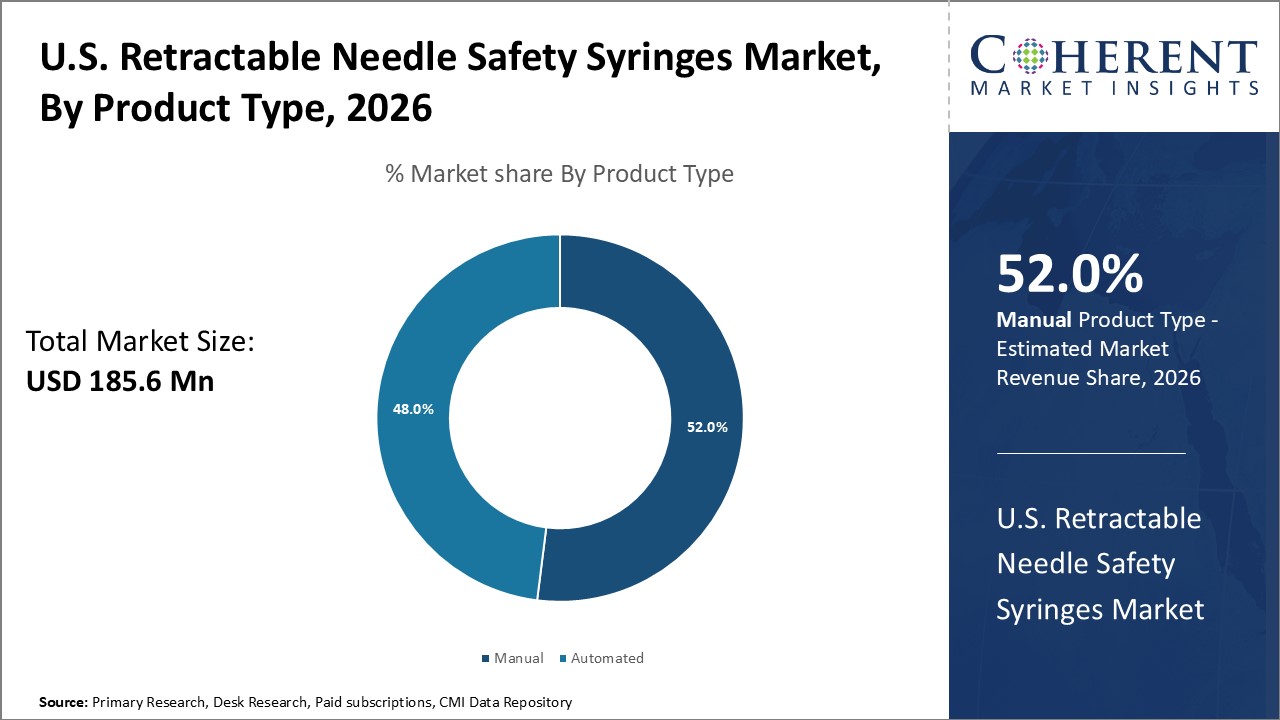
U.S. Retractable Needle Safety Syringes Market is estimated to be valued at USD 185.6 Mn in 2026 and is expected to reach USD 257.7 Mn in 2033, exhibiting a compound annual growth rate (CAGR) of 4.8% from 2026 to 2033.
Analysts’ Views on the U.S. Retractable Needle Safety Syringes Market:
Increasing research and development activities by key players are expected to drive the market growth over the forecast period. For instance, in July 2022, Sharps Technology, a U.S.-based medical device manufacturer, acquired Safeguard Medical’s smart syringe manufacturing facility for US$ 2.5 million. The acquisition aims to expand Sharps Technology’ manufacturing capabilities to address the increasing demand for smart syringes. Safeguard Medical is a U.S.-based manufacturer of smart syringes and emergency medical technologies.
Figure 1. U.S. Retractable Needle Safety Syringes Market Share (%), By Product Type, 2026
To learn more about this report, Request Free Sample
U.S. Retractable Needle Safety Syringes Market - Drivers
U.S. Retractable Needle Safety Syringes Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 185.6 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 4.8% | 2033 Value Projection: | USD 257.7 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Becton, Dickinson and Company, Smiths Medical, Revolutions Medical Corporation, Retractable Technologies Inc., SOL-MILLENNIUM, Axel Bio Corporation, Inc., DMC Medical Limited, Medicina, Duopross Meditech Corp., and MHC Medical Products, LLC. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
U.S.
faced problems with the transportation of things from one place to another.
retractable needle safety syringes market.Becton, Dickinson and Company, a medical technology company received orders for two billion injectable devices in syringes and needles. Moreover, the Biomedical Advanced Research and Development Authority (BARDA) estimated in January 2020 that 650 million to 850 million syringes and needles will be needed to administer vaccines in the U.S. alone.
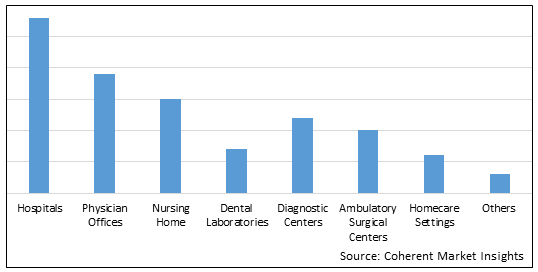
Figure 2. U.S. Retractable Needle Safety Syringes Market Share (%), By End User, 2026
To learn more about this report, Request Free Sample
U.S. Retractable Needle Safety Syringes Market Segmentation:
The U.S. retractable needle safety syringes market report is segmented based on product type, end user, and distribution channel.
U.S. Retractable Needle Safety Syringes Market: Key Developments
U.S. Retractable Needle Safety Syringes Market: Key Trends
One-inch needles are typically used for intravenous, intramuscular, and subcutaneous injections in medical settings. It is a long, somewhat thin needle. Injections into the femoral vein, which is somewhat close to the surface, may be beneficial. The needle stops any possible damage or scarring.
In 2021, hospitals generated a bigger percentage of revenue than clinics and ambulatory surgery centers. During the anticipated time frame, the sector is anticipated to own the majority of the market share. This can be explained by pointing to hospitals' higher foot traffic, patient admissions, and patient treatments when compared to other institutions.
U.S. Retractable Needle Safety Syringes Market: Restraint
The high cost of safety syringes: The high cost of safety syringes, growing cases of needle-stick injuries, and the availability of alternate drug delivery methods are major factors expected to hinder growth of the U.S. retractable needle safety syringes market. For instance, there are various alternative drug delivery methods such as oral, nasal, buccal, rectal, vaginal, sublingual, cutaneous, transdermal, ocular, otic, and others. Moreover, needle-stick injuries are known to occur frequently in healthcare settings and can be serious.
Key players are focusing on developing advanced retractable needle safety syringes that are safe and cost-effective, which is expected to drive market growth over the forecast period.
U.S. Retractable Needle Safety Syringes Market - Key Players
The major players operating in the U.S retractable needle safety syringes market include Becton, Dickinson and Company, Smiths Medical, Revolutions Medical Corporation, Retractable Technologies Inc., SOL-MILLENNIUM, Axel Bio Corporation, Inc., DMC Medical Limited, Medicina, Duopross Meditech Corp., and MHC Medical Products, LLC.
Primary Research Interviews:
Databases:
Magazines:
Journals:
Newspapers:
Associations:
Public Domain Sources:
U.S. Retractable Needle Safety Syringes Market - Definition: A retractable needle is one that is spring-loaded and retracts into the barrel of the syringe when the plunger is completely depressed after the injection is given. A syringe with a retractable needle works similar to a self re-sheathing needle. After the needle is fully depressed and all fluids are injected into the patient, a spring or gas cell enacts the needle and retracts it fully into the body of the barrel, where it is locked in place.
Share
Share
Manisha Vibhute is a consultant with over 5 years of experience in market research and consulting. With a strong understanding of market dynamics, Manisha assists clients in developing effective market access strategies. She helps medical device companies navigate pricing, reimbursement, and regulatory pathways to ensure successful product launches.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients