Global telmisartan market is estimated to be valued at USD 4.54 Bn in 2026 and is expected to reach USD 6.01 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 4.1% from 2026 to 2033.
Discover market dynamics shaping the industry: Request Free Sample
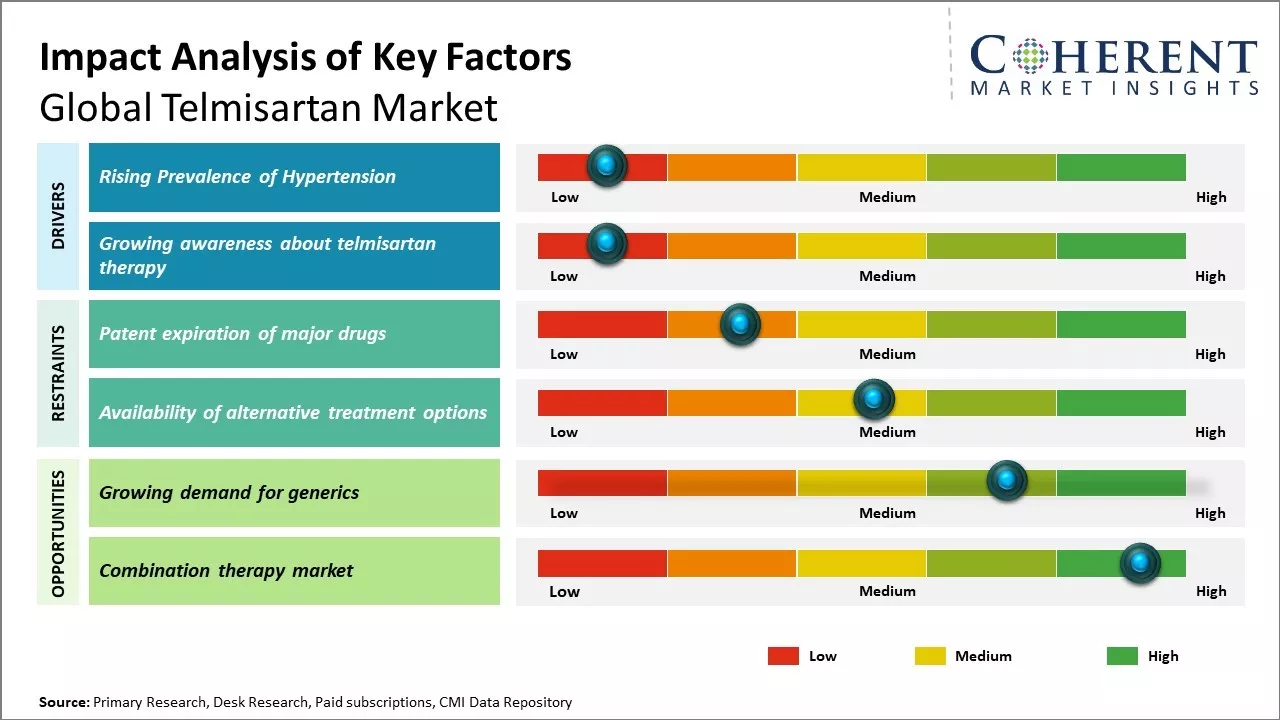
Global telmisartan market growth is driven by increasing demand for effective drugs for treatment of hypertension. Telmisartan is mainly prescribed for the primary treatment of hypertension and can effectively lower blood pressure levels. It is also used to reduce the risk of cardiovascular events for patients who cannot tolerate ACE inhibitors. Favorable government policies for prevention and control of hypertension along with rising healthcare spending can also drive the market growth. However, availability of generic substitutes once patents expire can hamper the market growth.
Rising prevalence of hypertension
Rising prevalence of hypertension can drive the market growth. According to the World Health Organization, hypertension is one of the leading causes of preventable death worldwide as it significantly raises the risks of heart disease and stroke. High blood pressure increases the workload of the heart which can eventually lead to damage of key organs and vessels in the body. If uncontrolled for many years, it progressively impairs the function of arteries and major organs. According to the data from National Health Surveys, hypertension affects over 1.13 billion people worldwide. Its occurrence varies across countries and regions depending on lifestyle habits and risk factors prevalent in different populations. Rapid aging of populations and sedentary jobs and unhealthy dietary patterns linked to urbanization contribute to prevalence of hypertension. More people are now consuming calorie-dense processed foods high in sodium, sugar, and unhealthy fats. At the same time, physical activity levels have fallen sharply in both developed and developing nations. These lifestyle transitions have contributed significantly to the hypertension epidemic. Considering the enormity of the problem and its severe ramifications, it has become imperative for healthcare systems globally to step up screening as well as provide effective long-term treatment solutions. Telmisartan has established itself as one of the most attractive medical options due its strong efficacy and tolerability profile. Its widespread adoption by physicians can boost its demand. For instance, as per a joint study by World Health Organization (WHO) and Imperial College London, the number of adults aged 30–79 years with hypertension has increased from 650 million to 1.28 billion over the past three decades. Published in The Lancet Journal, the study underscores that nearly half of these individuals were unaware of their condition, highlighting significant gaps in diagnosis and treatment.
Get actionable strategies to beat competition: Request Free Sample
Growing awareness about telmisartan therapy
Global telmisartan market growth is driven by growing awareness about its effectiveness and safety in the treatment of hypertension. Telmisartan is an angiotensin receptor blocker (ARB) that is widely prescribed by cardiologists and vascular physicians for management of high blood pressure. It is known to be well-tolerated with minimal side effects as compared to other anti-hypertensive drugs. For instance, as per the report published by the American Heart Association in 2021, long term use of telmisartan lowered the risk of stroke and dementia in elderly hypertensive patients. As per a meta-analysis published by European Journal of Preventive Cardiology in 2020, telmisartan is superior over other ARBs in reducing cardiovascular and all-cause mortality. Growing evidence of telmisartan's organ protective qualities beyond blood pressure control has increased physician's preference for the drug. Telmisartan has also gained traction due to its affordability and once-daily dosing regimen. This ensures high patient compliance which is crucial for effective management of lifelong conditions like hypertension. According to the data from World Health Organization in 2022, non-adherence to medication is a major public health challenge especially in developing nations. The simplicity and low cost of telmisartan therapy makes it suitable for improving access and adherence to treatment in resource-constrained settings.
Key Takeaways from Analyst:
Global telmisartan market growth is driven by increasing prevalence of hypertension worldwide. Rising geriatric population susceptible to hypertension can boost demand for telmisartan as age is a primary risk factor. However, availability of generic alternatives once patents of major brands expire can hamper the market growth. Stringent regulations pertaining to approval of generic drugs can limit rapid uptake of low-cost substitutes.
Asia Pacific region dominated the global market share last year due to the large patient pool suffering from hypertension in countries like China and India. Within the region, India is projected to encounter highest growth, owing to rising healthcare expenditure and expanding insurance coverage. Meanwhile, markets in North America and Western Europe will continue expanding at a modest pace as focus shifts to combination therapies.
Rising demand for fixed-dose combinations offering enhanced efficacy and adherence can offer lucrative opportunities for telmisartan market players. Furthermore, approval of telmisartan for treatment of diabetic kidney disease indication beyond hypertension has strengthened its clinical profile. However, long patent exclusivity maintains bargaining power of innovator companies currently.
Market Challenges: Patent expiration of major drugs
The patent expiration of major drugs in the recent years can hamper the global telmisartan market growth. When blockbuster drugs lose their patent protection, pharmaceutical companies experience a sharp decline in revenues from these products as cheaper generic versions enter the market. For instance, the patent for telmisartan drug expired in many regions including the U.S. in December 2012, which allowed other companies to manufacture and sell generic versions. Major companies such as Boehringer Ingelheim which developed and marketed telmisartan under the brand name Micardis witnessed decline in sales gradually as cheaper generics gained market share. As per data from the U.S. Food and Drug Administration, the approval rate of generic drug applications had increased steadily after 2012 with numerous generic manufacturers launching biosimilar versions of telmisartan hitting the market. The increased availability of low-cost generics created intense price competition which made it difficult for innovator companies to sustain earlier revenue levels. While the overall consumption and volume sales of telmisartan increased due to widespread affordability, the average selling price kept declining annually impacting the profit margins severely. This situation restrained companies pursuing more innovation in this segment.
As per data from World Health Organization, around 60% of the global consumption of telmisartan in 2020 was fulfilled by generic drug manufacturers as compared to only 10% prior to 2012 patent expiration reflecting the shift towards lower-cost alternatives over time.
Market Opportunities: Growing demand for generics
Growing demand for generic drugs around the world offers opportunity for companies in the global telmisartan market. In the recent years, there has been a significant shift towards more affordable healthcare options as the costs of branded pharmaceuticals continue to rise rapidly. More patients and governments are looking to treat health conditions while also managing tight budgets. This preference for lower-cost medicines has fueled the expansion of the generic drug industry.
Telmisartan is a widely used angiotensin receptor blocker (ARB) that treats hypertension and reduces the risk of cardiovascular disease. As the patents on the major branded versions start to expire, many new generic alternatives enter the market. According to data from the World Health Organization, over 50% of all drug prescriptions dispensed worldwide are now filled with a generic medication. This trend is expected to gain further momentum in developing nations struggling with escalating healthcare expenditures. As the population ages and non-communicable diseases like heart disease and diabetes rise globally, the treatment needs will increase substantially. Affordable versions of essential drugs like telmisartan will be pivotal in providing universal access and improving health outcomes. Companies with a strong focus and capabilities in generics will be best positioned to tap into this massive potential. These can develop high quality copies, obtain regulatory approvals, aggressively market their telmisartan offerings and distribute them widely.
Discover high revenue pocket segments and roadmap to it: Request Free Sample
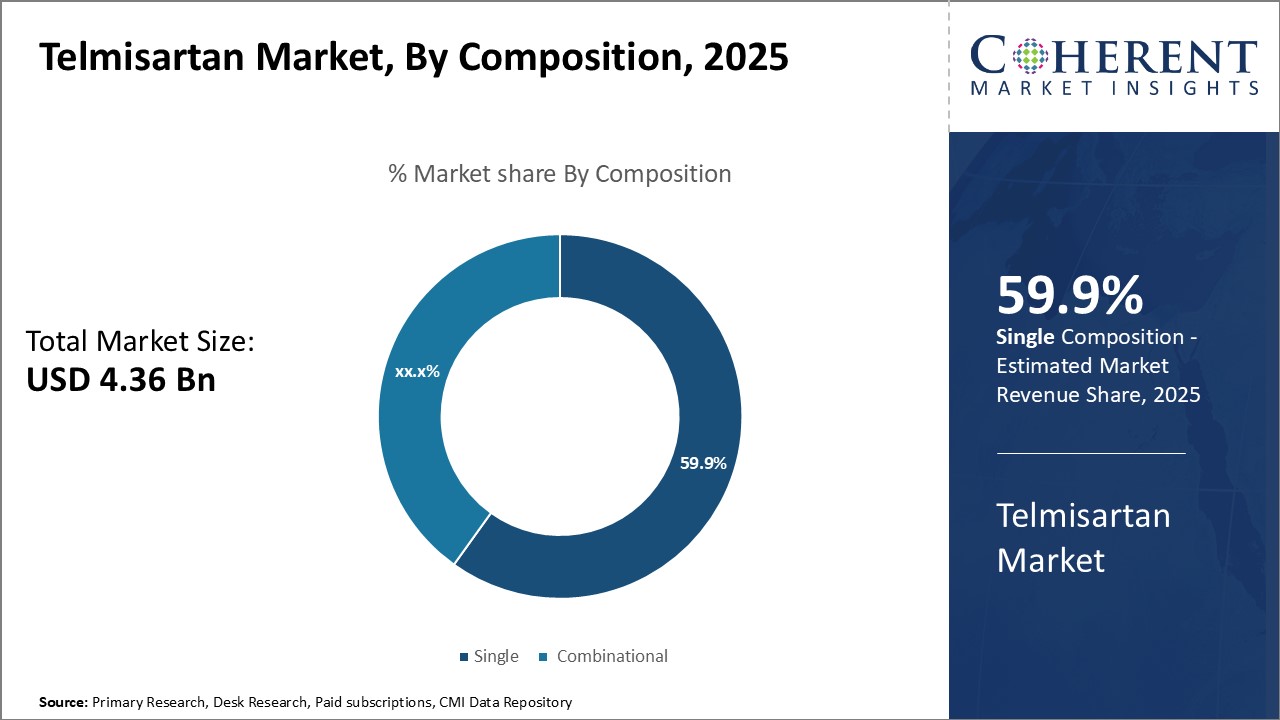
By Composition - Favorable safety and efficacy profile drives growth of single composition segment
By composition, single segment is estimated to contribute the highest market share of 60.9% in 2026, due to its favorable safety and efficacy profile. Telmisartan is generally well-tolerated when used as a monotherapy for hypertension and other cardiovascular conditions. Compared to combination therapies, single agent telmisartan reduces the risk of drug-drug interactions and other safety issues that can arise from polypharmacy. It also provides a simple dosing regimen that improves medication adherence. Furthermore, clinical studies indicate telmisartan as a single agent is as effective as ACE inhibitor or calcium channel blocker monotherapies in achieving target blood pressure levels. Its long duration of action allows once-daily dosing, enhancing convenience. Majority of patients, especially in the early stages of treatment, prefer single therapies to minimize pill burden. This drives strong physician preference and patient demand for the telmisartan single agent over alternatives.
To learn more about this report, Request Free Sample
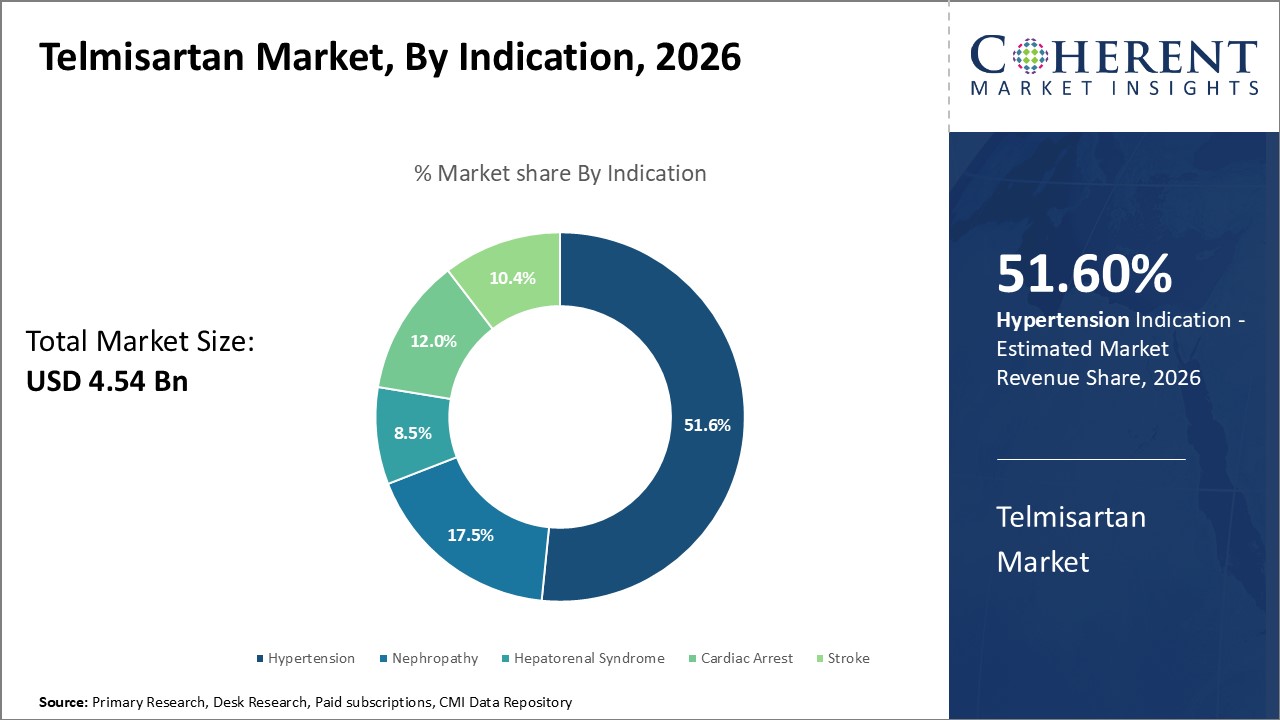
By Indication - Hypertension indication powers growth in indication segment
By indication, hypertension segment is estimated to contribute the highest market share of 51.6% in 2026 due to the huge patient pool and clinical need. Telmisartan is a well-established antihypertensive therapy and one of the most commonly prescribed angiotensin II receptor blockers worldwide for hypertension. It provides effective blood pressure control and cardiac protection with a good tolerability profile. Given that hypertension affects over 1.13 billion people globally and remains inadequately managed, there is huge clinical demand for effective and well-tolerated treatments like telmisartan. Rising lifestyle diseases, lack of exercise, obesity, and consumption of salt, sugar, and fat-rich diets have greatly increased the incidence of hypertension.
By Dosage Form - Tablets drive growth in dosage form segment
By dosage form, tablets segment is estimated to contribute the highest market share of 41.6% in 2026 due to various inherent advantages over other dosage forms. Tablets are the most convenient oral solid dosage form with benefits of stability, portability and easy administration. These do not require reconstitution like injections. Their manufacturing is also less complex and costly as compared to alternative dosage forms like capsules. Tablets allow accurate dosage, are easy to swallow as compared to large capsules, and provide consistent & rapid drug release for optimal therapeutic effect. This convenience leads to high patient preference and compliance for telmisartan tablets over other dosage forms. Furthermore, tablets are overwhelmingly prescribed by physicians worldwide as the standard oral dosage form in hypertension treatment.
Need a Different Region or Segment? Request Free Sample
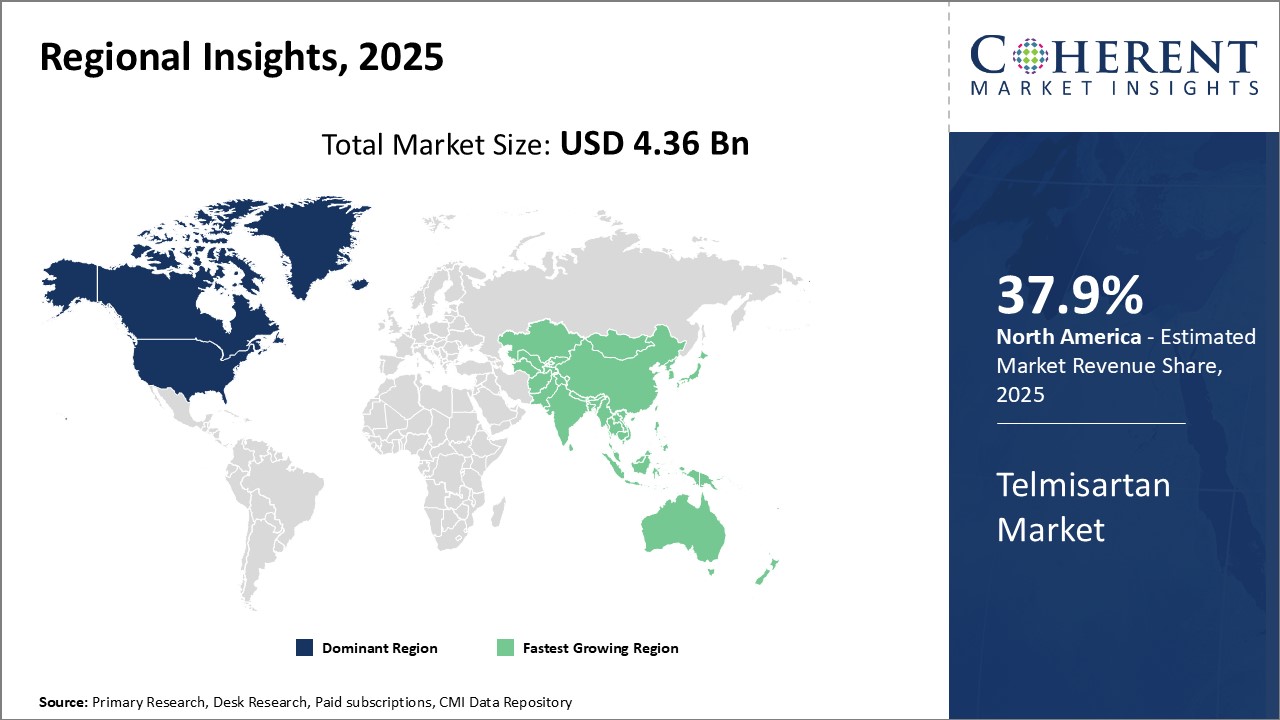
North America dominates the global telmisartan market with an estimated market share of 37.9% in 2026. The presence of leading pharmaceutical companies along with sophisticated healthcare infrastructure and growing prevalence of hypertension drives the market growth in North America. The region is a net exporter of Telmisartan APIs and formulations to other parts of the world. The strong distribution networks and global supply chain of American pharmaceutical companies plays a vital role in establishing North America's dominance in this market.
Asia Pacific has emerged as the fastest growing market for telmisartan globally. Countries like India, China and Japan have contributed majorly to the rapid market growth in this region. Favorable government policies to promote local manufacturing and cheaper cost of operations have attracted many global pharmaceutical companies to set up manufacturing plants in Asia Pacific region. Rising healthcare spending, growing medical needs of vast population and increasing focus on generics also drives the market growth. T Asia Pacific telmisartan market growth is primarily driven by rising domestic consumption as hypertension prevalence is surging rapidly, especially in middle-income countries like India and China. Asia Pacific has shown high potential in telmisartan market and anticipated to surpass other regions in the coming years, subject to sustaining its growth momentum.
Telmisartan Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 4.54 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 4.1% | 2033 Value Projection: | USD 6.01 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
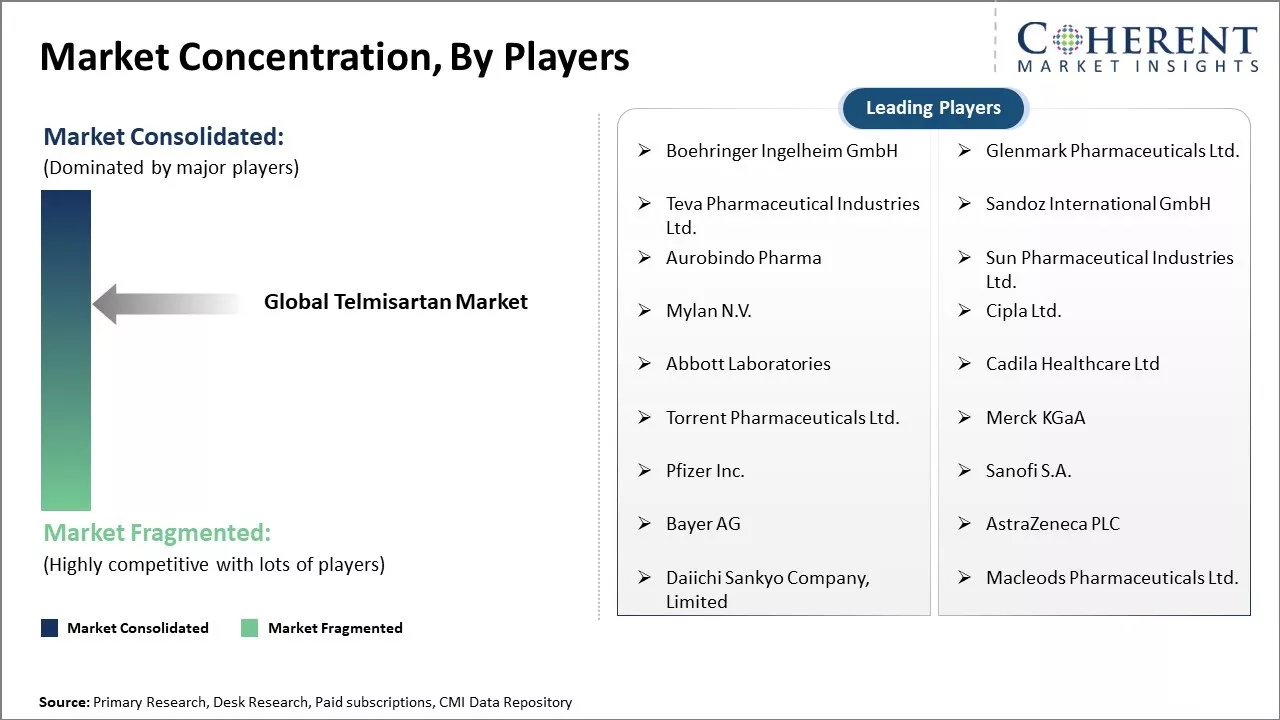
| Companies covered: |
Boehringer Ingelheim GmbH, Glenmark Pharmaceuticals Ltd., Teva Pharmaceutical Industries Ltd., Sandoz International GmbH, Aurobindo Pharma, Sun Pharmaceutical Industries Ltd., Mylan N.V., Cipla Ltd., Abbott Laboratories, Cadila Healthcare Ltd, Torrent Pharmaceuticals Ltd., Merck KGaA, Pfizer Inc., Sanofi S.A., Bayer AG, AstraZeneca PLC, Daiichi Sankyo Company, Limited, Macleods Pharmaceuticals Ltd. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
*Definition: The global Telmisartan market refers to the market for a specific pharmaceutical drug called Telmisartan. Telmisartan is primarily used to treat high blood pressure (hypertension) and reduce the risk of cardiovascular events such as heart attacks and strokes. It belongs to the class of medications known as angiotensin II receptor blockers (ARBs), which work by relaxing blood vessels, thereby lowering blood pressure.
Primary Research Interviews:
Databases:
Magazines:
Journals:
Newspapers:
Associations:
Public Domain Sources:
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients