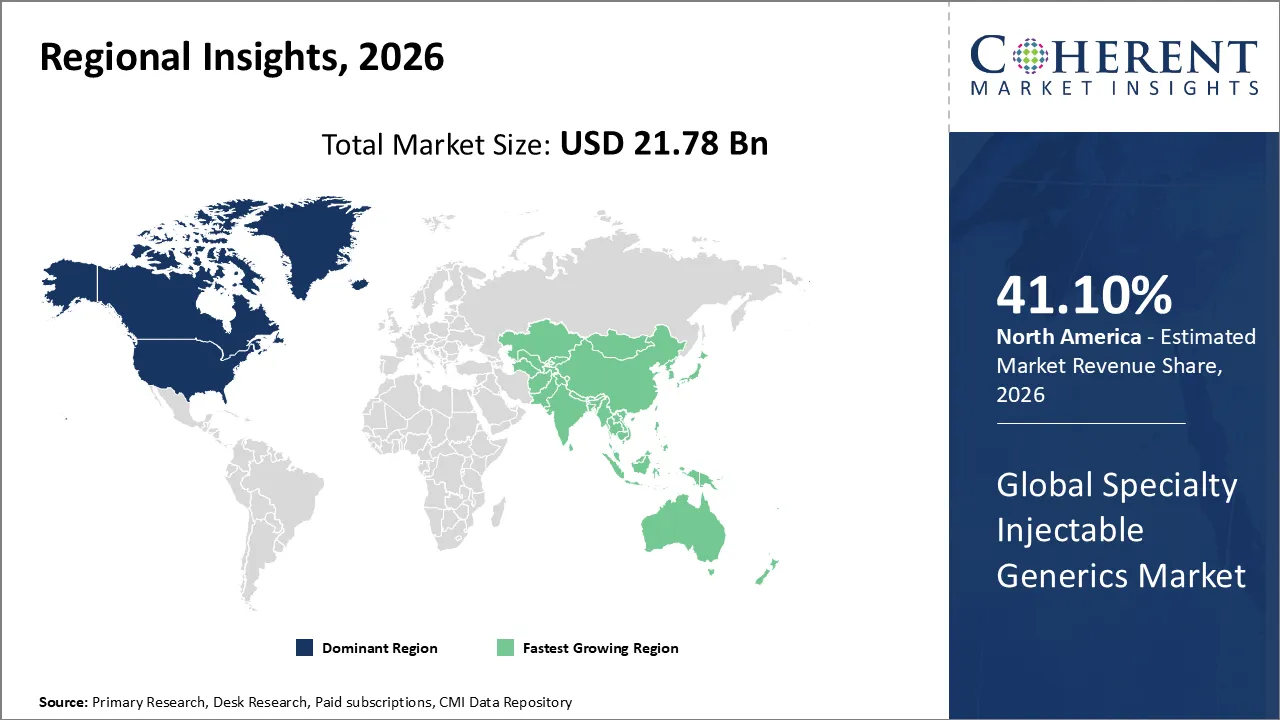
The Specialty Injectable Generics Market is estimated to be valued at USD 21.78 Bn in 2026 and is expected to reach USD 36.62 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 7.8% from 2026 to 2033.
The specialty injectable generics market delivers complex injectable therapies across hospital, oncology, and chronic care settings. Increasing demand for cost-effective alternatives to branded biologics, along with the rising prevalence of chronic and life-threatening diseases, drives market growth. Advances in sterile manufacturing and delivery technologies accelerate adoption, while supportive regulatory frameworks and expanding hospital and outpatient infrastructure enable wider use. The market offers biosimilars, oncology injectables, and other specialty generics, promoting affordable, accessible, and clinically effective injectable treatments worldwide.
|
Current Events |
Description and its impact |
|
Regulatory and Policy Changes in Key Markets |
|
|
Supply Chain Disruptions and Raw Material Constraints |
|
|
Macroeconomic and Geopolitical Influences |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Biologics hold the largest market share of 53.3% in 2026. The Specialty Injectable Generics Market expands as companies capitalize on the expiration of patents for high-value biologic drugs, offering cost-effective alternatives. Rising cases of chronic and complex diseases boost demand for advanced injectable therapies, while healthcare systems actively pursue affordable treatment options. Manufacturers improve production efficiency and patient convenience through innovations in formulation, delivery, and sterile manufacturing technologies. Supportive regulatory pathways and increasing clinical acceptance further drive adoption, enabling biologic generics to reach a wider patient population worldwide. In December 2025, Sun Pharmaceutical Industries, a global specialty pharmaceutical leader, unveiled Ilumya (tildrakizumab) in India for treating moderate to severe plaque psoriasis. This novel biologic targets IL-23 and has achieved blockbuster status globally, with availability in 35 countries, including the United States.
Oncology expected to hold largest market share of 37.7% in 2026. The Oncology segment of the Specialty Injectable Generics Market expands as hospitals and clinics adopt affordable alternatives to costly branded cancer therapies. Rising cancer rates and increasing demand for targeted injectable treatments drive this growth. Manufacturers enhance efficiency by applying advances in sterile production and biosimilar development, while regulatory frameworks accelerate approval of complex generics. Healthcare providers actively prioritize cost-effective oncology solutions, improving accessibility and promoting broader integration of injectable generics into cancer treatment protocols worldwide.
In November 2025, Zydus Lifesciences Ltd announced that it received final approval from the US Food and Drug Administration for its generic Leuprolide Acetate injection, indicated for the palliative treatment of advanced prostate cancer.
Hospital Pharmacies acquired the prominent market share of 46.6% in 2026. Hospital pharmacies actively boost the Specialty Injectable Generics Market by supplying cost-effective, high-demand injectable therapies for both inpatient and outpatient care. They secure affordable generics and biosimilars through centralized procurement and large-scale purchasing, ensuring critical treatments remain continuously available. Rising rates of chronic and complex diseases increase their reliance on injectable therapies, while advances in sterile manufacturing and convenient delivery systems enhance operational efficiency. By integrating these products into formularies and clinical protocols, hospital pharmacies actively expand the adoption of specialty injectable generics. For instance, in December 2025, Danish drugmaker Novo Nordisk unveiled diabetes drug Ozempic in India.
To learn more about this report, Request Free Sample
North America dominates the overall market with an estimated share of 41.10% in 2026. The North America Specialty Injectable Generics Market expands as hospitals, oncology centers, and chronic care facilities increasingly adopt affordable alternatives to expensive branded injectables. Rising cases of complex and chronic diseases drive higher usage, while regulatory frameworks actively facilitate faster approval of biosimilars and complex generics. Strong healthcare infrastructure and centralized procurement systems allow efficient distribution and adoption. Manufacturers apply advances in sterile production and innovative delivery technologies, and growing clinical acceptance further strengthens the market, establishing North America as a leading region for specialty injectable generics. For instance, in November 2025, Lupin Limited has introduced Risperidone Long-Acting Injectable in the U.S. market with 180-day Competitive Generic Therapy exclusivity. The antipsychotic treatment is available in 25 mg, 37.5 mg, and 50 mg strengths.
The Asia Pacific Specialty Injectable Generics Market grows as hospitals and specialty clinics increasingly adopt advanced injectable therapies while healthcare systems modernize. Greater awareness of cost-effective alternatives drives use, especially in oncology, chronic, and infectious disease treatments. Governments and regulatory authorities actively support biosimilar and generic approvals, and local manufacturers strengthen supply chains. Rising demand for outpatient and home-care injectable solutions, along with advancements in sterile manufacturing and delivery technologies, actively fuels the region’s rapid development in specialty injectable generics.
The United States Specialty Injectable Generics Market grows as hospitals, clinics, and specialty care centers actively adopt cost-effective alternatives to branded injectables. Increasing cases of chronic and complex diseases drive higher demand for injectable therapies in oncology, autoimmune, and critical care treatments. Regulatory authorities support the market through streamlined approval pathways, promoting the launch of biosimilars and complex generics, while manufacturers enhance efficiency and patient convenience with advanced production and delivery technologies. Centralized procurement and formulary integration further boost adoption, positioning the U.S. as a key market for specialty injectable generics. For instance, Avenacy has launched Dexmedetomidine Injection, USP in the United States as an FDA-approved therapeutic generic equivalent to Precedex®.
China’s Specialty Injectable Generics Market grows rapidly as hospitals and specialty clinics actively adopt affordable injectable therapies to address increasing patient demand. Rising cases of chronic, oncologic, and infectious diseases drive extensive use of generics and biosimilars. The government and regulatory authorities actively support approvals through evolving policies, while local manufacturers strengthen supply chains. Advances in sterile production, innovative delivery systems, and hospital procurement practices improve accessibility, positioning China as a dynamic and fast-developing market for specialty injectable generics.
The market increasingly emphasizes biosimilars and complex biologic injectables as patents on high-cost branded drugs expire. Manufacturers focus on replicating efficacy and safety profiles while offering cost-effective alternatives. Healthcare providers actively adopt these therapies to manage rising treatment costs and expand patient access. The trend also reflects growing confidence among clinicians and regulators in biosimilar safety, fueling broader acceptance and integration into hospital formularies and outpatient care protocols worldwide.
Hospitals, oncology centers, and specialty clinics drive demand for injectable generics due to high patient throughput and critical treatment needs. Centralized procurement and formulary management support large-scale adoption, while end-users prioritize reliable supply and affordability. Increased integration into clinical workflows ensures that injectable generics are used efficiently for chronic, acute, and specialty care treatments, reinforcing hospitals and specialty centers as key channels shaping the market’s growth and long-term sustainability.
The escalating costs of branded biologics and specialty injectables present a significant opportunity for generics and biosimilars. Hospitals, clinics, and outpatient centers actively seek affordable alternatives to maintain patient access while managing budgets. Manufacturers can leverage this demand by introducing clinically equivalent, lower-cost injectable therapies that address chronic, oncology, and critical care needs, creating value for healthcare providers, payers, and patients while capturing a growing share in a cost-conscious market.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 21.78 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7.8% | 2033 Value Projection: | USD 36.62 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Pfizer, Inc., Teva Pharmaceutical Industries Ltd., Novartis AG, Baxter International Inc., Mylan N.V., Fresenius SE & Co. KGaA, Endo International Plc., Hikma Pharmaceuticals PLC, Lupin Ltd., Sagent Pharmaceuticals, Inc., Cipla Limited , Sun Pharmaceutical Industries Limited, and Dr. Reddy's Laboratories. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients