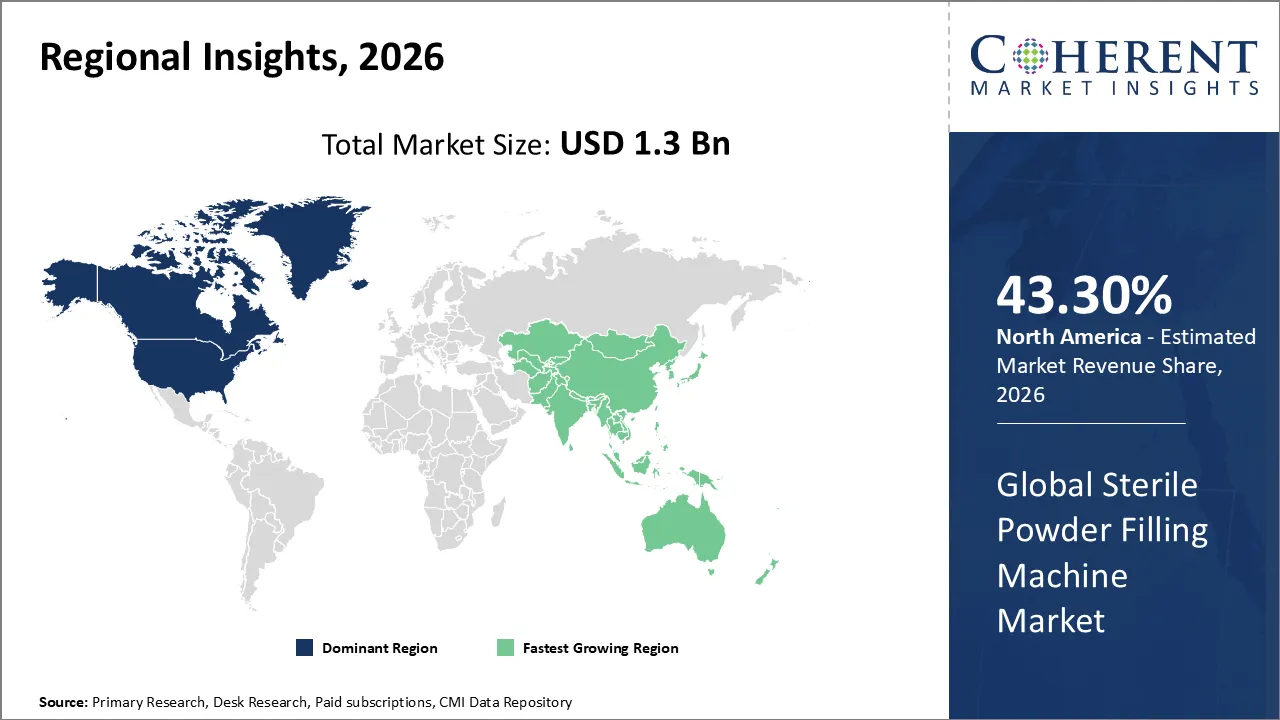
The Sterile Powder Filling Machine Market is estimated to be valued at USD 1.3 Bn in 2026 and is expected to reach USD 2.7 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 5.1% from 2026 to 2033.
The Sterile Powder Filling Machine market focuses on machines that precisely and hygienically fill powdered pharmaceutical products into vials, syringes, and cartridges while maintaining strict sterile conditions. Growing production of complex injectable drugs and vaccines, combined with the pharmaceutical industry’s emphasis on contamination‑free processing, fuels market expansion. Manufacturers increasingly adopt advanced automated filling technologies, and pharmaceutical and biotechnology companies actively drive demand for reliable, high‑precision sterile filling solutions across global production facilities.
|
Current Events |
Description and its impact |
|
Geopolitical and Regulatory Developments |
|
|
Technological Advancements and Industry Innovation |
|
|
Regional Market Expansion and Emerging Markets |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Syringes acquired the prominent market share of 36.6% in 2026. The rising demand for syringe‑ready formats fuels the Sterile Powder Filling Machine market as manufacturers produce more prefilled, user‑friendly delivery systems. Pharmaceutical companies invest in machines that fill sterile powders into syringes because healthcare providers and patients prioritize accuracy, convenience, and lower contamination risk. Advancements in automation and precision dosing reinforce this shift, while expanding production of biologics and specialty injectables drives the need for dependable, aseptic syringe filling solutions across the pharmaceutical industry. For instance, in January 2025, Vetter, a global Contract Development and Manufacturing Organization (CDMO), has announced to launch the latest version of its proprietary V-OVS syringe closure system. The new V-OVS next builds on the proven performance of the original system, enhancing support for and differentiation of a wide range of sterile injectable products.
20 to 60 Fills/Minute hold the largest market share of 36.6% in 2026. The 20 to 60 fills per minute segment of the Sterile Powder Filling Machine market thrives as pharmaceutical companies seek mid‑level production flexibility for small to medium batches. Manufacturers rely on this speed range to produce niche biologics, specialty injectables, and pilot batches that demand precision and strict sterility. Increasing adoption of automated, contamination‑resistant filling systems and the expanding use of dry powder formulations drive companies to invest in reliable machines that deliver accurate dosing while maintaining moderate throughput.
Nuclear Medicine expected to hold largest market share of 41.1% in 2026. The expansion of nuclear medicine drives the Sterile Powder Filling Machine market by increasing demand for aseptic systems used in radiopharmaceuticals and injectable therapies. Manufacturers produce sterile pharmaceutical agents for nuclear diagnostic procedures (such as PET and SPECT) and targeted radiotherapies, requiring precise dosing and contamination‑free handling. Healthcare providers and pharmaceutical companies implement automated, closed‑system processes to enhance safety, comply with regulations, and support emerging theranostic treatments, prompting greater investment in advanced, high‑quality sterile filling technologies.
To learn more about this report, Request Free Sample
North America dominates the overall market with an estimated share of 43.3% in 2026. In North America, strong pharmaceutical and biotech activity drives the Sterile Powder Filling Machine market, with the United States leading demand through its advanced healthcare infrastructure and strict regulatory oversight that encourages manufacturers to adopt aseptic solutions. Producers implement automated, flexible systems to handle various sterile formats, including vials and syringes, while Canadian manufacturers upgrade their production lines to maintain quality standards. Investments in biologics, vaccines, and modernization of fill‑finish operations further propel market growth across the region. For instance, Nelipak, a Netherlands-based healthcare packaging supplier, has opened its first North American flexible packaging facility in Winston-Salem, NC. The site actively produces high-quality sterile-barrier packaging solutions, supporting customers’ sustainability goals and business continuity.
In the Asia Pacific region, pharmaceutical manufacturers in China, India, Japan, and Southeast Asia are expanding production of sterile injectables, vaccines, and biosimilars, driving growth in the Sterile Powder Filling Machine market. Companies invest in healthcare infrastructure and quality control systems to upgrade automated and mid‑speed filling lines that maintain aseptic conditions. Regional firms implement flexible machines to serve domestic and export markets, while cost‑effective solutions and the rise of biotech hubs promote wider adoption of sterile powder filling equipment across the region. For instance, WuXi STA, a subsidiary of WuXi AppTec, has launched a new parenteral formulation manufacturing line at its Wuxi, China site. The advanced sterile lipid nanoparticle (LNP) facility enables the site to deliver formulation development and manufacturing for oligonucleotides and their conjugates.
In the United States, strong pharmaceutical and biotechnology production drives the Sterile Powder Filling Machine market as manufacturers expand aseptic capacity to supply sterile injectables, vaccines, and biologics. U.S. companies adopt automated, closed‑system filling equipment to meet strict FDA sterility and quality standards while enhancing efficiency and reducing contamination. Contract manufacturers increase fill‑finish capabilities to strengthen domestic supply and address growing demand for flexible, multi‑format sterile powder filling solutions across the industry.
In India, local pharmaceutical manufacturers are driving growth in the Sterile Powder Filling Machine market by scaling up sterile injectable and vaccine production to meet domestic and export demand. Companies adopt automated, aseptic powder filling systems to comply with global quality standards and WHO‑GMP guidelines, while government initiatives and infrastructure investments enable technological upgrades. Indian biotech and pharmaceutical plants increasingly implement cost‑effective, flexible machines capable of handling diverse formulations and batch sizes, supporting broader adoption of advanced sterile filling solutions.
Manufacturers are increasingly adopting automated sterile powder filling machines with advanced control systems to improve precision, reduce human error, and enhance production efficiency. Smart technologies, including IoT-enabled monitoring, predictive maintenance, and real-time data analytics, allow companies to optimize operations, maintain consistent quality, and ensure compliance with stringent regulatory standards. This trend reflects the broader move toward Industry 4.0 in pharmaceutical manufacturing, where connected and intelligent systems support aseptic production and reduce operational risks.
The market is witnessing growing adoption of prefilled syringes, cartridges, and vials to meet patient convenience and dosage accuracy requirements. Manufacturers increasingly prefer machines that can switch between multiple container types and batch sizes with minimal downtime. This flexibility helps pharmaceutical companies serve niche biologics, vaccines, and specialty injectables while maintaining sterility, supporting small and large-scale production simultaneously, and reducing operational costs linked to frequent equipment changeovers.
The rising development of biologics, vaccines, and high-value specialty injectables presents a significant opportunity for sterile powder filling machine manufacturers. These products require precise dosing, strict aseptic conditions, and contamination-free handling. Companies can expand offerings with advanced automated machines that handle sensitive powders and support small to mid-sized batches, enabling pharmaceutical firms to meet increasing demand while ensuring product quality and compliance with regulatory standards, creating new avenues for market penetration and innovation.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1.3 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 5.1% | 2033 Value Projection: | USD 2.7 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Duke Technologies, Adinath International, Multi Pack Machinery, Harsiddh Engineering, SB Machines, OPTIMA Packaging, Ambica Pharma Machines Pvt. Ltd, N.K. INDUSTRIES, COZZOLI MACHINE COMPANY, METACRAFTS Packaging Machinery, Romaco S.r.l., Lodha International LLP, SHIBUYA CORPORATION, MAR USA Inc., PPS A/S, Parth Engineers & Consultant, Bausch + Ströbel Maschinenfabrik Ilshofen GmbH + Co. KG, Syntegon, DEC Group, Dosa Srl, Harro Höfliger Verpackungsmaschinen GmbH, Tofflon Science, M&O Perry Industries, Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients