Viral Vector And Plasmid Dna Testing Services Market is estimated to be valued at USD 466.7 Mn in 2026 and is expected to reach USD 2,353.1 Mn in 2033, exhibiting a compound annual growth rate (CAGR) of 26.0% from 2026 to 2033.
Analysts’ Views on Global Viral Vector and Plasmid DNA Testing Services Market:
Increasing expansion of geographical presence by the market players by opening new manufacturing center is expected to foster the grot hog global viral vector and plasmid DNA testing services market over the forecast period. For instance, in July 2022, Charles River Laboratories International, a pharmaceutical company, announced the opening of its High Quality (HQ) Plasmid DNA manufacturing Centre of Excellence at Bruntwood SciTech's Alderley Park in Cheshire, U.K. This expansion will further help the company in strengthening their geographical footprints in the global market.
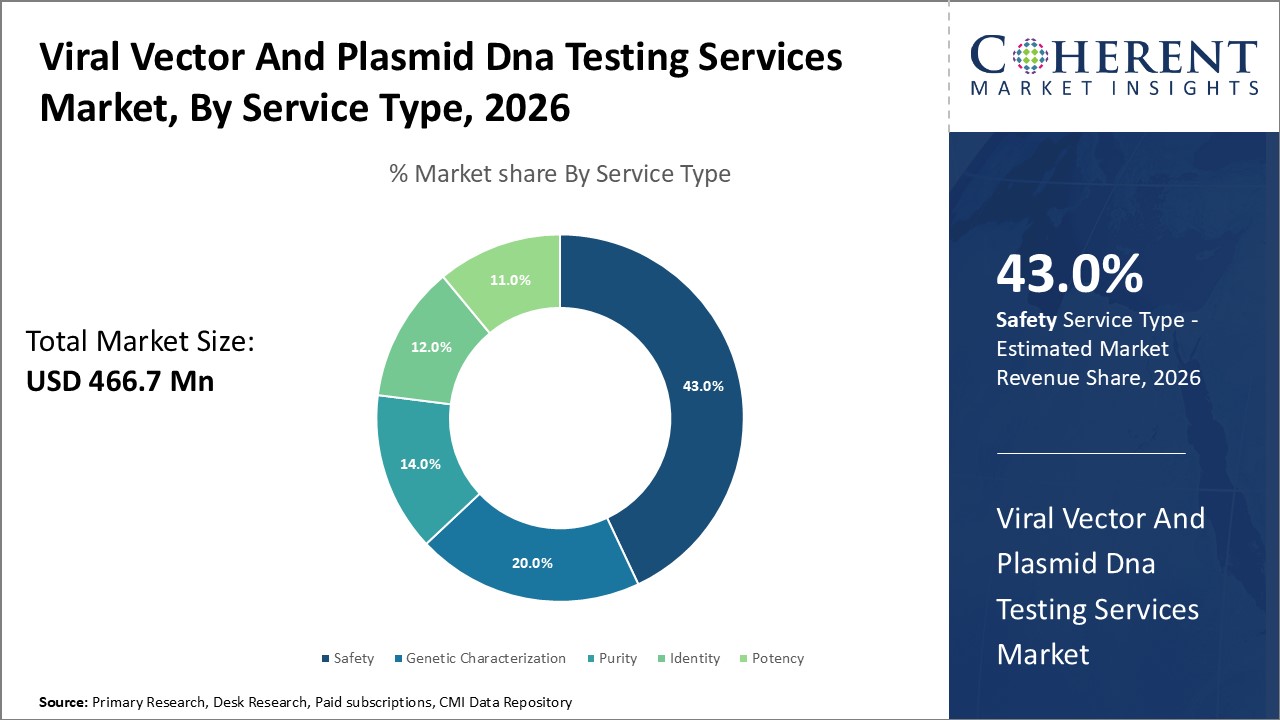
Figure 1. Global Viral Vector and Plasmid DNA Testing Services Market Share (%), By Service Type, 2026
To learn more about this report, Request Free Sample
Global Viral Vector and Plasmid DNA Testing Services Market– Drivers
Increasing number of inorganic growth strategies such as partnership and others
Increasing adoption of inorganic growth strategies such as collaboration, partnership and others by the key market players is expected to fuel the segmental growth of the over the forecast period. For instance, in October 2022, Charles River Laboratories International, Inc., a pharmaceutical company and Nanoscope Therapeutics, Inc., a clinical-stage biotechnology company developing gene therapies for retinal degenerative diseases, announced a comprehensive manufacturing collaboration utilizing Charles River's extensive contract development and manufacturing (CDMO) services in both plasmid DNA and viral vectors.
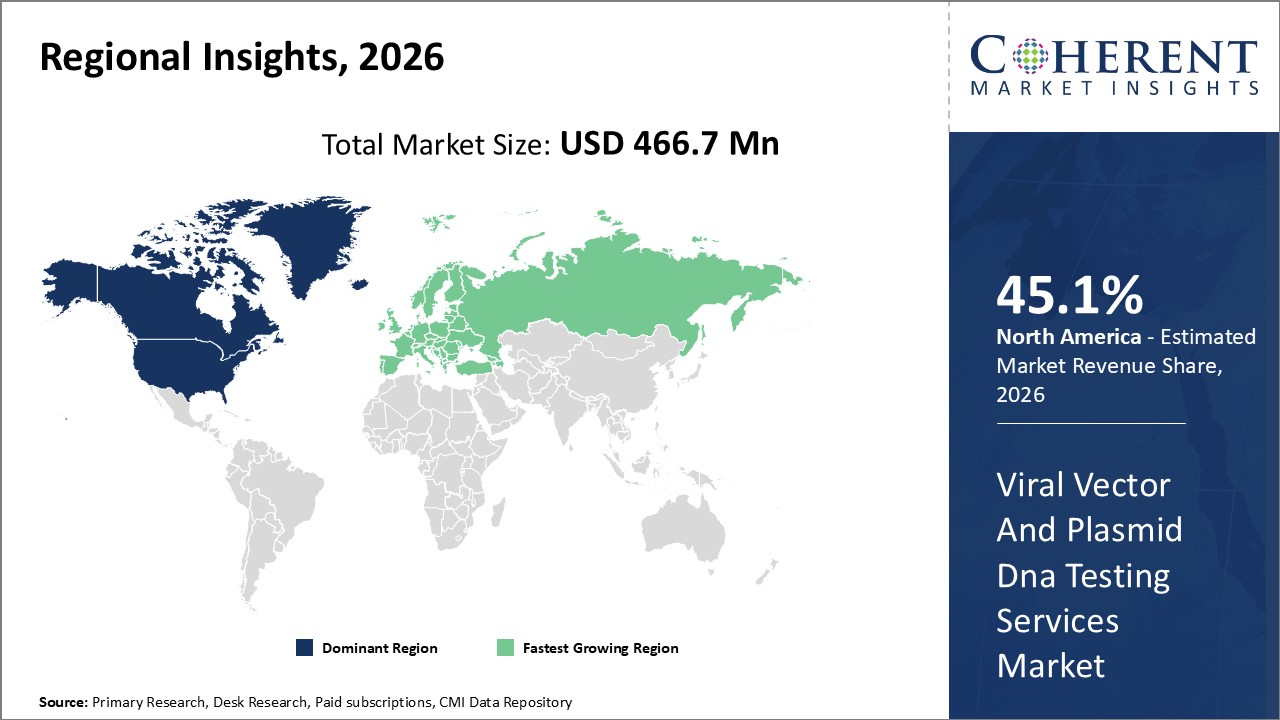
Figure 2. Global Viral Vector and Plasmid DNA Testing Services Market Value (US$ Million), By Region, 2026
To learn more about this report, Request Free Sample
Global Viral Vector and Plasmid DNA Testing Services Market- Regional Analysis
Among region, North America is estimated to hold a dominant position in the global viral vector and plasmid DNA testing services market over the forecast period, the presence of key players in the region who are focused on facility expansions to increase production and research & development activities. For instance, in October 10, 2026, VGXI Inc., a specialized contract developer and manufacturer (CDMO), announced an opening of its new headquarters and manufacturing facility located at Deison Technology Park in Conroe, Texas. U.S. for developing the next generation of gene therapies and vaccines
For instance, in April 2020, INOVIO pharmaceuticals Inc., a biotechnology company, entered into an agreement to expand its manufacturing partnership with the Germany-based contract manufacturer Richter-Helm BioLogics GmbH & Co. KG, to support large-scale manufacturing of INOVIO's investigational DNA vaccine INO-4800, which currently is in Phase 1 clinical testing in the U.S.
Viral Vector and Plasmid DNA Testing Services Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 466.7 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 26.0% | 2033 Value Projection: | USD 2,353.1 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Charles River Laboratories, Inc., WuXi AppTec Co., Ltd., Cobra Biologics and Pharmaceutical Services, Merck KgaA, Lonza, Eurofins Scientific, FinVector Vision Therapies, Advanced Bioscience Laboratories, Inc., Takara Bio Inc., ViruSure GmbH, Genezen Laboratories, Akron Biotech., Catalent, Inc, AcuraBio., CATUG Biotechnology., Creative Biogene and Aldevroz |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Viral Vector and Plasmid DNA Testing Services Market- Segmentation
Global viral vector and plasmid DNA testing services market report is segmented into by service type, by end user and by region.
Based on Service Type, the market is segmented into safety, genetic characterization, purity, identity and potency. Out of which, safety segment is expected to hold a dominant position in the global viral vector and plasmid DNA testing services market during the forecast period, owing to increasing number of inorganic growth strategies such as collaboration and others
Based on End User, the market is segmented into pharmaceutical and biotechnology industries and research organizations. Out of which, pharmaceutical and biotechnology industries segment is expected to dominate the market over the forecast period, owing to increasing research and development activities using viral vector and plasmid DNA for various therapeutic applications such as oncology and others
Based on Region, global viral vector and plasmid DNA testing services market is segmented into North America, Latin America, Europe, Middle East, Asia Pacific, and Africa. Out of which, North America is expected to dominate the market over the forecast period, due to increasing inorganic growth strategies such as agreement , partnership and others by key players in the region.
Among all segmentation, pharmaceutical and biotechnology industries segment has the highest potential key players are focusing on adoption of inorganic growth strategies such as partnerships, and collaborations, in order to strengthen their market presence in the global viral vector and plasmid DNA testing services market. Plasmid DNA (pDNA) starting material is an essential component of BioNTech’s mRNA-based vaccine manufacturing process.
Global Viral Vector and Plasmid DNA Testing Services Market- Cross Sectional Analysis
Among service type, safety segment held a dominant position in North America region over the forecast period due to increasing number of inorganic growth strategies such as collaboration and others by the market players for product development. Under this agreement, AGC Biologics will manufacture the plasmid DNA intermediate for the vaccine.
Global Viral Vector and Plasmid DNA Testing Services Market- Key Developments
In January 2022, Sarepta Therapeutics, Inc., a biotechnology company, and Aldevron, A corporate company, collaborated for the supply of GMP grade plasmid to meet the needs of Sarepta’s gene therapy clinical trials and commercial supply.
INOVIO plans to conduct the global INNOVATE Phase 3 segment in multiple countries, including Brazil, with partner Advaccine Biopharmaceuticals Suzhou Co., Ltd. (Advaccine).
In July 2021, Thermo Fisher Scientific, a pharmaceutical company, announced the opening of its new cGMP (Current good manufacturing practices) plasmid DNA manufacturing facility in Carlsbad California, U.S.
In June, 2020, Genprex, Inc., a clinical-stage gene therapy company, announced collaboration with Aldevron, a biotechnology company, in order to manufacture GMP plasmid DNA for Genprex's Oncoprex gene therapy portfolio.
Global Viral Vector and Plasmid DNA Testing Services Market- Key Trends
Increasing number of investment by market players for product development
Increasing number of investment by the market players in for product development is expected to foster the market growth over the forecast period. For instance, in September, 2022, Trans-Atlantic private equity healthcare specialist, ArchiMed, invested in Bielefeld, Germany-based PlasmidFactory. PlasmidFactory is the contract manufacturer and service provider for plasmid and minicircle DNA.CAR-Tcell applications).
Partnership by key market players
Key players are focused on partnerships to strengthen their position, is expected to fuel the global viral vector and plasmid DNA testing services market over the forecast period. For instance, on October 17, 2022, GenScript ProBio, a global CDMO, and GeneCraft, a global research of lung cancer mechanism research, announced that they had entered into a strategic partnership MOU concerning the development and production of a new drugs needed for RX001. GenScript ProBio and GeneCraft have agreed to strengthen their cooperation in the AAV gene therapy field through this MoU. GenScript ProBio and GeneCraft are in the process of signing a contract for plasmid and AAV development and production for the GeneCraft's own new drug candidate Pan-KRAS non-small cell lung cancer anti-cancer gene therapy (RX001) developed as a new drug.
Global Viral Vector and Plasmid DNA Testing Services Market: Restraints
Requirement of highly Skilled Labor
Production of recombinant plasmid DNA requires advanced manufacturing equipment and facilities and highly experienced and skilled labor. Limited number of Contract Development and Manufacturing Organizations (CDMOs) and biopharmaceutical companies have enough resources at all stages of the product development cycle. Majority of CDMOs providing commercial and clinical manufacturing and development services and academic laboratories providing early phase support, are running at high capacity, typically with longer wait list.
Challenges Faced During DNA Supply
Biotech and pharmaceutical companies that are majorly focused on advanced therapy manufacturing face challenges associated with their plasmid DNA supply, which includes slow or delayed production and poor DNA quality, and impacting their ability to meet timelines and regulatory demands. However, the regenerative medicine industry is also experiencing challenges and limitations with genetic medicines. Almost, every genetic medicines today are dependent on plasmid DNA as either critical starting material or API (Application Programming Interface), which presents challenges in advanced therapy manufacturing.
Global Viral Vector and Plasmid DNA Testing Services Market- Key Players
Major players operating in the global viral vector and plasmid DNA testing services market include Charles River Laboratories, Inc., WuXi AppTec Co., Ltd., Cobra Biologics and Pharmaceutical Services, Merck KgaA, Lonza, Eurofins Scientific, FinVector Vision Therapies, Advanced Bioscience Laboratories, Inc., Takara Bio Inc., ViruSure GmbH, Genezen Laboratories, Akron Biotech., Catalent, Inc, AcuraBio., CATUG Biotechnology., Creative Biogene and Aldevroz
Share
Share
Abhijeet Kale is a results-driven management consultant with five years of specialized experience in the biotech and clinical diagnostics sectors. With a strong background in scientific research and business strategy, Abhijeet helps organizations identify potential revenue pockets, and in turn helping clients with market entry strategies. He assists clients in developing robust strategies for navigating FDA and EMA requirements.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients