The next generation sequencing market is estimated to be valued at USD 21.76 Bn in 2026 and is expected to reach USD 57.54 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 14.9% from 2026 to 2033.
To learn more about this report, Request Free Sample
The increased research and development for precision medicine, reduced sequencing costs, and advances in data integration and analysis are driving the growth of this market. The next generation sequencing market is witnessing significant technological upgrades with new product launches focusing on automation, compact size, and improved data analytics capabilities. Leading players are investing in key areas like direct-to-consumer genetic testing, non-invasive prenatal testing, oncology, and agricultural and animal research to boost market revenues. Wider clinical applications and therapeutic areas are expected to expand the potential of next generation sequencing in the coming years.
|
Current Event |
Description and its Impact |
|
US-China Geopolitical Trade Tensions |
|
|
Precision Medicine Initiative Expansion and Funding |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
NGS plays a pivotal role in cancer care by identifying genetic mutations that drive tumor growth. It enables:
NGS is used for non-invasive prenatal testing (NIPT) to screen for chromosomal abnormalities like Down syndrome. In newborns, it helps diagnose rare genetic disorders early, allowing timely interventions.
Whole-exome and whole-genome sequencing help uncover the genetic basis of rare diseases that are difficult to diagnose through conventional methods. This is especially valuable in pediatric and neurological cases.
NGS enables rapid identification of pathogens, tracking of outbreaks, and analysis of antimicrobial resistance. It was instrumental during the COVID-19 pandemic for variant surveillance.
By analyzing how genes affect drug metabolism, NGS supports personalized prescribing, reducing adverse drug reactions and improving efficacy.
NGS helps in HLA typing and immune repertoire profiling, improving donor-recipient matching and understanding immune responses.
Large-scale sequencing projects are identifying genetic risk factors for common diseases, paving the way for preventive screening and lifestyle interventions tailored to individual genetic profiles.
In terms of technology, the reagents & consumables segment is estimated to hold 59% the highest share of the market in 2026, owing to the growing demand for consumables and reagents among end-users. Extensive R&D activities in the fields of genomics and precision medicine have fueled the adoption of next-generation sequencing technologies. Since these high-throughput sequencing platforms require regular procurement of reagents and other associated consumables for their operation, this segment experiences consistent sales. Moreover, the decreasing costs of reagents and kits have made sequencing techniques more affordable for clinical research. Continuous technological advancements are also improving the efficiency and throughput of sequencing assays, leading to higher consumption of sequencing reagents, primers and enzymes among laboratories. The non-invasiveness and high sensitivity of NGS techniques further support the increased uptake of sequencing tests, thereby propelling the reagents and consumables segment.
For instance, in July 2025, QIAGEN expanded its next-generation sequencing (NGS) portfolio with the launch of QIAseq xHYB Long-Read Panels. Designed to deliver high-resolution genomic insights, the panels enable hybrid capture of long DNA fragments for comprehensive variant detection. This innovation supports precision medicine and enhances research capabilities across oncology, rare diseases, and hereditary conditions.
To learn more about this report, Request Free Sample
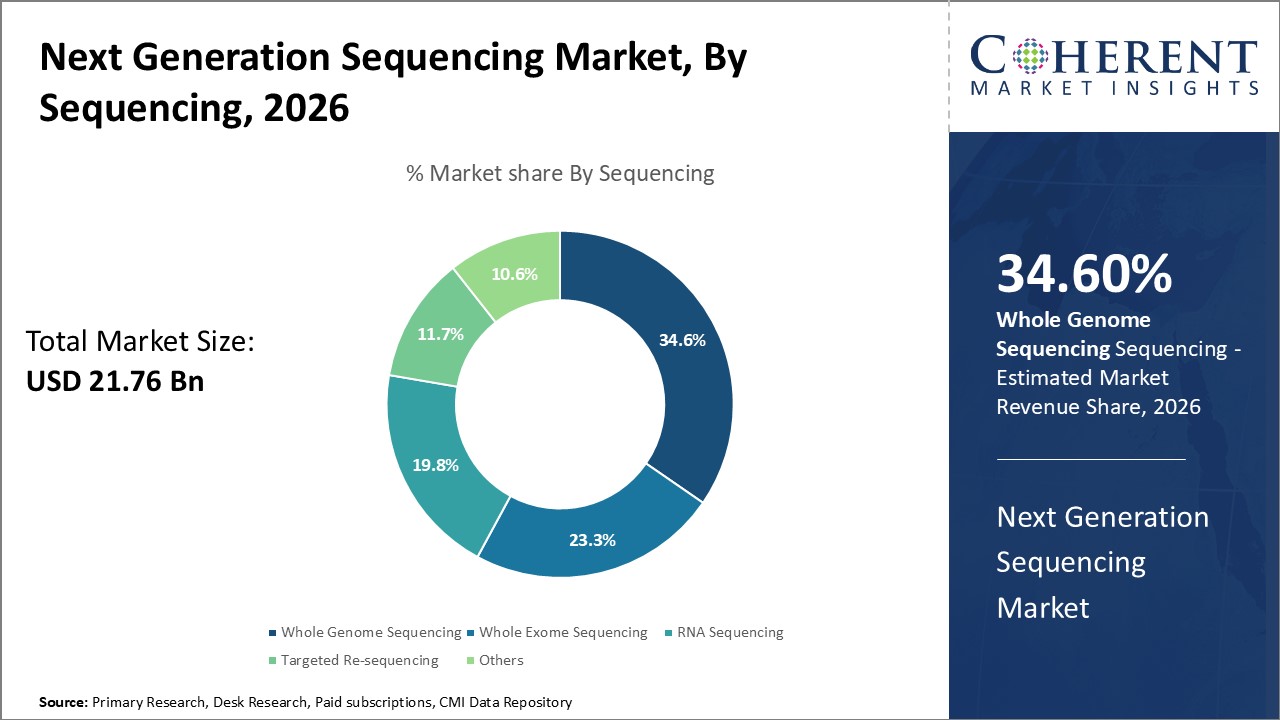
In terms of sequencing type, the whole genome sequencing segment hold the highest share of the next generation sequencing market and is estimated to hold 34.6% of the market share in 2026. Reduced costs and enhanced speed of whole genome sequencing have enabled its use in diverse areas such as oncology, reproductive health and agricultural biotechnology. For instance, whole genome profiling is increasingly utilized for comprehensive tumor characterization and mutation detection in cancer. It also aids in newborn screening, infectious disease investigations and preimplantation genetic diagnosis. Moreover, whole genome analysis finds application in identifying genetic variations associated with drug response and trait mapping in plants and animals. Initiatives by governments and private agencies to sequence thousands of human genomes have further fostered the adoption of whole genome sequencing procedures. The proven effectiveness of WGS in precision diagnostics and personalized care makes it a preferred sequencing type.
For instance, in May 2025, MGI Tech launched the DNBSEQ-G400RS High-throughput Sequencing Set in Europe, designed to enhance clinical sequencing efficiency and data quality. The new kit supports a wide range of applications including whole genome and targeted sequencing, offering improved flexibility and scalability for laboratories. This release marks a significant step in advancing precision medicine across the region.
In terms of application, the personalized medicine segment holds the highest share of the next generation sequencing market and is estimated to hold 40% of the market share in 2026. Advancements in genomic sciences and bioinformatics have enabled sequencing techniques to study individual patient's DNA, RNA and genomes. This revolutionized drug development processes wherein treatments can be customized based on a person's genetic profile. For instance, comprehensive genomic profiling via NGS aids in identifying therapeutic targets and biomarker-driven clinical trials. It also supports development of companion diagnostics to select appropriate patient populations for particular therapies. Thus, sequencing is widely utilized to develop effective and safer personalized treatment regimens. Furthermore, sequencing assists in disease screening, monitoring treatment response and detecting resistance at a molecular level. These merits have boosted the integration of NGS-based genomic data in clinical management across varied therapeutic areas like oncology and cardiology. The shift towards precision medicines will subsequently spur demand from the personalized medicine application segment over the coming years.
For instance, in April 2025, Illumina and Tempus announced a strategic collaboration to accelerate the clinical adoption of next-generation sequencing through genomic AI innovation. By integrating Illumina’s AI technologies with Tempus’s multimodal data platform, the partnership aims to enhance molecular profiling across major disease categories, advancing precision medicine and improving patient outcomes globally.
In terms of end user, the hospitals and clinics segment is expected to hold the largest share of the market in 2026, due to their growing reliance on NGS for precision diagnostics, personalized treatment planning, and rapid turnaround times. With in-house sequencing capabilities, integration into electronic health records, and expanding regulatory support, NGS is transforming clinical workflows and redefining genomic medicine in hospital settings.
For instance, in June 2024, Command Hospital Pune inaugurated a cutting-edge genome sequencing lab featuring next generation sequencing (NGS) facilities. This new setup aims to advance precision diagnostics and genomic research within the Armed Forces Medical College (AFMC), reinforcing the hospital’s role in delivering high-quality, technology-driven healthcare solutions.
To learn more about this report, Request Free Sample
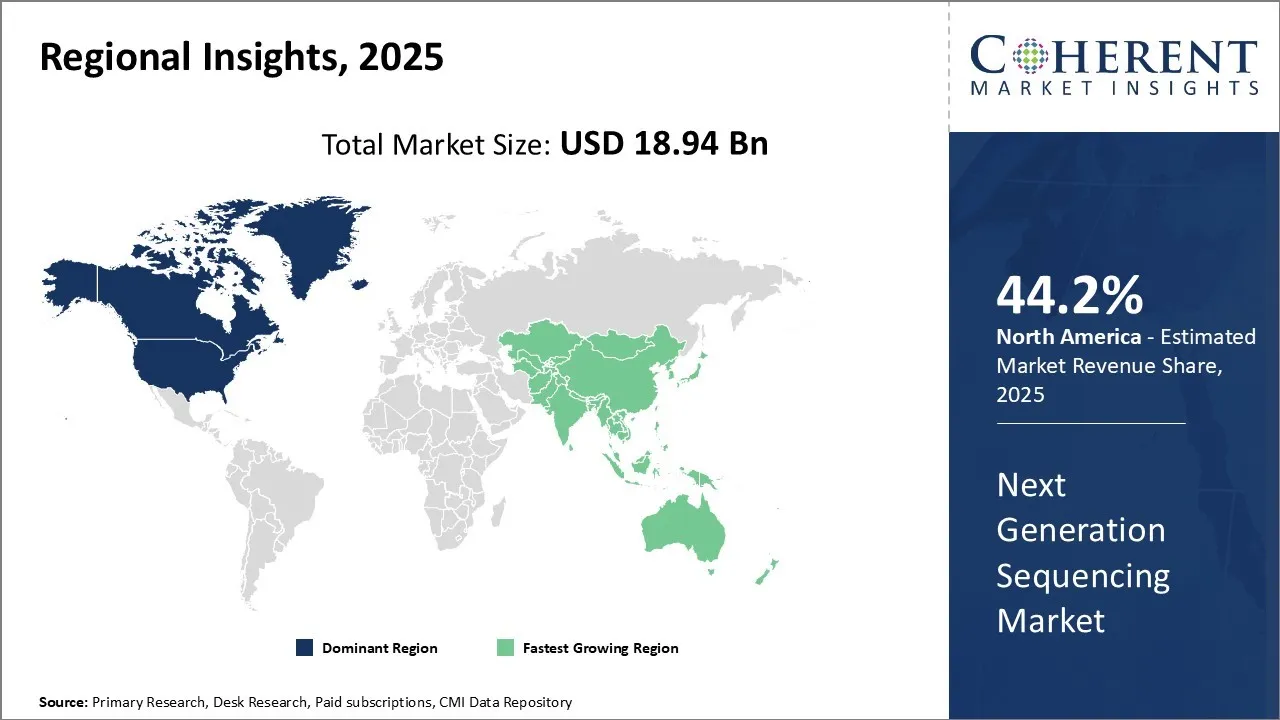
North America has established itself as the dominant region in the next generation sequencing market and is estimated to hold 44.2% of the market share in 2026. With strong presence of global industry leaders and emergence of several startups, the region enjoys high adoption rates of newer sequencing technologies. Presence of sophisticated research infrastructure, availability of funding for genomics and precision medicine initiatives have paved the way for North America's leadership.
Several pharmaceutical and biotechnology companies in the region are investing significantly in applying NGS for drug discovery and development. Leading academic institutes are also supporting research in clinical genomics through sequencing-based techniques. With the objective of faster drug approvals and lower costs, the U.S. FDA has also been encouraging use of biomarkers and companion diagnostics, driving use of NGS in clinical settings. The region also has robust bioinformatics capabilities helping accelerate genomic research. High disposable incomes allow for superior infrastructure and talent availability in the region.
For instance, in June 2026, bioMérieux announced the acquisition of Day Zero Diagnostics, a U.S.-based company specializing in next-generation sequencing (NGS) and AI-powered infectious disease diagnostics. The move strengthens bioMérieux’s capabilities in rapid pathogen identification and antimicrobial resistance profiling, supporting earlier and more targeted treatment decisions. This strategic expansion reinforces its commitment to advancing precision microbiology.
Asia Pacific is expected to exhibit the fastest growth of the market, due to a combination of healthcare needs, technological advancements, and supportive government initiatives. Rising cases of cancer, rare diseases, and infectious conditions have increased the need for advanced molecular diagnostics. National genome projects in countries like India, Japan, and Thailand are accelerating NGS adoption, while falling sequencing costs and improved platform efficiency have made the technology more accessible. Public-private collaborations and expanding research infrastructure are further driving growth. Additionally, regulatory support and the integration of NGS into clinical guidelines are promoting its routine use in precision medicine across the region.
For instance, in May 2026, Gene Solutions and NEWCL partnered to launch an advanced Next Generation Sequencing (NGS) laboratory in Taiwan, focusing on prenatal and oncology genetic testing. The facility aims to enhance early disease detection and personalized treatment through high-precision genomic analysis. This collaboration marks a significant step in expanding precision medicine capabilities across Asia.
The United States remains the global leader in the Next Generation Sequencing market, driven by widespread clinical adoption, substantial research funding, and a thriving biotech ecosystem. Its advanced healthcare infrastructure, strong regulatory support, and ongoing innovation in genomics and precision medicine continue to fuel demand for NGS technologies across diagnostics, oncology, and rare disease research.
For instance, in September 2026, Illumina introduced the Illumina Complete Long-Read technology, offering a cost-effective and scalable solution for comprehensive genomic analysis. Designed to integrate seamlessly with existing workflows, the new platform enables accurate detection of structural variants and complex regions. This innovation enhances clinical and research applications, advancing precision medicine and whole-genome sequencing capabilities.
China is rapidly becoming a major force in the Next Generation Sequencing market, driven by national genome initiatives, expanding genomics research, and a booming biotech sector. Government investments and innovations from companies like MGI Tech are making NGS more scalable and accessible. Rising demand for precision medicine and diagnostics further fuels market growth across the region.
For instance, in September 2026, China’s MGI Tech launched the T7+, a next-generation high-throughput sequencer designed to accelerate genomic data mining. Capable of sequencing up to 150,000 samples annually, the T7+ enhances efficiency in large-scale research and clinical applications. This innovation marks a significant leap in precision medicine and global genomics capabilities.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 21.76 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 14.9% | 2033 Value Projection: | USD 57.54 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Thermo Fisher Scientific Inc., Agilent Technologies Inc, bioMérieux SA, F. Hoffmann-La Roche Ltd, QIAGEN, Illumina Inc., Oxford Nanopore Technologies plc., BGI, PerkinElmer Genomics, Bio-Rad Laboratories Inc., Pacific Biosciences, 10x Genomics, Promega Corporation, Macrogen Inc., Eurofins Scientific, DNASTAR Inc., and CD Genomics |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
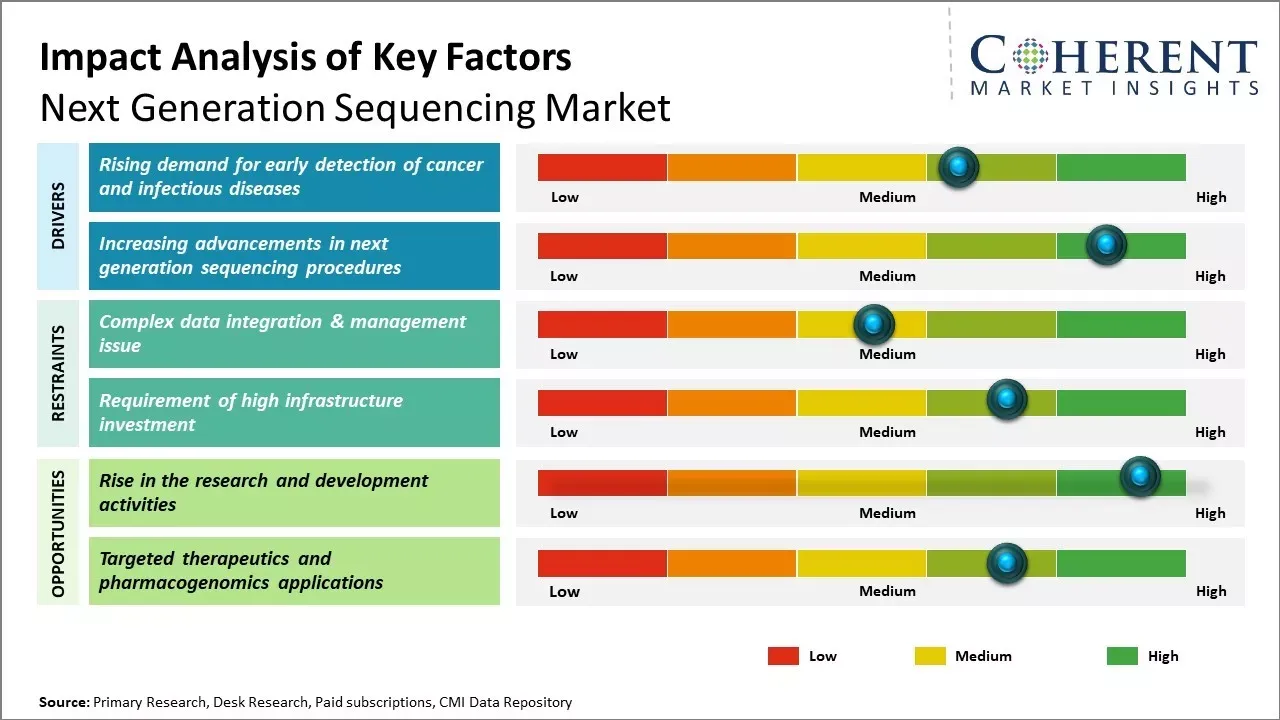
The chances of successful treatment increase when cancer and infectious diseases are detected at the early stage. One of the major factors that drive growth of the next generation sequencing market is the rising prevalence of cancer and other infectious diseases such as tuberculosis, meningococcal disease, etc.
For instance, in January 2025, Oxford Gene Technology (OGT) launched the SureSeq Myeloid MRD Panel, offering enhanced minimal residual disease (MRD) detection for myeloid malignancies. Designed for high sensitivity and specificity, the panel supports personalized monitoring and treatment decisions in hematologic cancers. This advancement strengthens OGT’s NGS portfolio and its commitment to precision oncology solutions.
The declining cost of sequencing per genome is improving the affordability and allowing new clinical and research applications. The developments in single-cell sequencing and spatial transcriptomics provide new opportunities in precision medicine and disease research. Additionally, the sequencing of various genomes is helping in better understanding of thousands of genetic disorders and opening avenues for new treatment development.
The NGS market is undergoing an accelerated phase of technological commoditization in short-read sequencing while simultaneously entering a differentiation era driven by long-read platforms, integrated workflows, and clinical-grade claims. Short-read throughput and reagent cost reductions have removed many traditional barriers to scale, pushing sequencing from an instrument-led premium service to a high-volume commodity for large labs and population projects; several industry reports and vendor announcements indicate roadmap-driven cost targets in the low-hundreds to sub-hundred dollars per human genome, a dynamic that inherently compresses per-sample margins for incumbent suppliers and forces a shift in where value is captured.
Clinical translation is the primary battleground for differentiated pricing and durable revenue. Oncology and prenatal testing exemplify where sequencing has moved from research to standard-of-care workflows: professional oncology guidelines and real-world registry studies now recommend or report tumor NGS for defined indications, and implementation studies show clinically actionable changes and therapy adjustments in a measurable minority of tested patients — evidence that underpins payers’ and hospital systems’ willingness to reimburse for panels and companion diagnostics rather than generic sequencing runs. The result is that companies that pair validated assays, regulatory claims, and robust bioinformatics will sustain margins even as raw sequencing commoditizes.
Long-read technologies and non-sequencing chemistries are the obvious vectors for future premiumization. Permanent advantages will accrue to platforms that deliver clinically validated long reads (structural variants, phasing), low-complexity sample prep, and end-to-end solutions (from library prep to interpretation). This is visible in vendor product announcements and executive strategies emphasising companion diagnostics and pharma partnerships — moves intended to lock customers into higher-value, regulated workflows rather than open-ended research runs. Illumina’s recent pharma partnership activity and similar moves across suppliers illustrate this strategic pivot towards higher-value clinical claims and co-development.
*Definition: The next generation sequencing market involves technologies and products that are used for DNA sequencing. It allows rapid sequencing of genomes and includes techniques such as shotgun sequencing, sequencing by synthesis, ion semiconductor sequencing, single-molecule real-time sequencing, nanopore sequencing, and miniature genome sequencing. Next generation sequencing enables faster and more affordable DNA analysis, helping to advance genomic research and its applications in healthcare, agriculture, and other fields.
Share
Share
Nikhilesh Ravindra Patel is a Senior Consultant with over 8 years of consulting experience. He excels in market estimations, market insights, and identifying trends and opportunities. His deep understanding of the market dynamics and ability to pinpoint growth areas make him an invaluable asset in guiding clients toward informed business decisions. He plays a instrumental role in providing market intelligence, business intelligence, and competitive intelligence services through the reports.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients