The global respiratory devices market is estimated to be valued at USD 29.21 Bn in 2026 and is expected to reach USD 62.19 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 11.4% from 2026 to 2033.
Discover market dynamics shaping the industry: Request Free Sample
Rising geriatric population, increasing prevalence of chronic respiratory diseases such as chronic obstructive pulmonary disease (COPD) and asthma, and rising airborne diseases are some of the key factors expected to drive the demand for respiratory devices. Technological advancements in the field of respiratory care devices, rising healthcare expenditure, and growing access to health insurance in developing nations also provide opportunities for market growth. However, high cost of innovative and new respiratory devices and procedures may restrict the market growth to some extent over the coming years.
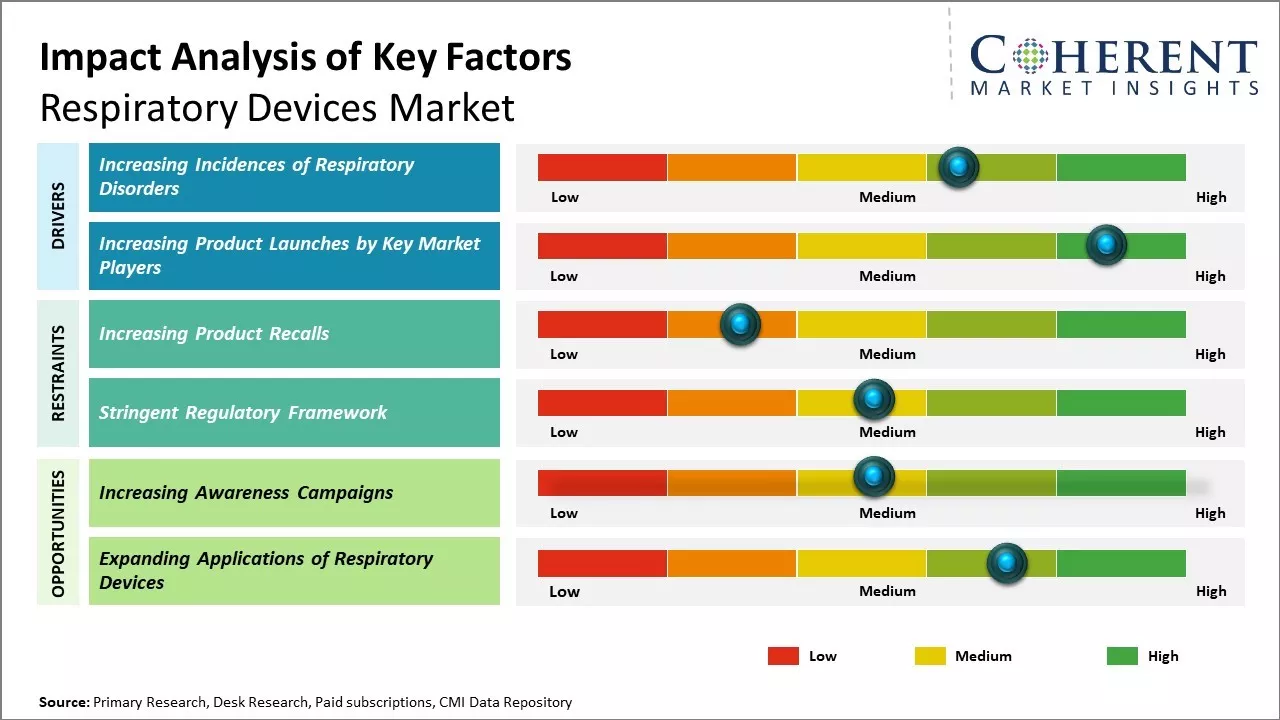
Increasing Incidences of Respiratory Disorders
The global respiratory devices market is being driven higher by the increasing prevalence of various respiratory diseases worldwide. Conditions, such as chronic obstructive pulmonary disease (COPD) and asthma continue affecting millions of people and are major health issues in both developed and developing countries. COPD in particular has risen to become a leading cause of death globally in recent years. The high instance of respiratory illnesses can be attributed to factors like rising air pollution levels, growing elderly population, and changing lifestyles involving smoking and lack of physical activity. For instance, according to the World Health Organization fact sheet released in March 2023, over 200 million individuals are affected by chronic obstructive pulmonary disease (COPD), causing 3.23 million deaths in 2019 and 65 million experiencing moderate-to-severe COPD. Moreover, sleep-disordered breathing affects around 1-6% of the adult population, which amounts to more than 100 million people. The urgent need to address this growing respiratory disease burden through effective therapeutic and diagnostic solutions is a key market growth catalyst. Device manufacturers are ramping up their product portfolios to meet the provider demand arising from the expanding patient pools.
Get actionable strategies to beat competition: Request Free Sample
Increasing Product Launches by Key Market PlayersIncreasing adoption of organic growth strategies, such as product launches, is expected to drive the market growth over the forecast period. For instance, on February 13, 2024, Masimo, a health technology company, announced the launch of Over-the-Counter fingertip pulse oximeter for medical use without a prescription.

To learn more about this report, Request Free Sample
Market Challenges – Increasing Product RecallsIncreasing number of product recalls by regulatory authorities, such as the U.S. Food and Drug Administration, are expected to hinder the market growth over the forecast period. For instance, in September 2022, the U.S. Food and Drug Administration recalled a class I BiPAP machine, a type of ventilator, which is manufactured by Koninklijke Philips N.V., a medical device company. The device was recalled because the machines may contain plastic in their motors that release volatile organic compounds which have the risk for users inhaling dangerous volatile organic compounds and the plastic could also cause device failure.
Market Opportunities – Increasing Awareness Campaigns
Increasing adoption of inorganic growth strategies such as launch of awareness campaigns. For instance, in November 2022, Reckitt Benckiser Group plc, a U.K.-based multinational consumer goods company, launched a new education and early detection campaign for patients with undiagnosed chronic obstructive pulmonary disease (COPD).
Discover high revenue pocket segments and roadmap to it: Request Free Sample
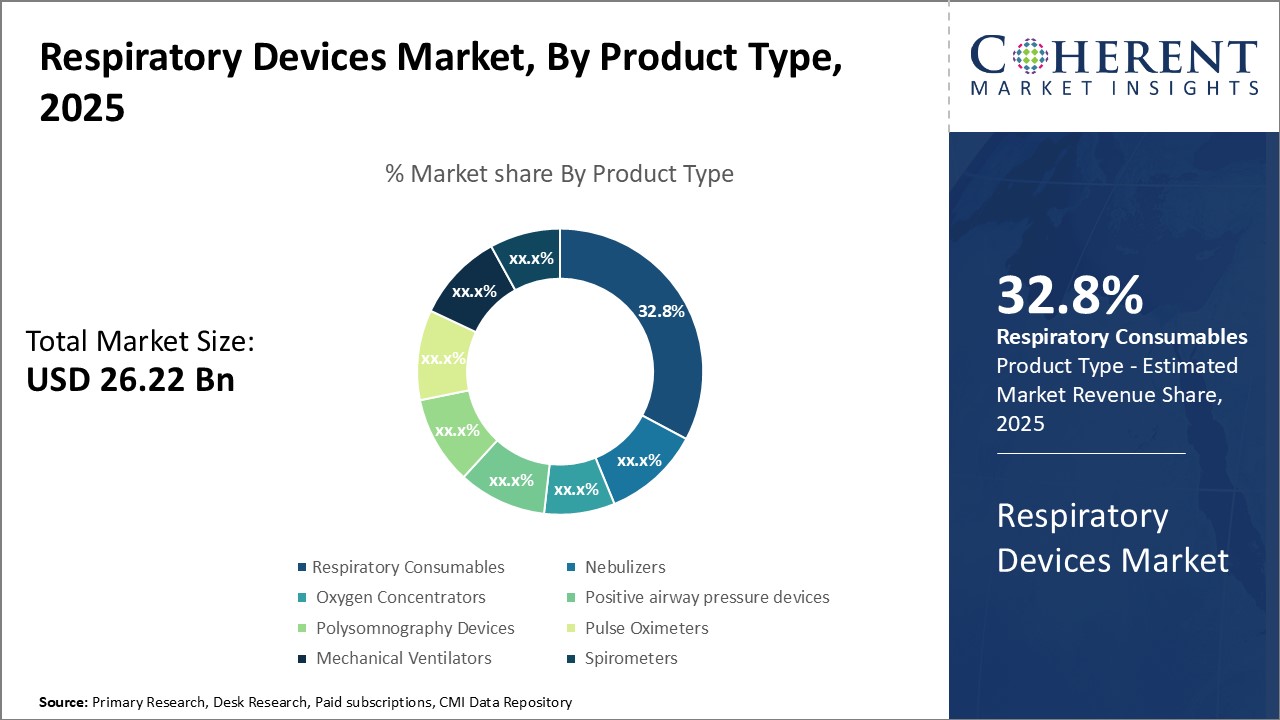
Insights, By Product Type: Meeting Demand for Home Healthcare Oxygen DeliveryThe product type segment includes respiratory consumables, nebulizers, oxygen concentrators, positive airway pressure devices, polysomnography devices, pulse oximeters, mechanical ventilators, and spirometers. The oxygen concentrators sub-segment is estimated to hold 33.8% of the market share in 2026 owing to the growing demand for home healthcare solutions among respiratory patients. As chronic respiratory conditions like COPD become more prevalent, there is an increasing need for convenient oxygen delivery methods that allow patients to maintain an active lifestyle outside of healthcare facilities. Oxygen concentrators address this need well by concentrating oxygen from ambient air and supplying a continuous flow of oxygen to patients through nasal cannulas or masks. Being lightweight and portable, oxygen concentrators enable independent oxygen delivery anywhere and do not require frequent refills like heavy oxygen cylinders. This has led to a growing preference for oxygen concentrators over other oxygen delivery methods among both patients and healthcare providers. For patients, oxygen concentrators provide mobility and freedom of movement which improves their quality of life. They no longer have to be tied to their homes or rely on heavy oxygen cylinders. This has a positive psychological impact. For healthcare systems, oxygen concentrators reduce costs associated with frequent cylinder refills and deliveries. They enable shifting of long-term oxygen care to the home instead of hospitals or nursing facilities. This has boosted the uptake of oxygen concentrators among chronic respiratory patients globally.
To learn more about this report, Request Free Sample
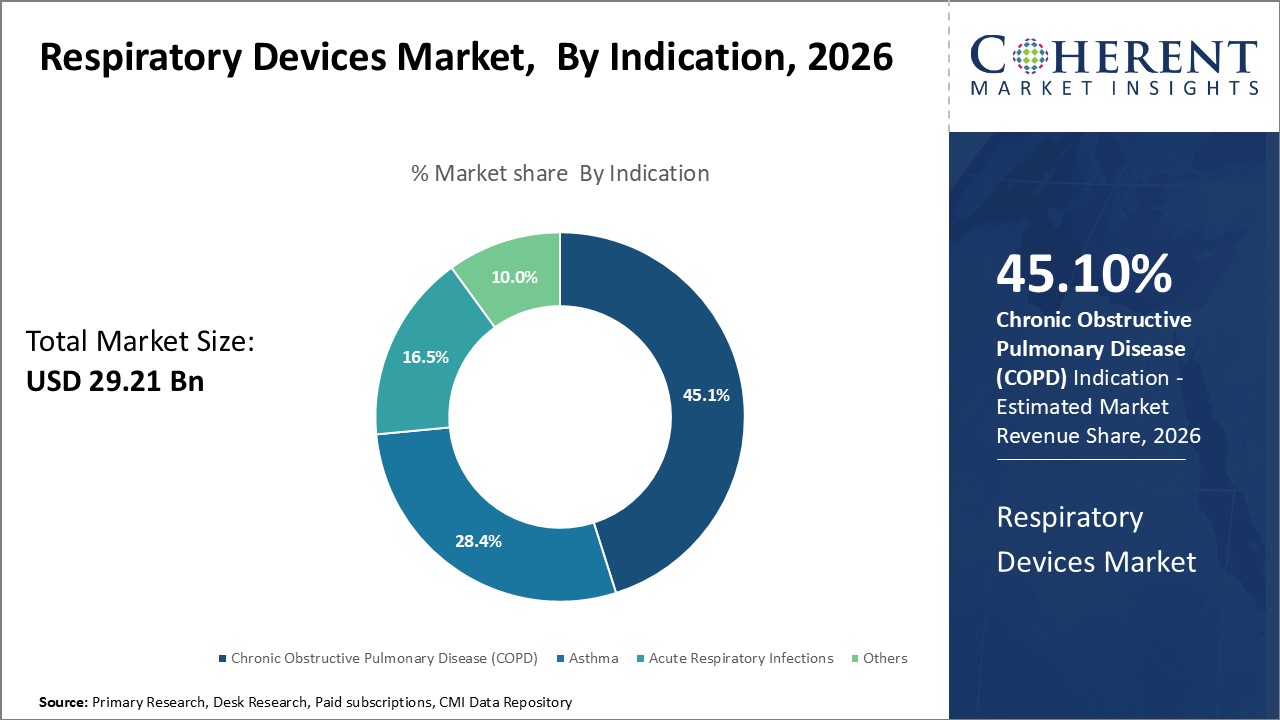
Insights, By Indication: Increasing Prevalence of COPD
The indication segment includes chronic obstructive pulmonary disease (COPD), asthma, acute respiratory infections, and others. The chronic obstructive pulmonary disease (COPD) sub-segment is estimated to hold 45.1% of the market share in 2026 to rising cases of COPD worldwide. COPD refers to a group of progressive lung diseases characterized by airflow obstruction and breathing-related problems. The primary risk factors for COPD development include smoking, air pollution, and occupational dust/chemical exposure. According to the latest World Health Organization estimates, over 340 million people worldwide are living with COPD, with the condition being among the top causes of mortality. While tobacco smoking has been the dominant cause historically, other factors like aging population and growing air pollution levels in urban areas are significantly increasing COPD prevalence globally. Severe COPD patients rely on long-term oxygen therapy, nebulizers and other respiratory devices to help manage their condition. Even in mild-moderate stages, periodic exacerbations often require acute care using inhalers, ventilators, or oxygen concentrators. With limited pharmacological treatment options available, respiratory devices therefore form the mainstay of both routine and crisis management for most COPD patients.
Insights, End User: Central Role in Acute Care Drives the Hospital Demand
The end user segment includes hospitals, ambulatory surgical centers, clinics, and others. The hospitals sub-segment is estimated to hold 42.2% of the market share in 2026 owing to the criticality of respiratory devices in acute care settings. Respiratory devices finds immense utility in hospital departments such as emergency rooms, intensive care units, and operation theatres where constant evaluation of patients' oxygen saturation is imperative. Their non-invasive nature and quick readings ensure minimal disruptions to patient care. Additionally, the larger scale of operations in hospitals translates to higher volumes of respiratory procurement.
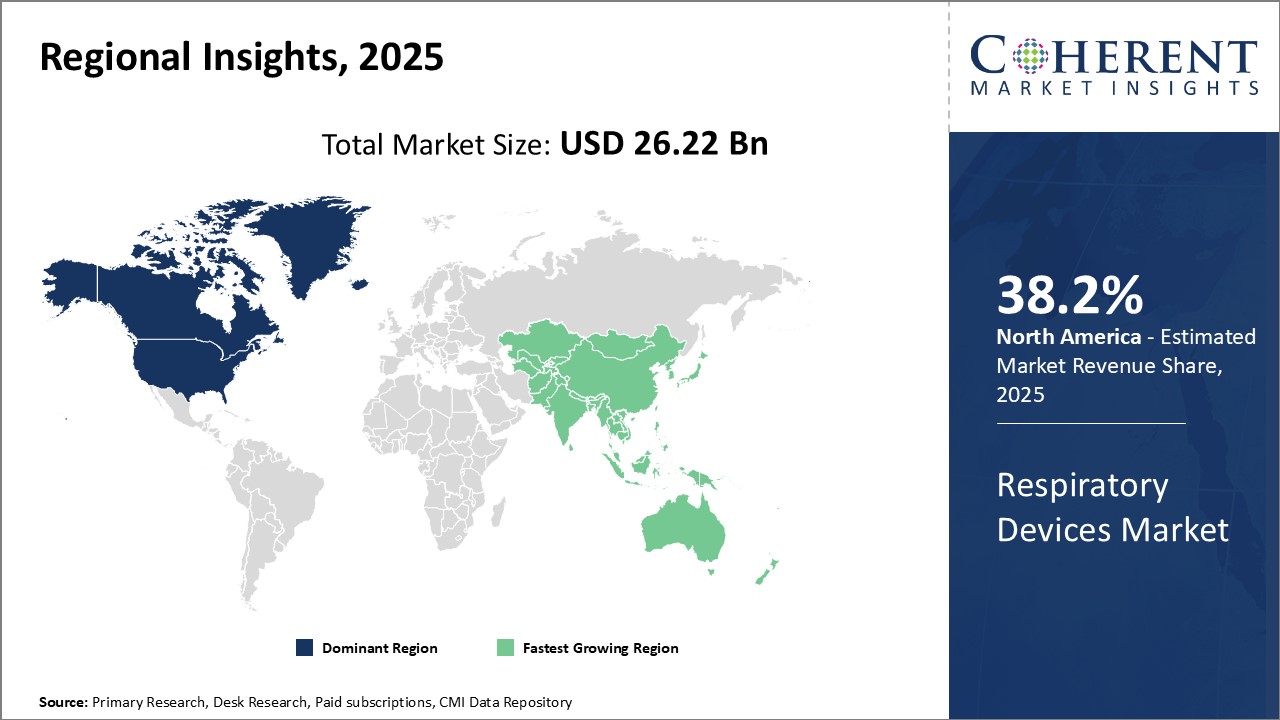
Need a Different Region or Segment? Request Free Sample
North America remains the dominant region in the global genome engineering market and is estimated to hold 38.2% of the market share in 2026 due to a strong presence of major device manufacturers, established healthcare infrastructure, and availability of favorable reimbursement policies. The U.S. accounts for the largest share owing to presence of major players and advanced healthcare system. These players have strong distribution network across the region ensuring easy accessibility of devices. Moreover, government initiatives to spread awareness about respiratory conditions have boosted adoption rate of respiratory therapies.
The Asia Pacific region is emerging as the fastest growing market for respiratory devices. Rapidly developing healthcare infrastructure, growing geriatric population susceptible to respiratory diseases and improving access to healthcare are some key factors contributing to the high growth. China and India are expected to lead the market in the coming years supported by huge patient population, increasing healthcare expenditure, and penetration of medical insurance. Manufacturers are focusing on these developing nations by establishing local manufacturing plants to cater growing demand and ensure availability of affordable devices. Several multinational companies have also entered partnerships with local players to leverage their distribution and insights of local market.
Respiratory Devices Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 29.21 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 11.4% | 2033 Value Projection: | USD 62.19 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Medtronic, Hamilton Medical, ResMed, Masimo, Teleflex Incorporated, Fisher & Paykel Healthcare Limited, Koninklijke Philips N.V., ICU Medical, Inc., Dragerwerk AG & Co. KGaA, TytoCare Ltd., VYAIRE, Medline Industries, Inc., OMRON Corporation, TNI medical AG, and Xplore Health Technologies Pvt. Ltd. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
*Definition: Respiratory devices are medical devices that are used to assist a patient suffering from breathing problems. These devices are used in the management and treatment of disorders such as fibrosis, asthma, Chronic Obstructive Pulmonary Disease (COPD), and Acute Respiratory Distress Syndrome (ARDS). There are a variety of diseases that benefit from these advanced tools such as oxygen concentrators, nebulizers, ventilators, etc. The respiratory device assists with the removal of mucus from the airways and improves pulmonary functions.
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients