative oncology and immunosuppressive treatments are shaping market trends and accelerating the adoption of mTOR inhibitors across the region. For instance, in November 2025, Hyderabad-based NATCO Pharma has launched the immunosuppressant generic Everolimus, an mTOR inhibitor used to prevent organ rejection in adult kidney and liver transplant patients, in the United States.
Healthcare providers across Europe are actively expanding the mTOR inhibitors market by integrating targeted therapies into oncology and transplant treatment protocols. Pharmaceutical companies are developing and launching new drugs and formulations, including oral and combination therapies, to meet diverse patient needs. Hospitals and specialty clinics are increasing patient access by leveraging advanced infrastructure and skilled medical staff. At the same time, rising awareness of precision medicine, along with supportive regulations and ongoing clinical research, is accelerating the adoption of mTOR inhibitors and establishing Europe as a leading region for innovative targeted therapies. For instance, Novartis announced that the EMA’s CHMP issued a positive opinion for Votubia (everolimus) dispersible tablets as an adjunctive treatment for patients aged two and older with TSC-related refractory partial-onset seizures.
Clinicians in the United States are actively driving the mTOR inhibitors market by increasingly using targeted therapies for cancer and post-transplant care. Biopharmaceutical companies are developing and launching novel oral and combination formulations while conducting clinical trials to expand indications and enhance treatment outcomes. Leading hospitals and specialty centers are improving patient access through advanced infrastructure and skilled healthcare professionals. At the same time, growing focus on precision medicine, along with supportive FDA regulations and heightened awareness of pathway-specific therapies, is accelerating the adoption of mTOR inhibitors, positioning the U.S. as a key hub for innovative targeted treatments. For instance, in March 2025, Faeth Therapeutics and The GOG Foundation have dosed the first patient in their Phase 2 trial combining PIKTOR—FTH-001 (serabelisib) and FTH-003 (sapanisertib)—with paclitaxel, exploring dual PI3Kɑ-mTORC1/2 inhibition in endometrial cancer.
Healthcare providers across the United Kingdom are actively driving growth in the mTOR inhibitors market by integrating targeted therapies into cancer treatment and post-transplant care. Pharmaceutical companies are developing and launching new oral and combination formulations while conducting clinical studies to expand their therapeutic use. Hospitals and specialized clinics are enhancing patient access through advanced infrastructure and skilled healthcare staff. At the same time, rising awareness of precision medicine, combined with supportive guidance from regulators like the MHRA, is accelerating the adoption of mTOR inhibitors, establishing the U.K. as an emerging hub for innovative targeted therapies.
Healthcare providers increasingly prefer targeted therapies like mTOR inhibitors over traditional chemotherapy for cancer management. Drugs such as Everolimus and Temsirolimus are gaining traction in treating renal cell carcinoma, breast cancer, and neuroendocrine tumors. Advances in molecular research and biomarker-based patient selection are enabling clinicians to deliver more personalized treatment plans, improving efficacy and safety. This trend highlights precision medicine as a key driver for adoption of mTOR inhibitors in modern oncology care.
Oral mTOR inhibitors, including Everolimus and Sirolimus, are becoming more popular due to patient convenience and home-based administration. Patients and healthcare providers favor oral therapies to reduce hospital visits, improve adherence, and facilitate long-term management of oncology and post-transplant conditions. Pharmaceutical companies are responding with highly bioavailable, patient-friendly formulations. The trend reflects a broader move toward outpatient care and self-administered therapies, enhancing patient quality of life while maintaining therapeutic effectiveness.
Oral mTOR inhibitors present a growing opportunity as patients and providers increasingly prefer convenient, self-administered therapies. Companies can develop highly bioavailable, patient-friendly formulations that reduce hospital visits and improve adherence. Expanding the availability of oral therapies in oncology and post-transplant care can enhance patient quality of life and support broader adoption of mTOR inhibitors across chronic and long-term treatment settings.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 6.6 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 5.8% | 2033 Value Projection: | USD 8.3 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Novartis AG, Hikma Pharmaceuticals PLC., Par Pharmaceutical, Teva Pharmaceutical Industries Ltd., Dr. Reddy’s Laboratories Ltd., Zydus Cadilla, Pfizer, Inc., Biocon, Alkem Laboratories Ltd., Accord Healthcare Inc., Gland Pharma Ltd., Apotex Inc., and Glenmark Pharmaceuticals Ltd. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients