The Global Patient Registry Software Market is anticipated to grow at a CAGR of 12.6% with USD 2.05 Bn share in 2026 and is expected to reach USD 4.72 Bn in 2033. This growth in the market is driven by rising burden of chronic diseases and increasing demand for real-world evidence (RWE) in clinical decision-making.
Commercial Registry Software has the biggest market share at 62% in 2026 because hospitals, specialty networks, pharma companies, and CROs usually prefer vendor-backed platforms that can work across multiple sites, work with EHR workflows, and support regulated reporting. That preference is backed up by the fact that healthcare delivery is becoming more digital. By 2025, 96% of non-federal acute care hospitals and 78% of office-based doctors had adopted certified EHRs. By 2023, 70% of hospitals were involved in all four areas of interoperable exchange.
The FDA says that product and disease registries are good sources of real-world data. Its guidance also talks about using new or existing registries to help make decisions about drugs and biologics. That makes it more likely that commercial platforms will have better auditability, validation, security, and control over workflows with multiple stakeholders. The CDC says that chronic diseases are the main cause of the country's $4.9 trillion in annual healthcare costs. 90% of U.S. healthcare spending is for people with chronic and mental health problems.
The American College of Cardiology started the CONNECT-HCM digital registry on August 19, 2025. At the time, it had already signed up more than 70 patients and connected registry data with EHRs to keep track of symptoms, imaging, treatments, and outcomes.
The Cancer Registry segment makes up 28% of the patient registry software market and is the most popular because oncology needs one of the most mature, standardized, and large-scale data infrastructures in healthcare. The base of demand is very large: In 2025, IARC reported 30 million new cancer cases, 9.7 million deaths, and 53.5 million people still alive five years after their diagnosis.
The number of people who survive cancer is rising, which means that long-term follow-up, tracking recurrences, analyzing treatment pathways, and monitoring outcomes are all becoming more important. The National Cancer Institute (NCI) estimates that there are 18.6 million cancer survivors in the U.S., which is 5.4% of the population. Oncology is becoming a top priority for regulatory-grade real-world evidence. FDA's registry guidance clearly talks about how to use new or existing registries to help make regulatory decisions.
The FDA held a public workshop on August 27, 2025, called "The Future of Registries in Oncology." It was about common data elements and best practices for developing oncology registries. On September 5, 2025, the CDC also released GenEDITS Plus 6.1.31 for cancer registries and EditWriter 6.0.77 on February 27, 2026.
To learn more about this report, Request Free Sample
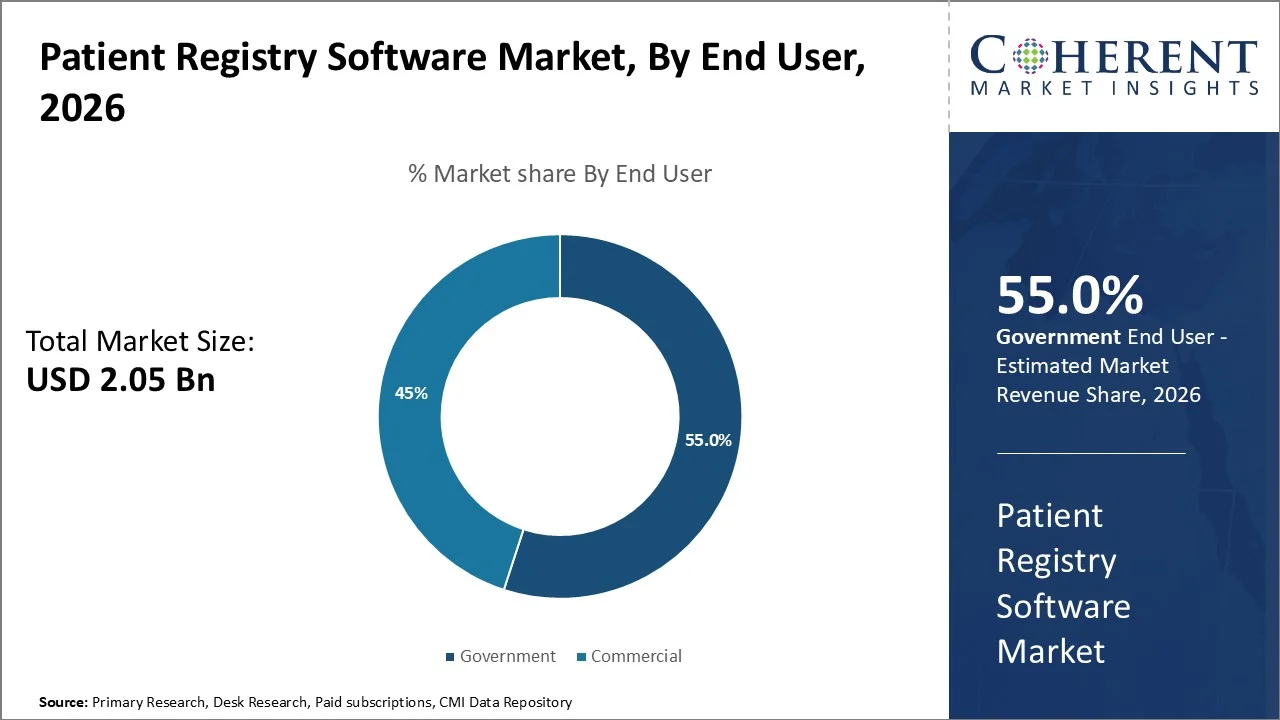
The Government segment makes up 55% of the patient registry software market. This is because public agencies run the biggest registries for surveillance and population-level monitoring. The CDC's National Program of Cancer Registries helps central registries in 46 states, the District of Columbia, Puerto Rico, the U.S. Pacific Island Jurisdictions, and the U.S. Virgin Islands, which together represent 97% of the U.S. population. Along with NCI's SEER program, these systems collect cancer data for the entire U.S. population. CDC's U.S. Cancer Statistics has covered all new cancers and deaths in all 50 states and D.C. from 2003 to 2022.
On September 5, 2025, CDC released GenEDITS Plus 6.1.31, which added the ability to process large registry files at the same time. CDC updated CRS Plus/TLC Plus, its main central-registry database tools, on September 11, 2025. CDC said that more than 40,000 people had signed up for the National Firefighter Registry for Cancer by February 10, 2026.
Registry platforms are getting better at using real-world evidence. According to the FDA the product or disease registries, electronic health records, medical claims, and digital health technologies are all good sources of real-world data (RWD). The FDA's CDER data also show that the number of protocol submissions related to RWE went up from 10 in FY2023 to 11 in FY2024. In FY2024, there were also 5 final study reports that used RWD/RWE. Four of the FY2024 protocol submissions used electronic health records, and seven used medical claims. This means that new ideas in registry software are more and more focused on structured data capture, compliance, analytics, and creating evidence that meets regulatory standards. (FDA)
|
Current Event |
Description and its Impact |
|
FDA Workshop Pushes for High-Quality Oncology Registries |
|
|
NORD Lowers Entry Barrier for Rare Disease Registries |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
To learn more about this report, Request Free Sample
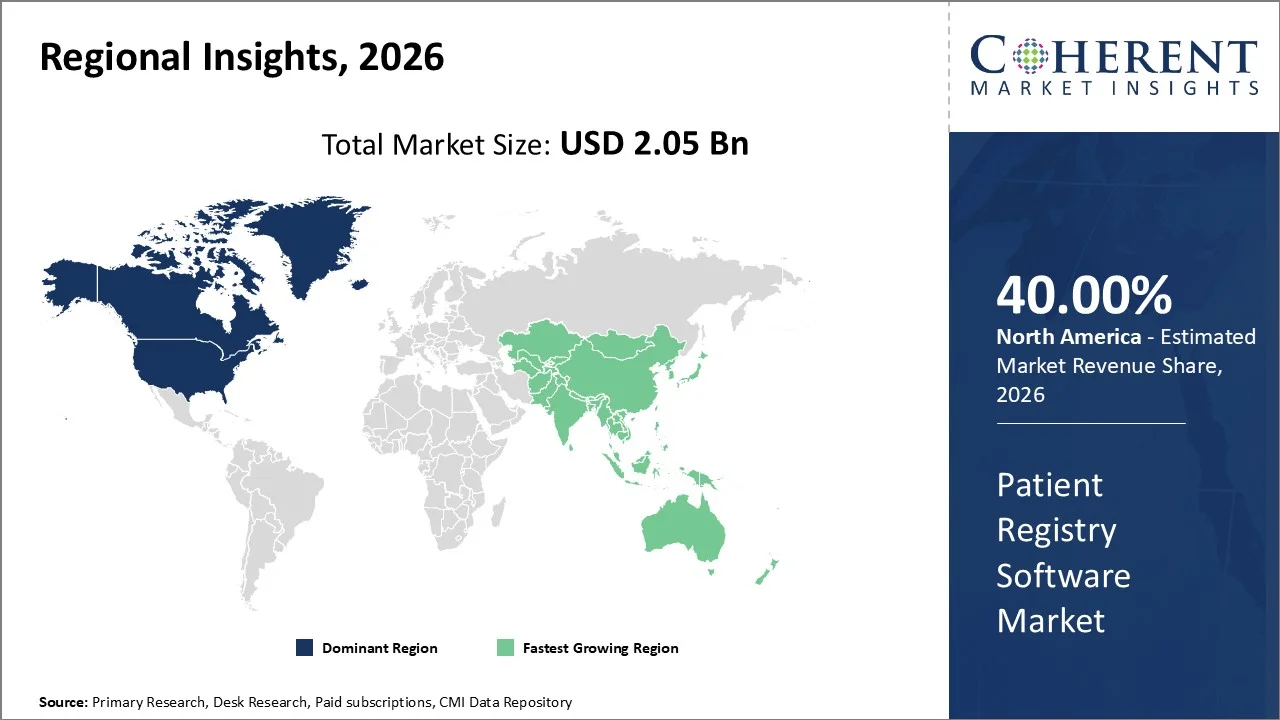
With a projected 40% market share in 2026, North America remains the largest region for patient registry software, supported by sophisticated healthcare IT and a robust regulatory environment. Additionally, the number of large vendors and widespread adoption by providers of electronic health records have helped. According to Centers for Medicare and Medicaid, US healthcare spending is projected to exceed USD 4.5 trillion, with an increasing amount of that investment being allocated to digital health solutions like registry systems.
The Asia Pacific region is forecasted to be the fastest growing region, with a compound annual growth rate of 14.8% between 2026-2033, due largely to accelerating digitization of healthcare and increasing government investments. An additional factor supporting increased investment in registry solutions in Asia Pacific countries like India and China is that both nations are developing and deploying national health databases and registries to help them manage rising levels of chronic disease among their populations. The world bank has documented steady growth in healthcare expenditures throughout the Asia Pacific, driven by economic expansion and policy changes.
The U.S. has one of the most advanced environments for patient registry software because it already has a strong digital base. By 2024, 9 out of 10 hospitals had allowed patients to access their records through an API, and 7 out of 10 will have done so through a standards-based API. This supports the need for registry software that can connect directly to hospital systems, automatically collect data, and make long-term tracking better.
India is becoming a high-potential market for patient registry software because it is building a huge digital health backbone. As of December 12, 2025, the Government of India said that 83.94 crore ABHA accounts had been made, 7.47 lakh healthcare professionals had signed up for ABDM, 4.42 lakh health facilities had signed up, and 79.71 crore health records had been linked to ABHA. This scale lays a solid groundwork for registry software that can help identify patients, link records, and share data between facilities.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 2.05 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 12.6% | 2033 Value Projection: | USD 4.72 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
IQVIA, Global Vision Technologies, Dacima Software, ImageTrend, Oracle, Health Catalyst, Conduent, Elekta, PatientCrossroads, McKesson, Liaison Technologies, Evado eClinical Solutions. |
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Some of the major key players in Patient Registry Software Market are IQVIA, Global Vision Technologies, Dacima Software, ImageTrend, Oracle, Health Catalyst, Conduent, Elekta, PatientCrossroads, McKesson, Liaison Technologies, Evado eClinical Solutions.
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients