The global abortion drugs market is expected to grow from USD 2,100.1 Mn in 2026 to USD 3,367.7 Mn by 2033, registering a compound annual growth rate (CAGR) of 7%. The global abortion drugs market is poised for significant expansion, fueled by the frequent and persistent occurrence of unintended pregnancies.
According to the United Nations Population Fund, about 121 million pregnancies each year are unplanned, and more than 60% of these end in abortion. Additionally, about 45% of all abortions are unsafe, which causes 5–13% of all maternal deaths worldwide.
(Source: The United Nations Population Fund)
Mifepristone is projected to hold a market share of 50.4% in 2026, attributed mainly to the drug's high efficacy, approval from relevant regulatory bodies, and standardization in the abortion process. When used with misoprostol, mifepristone-based therapy has success rates of over 95%–99%, which is much higher than other methods (Source: JAMA Network). The drug works by blocking progesterone, a hormone that is necessary for pregnancy to continue, so it can produce effective and predictable results, especially in the first 10 weeks of pregnancy. (Source: U.S. Food and Drug Administration)
For example, regulatory authorities like the U.S. Food and Drug Administration have approved mifepristone as part of a standardized medication abortion regimen, reinforcing its widespread clinical adoption. Its superiority in efficacy, safety profile (characterized by minimal complication rates), and integration into global treatment protocols collectively contribute to its preeminence in the abortion drugs market.
To learn more about this report, Request Free Sample
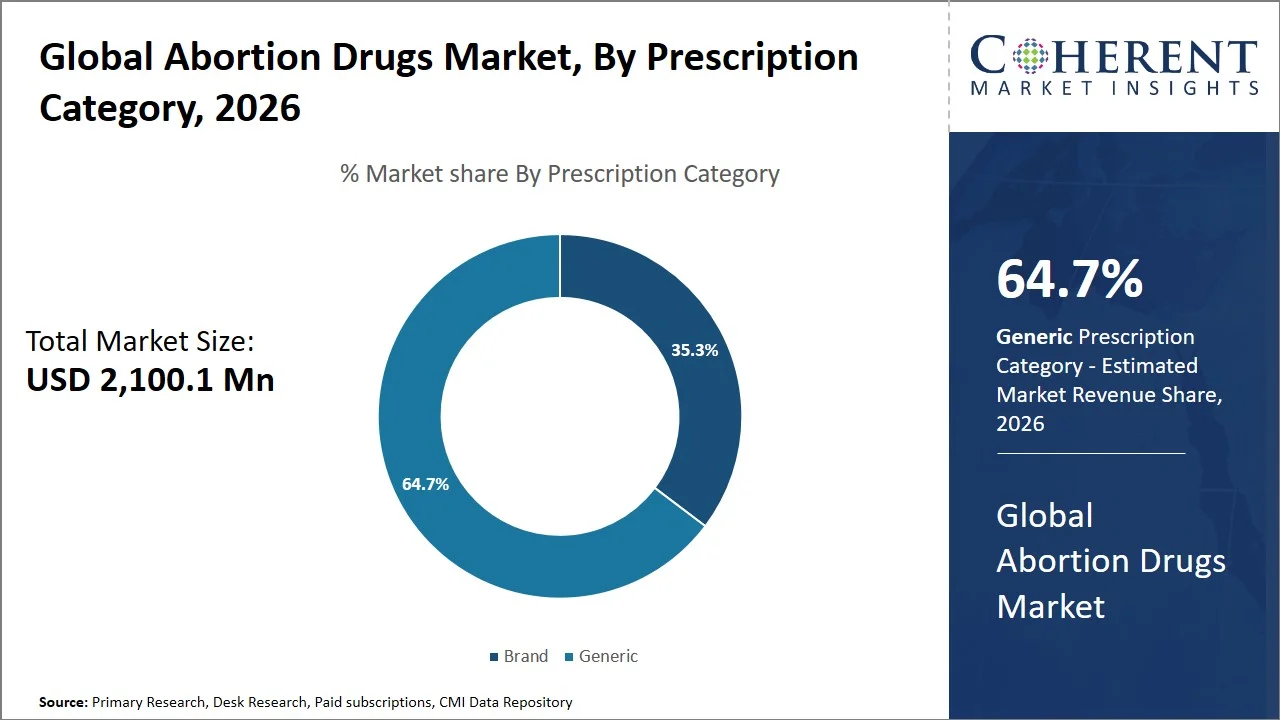
Generic is projected to hold a market share of 64.7% in 2026, owing to their cost effectiveness, equal efficacy, and robust governmental policies in favor of generics (such as the Pradhan Mantri Bhartiya Janaushadhi Pariyojana in India, the Volume-Based Procurement Program in China, and the Generic Drug User Fee Amendments in the U.S.). The drugs are composed of the same active chemicals as their branded versions but have to comply with stringent standards of safety, quality, and performance, as stipulated by health organizations, including the U.S. FDA (Source: U.S. Food and Drug Administration). They can be procured at a significantly lower price than their branded equivalents and therefore are considered more affordable by both developing countries and government-run health systems that need cost-effectiveness. The governments themselves work towards promoting generics to keep health costs low and improve access to medicines. Consequently, there exist huge manufacturing centers (such as Hyderabad and Ahmedabad in India, and Shanghai and Zhejiang in China) for generic drugs in developing countries such as India and China.
The up to 7 weeks segment is projected to hold a market share of 56.7% in 2026, attributed to greater efficiency and safety associated with the use of drug-based procedures for abortions at early pregnancy stages. Drug-based methods have demonstrated more favorable results, making them the best choice in the eyes of professionals. For instance, a study published in February 2025, by the National Library of Medicine reported that medical abortion using mifepristone–misoprostol achieved a success rate of approximately 97–98% for pregnancies up to 7 weeks, reinforcing its high efficacy in early gestation (Source: National Library of Medicine). The early identification of pregnancy and greater awareness on matters related to women’s health contribute to the success of such a procedure. Countries including the U.K. provide guidance based on clinical policies in line with the National Health Service, encouraging early medical abortion.
|
Current Events |
Description and its Impact |
|
Rising Regulatory Scrutiny & Legal Battles Around Mifepristone (2025–2026) |
|
|
Rapid Shift Toward Telehealth & Mail-Order Abortion Drug Distribution |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
(Source: Reuters, Guardian News & Media Limited)
To learn more about this report, Request Free Sample
To learn more about this report, Request Free Sample
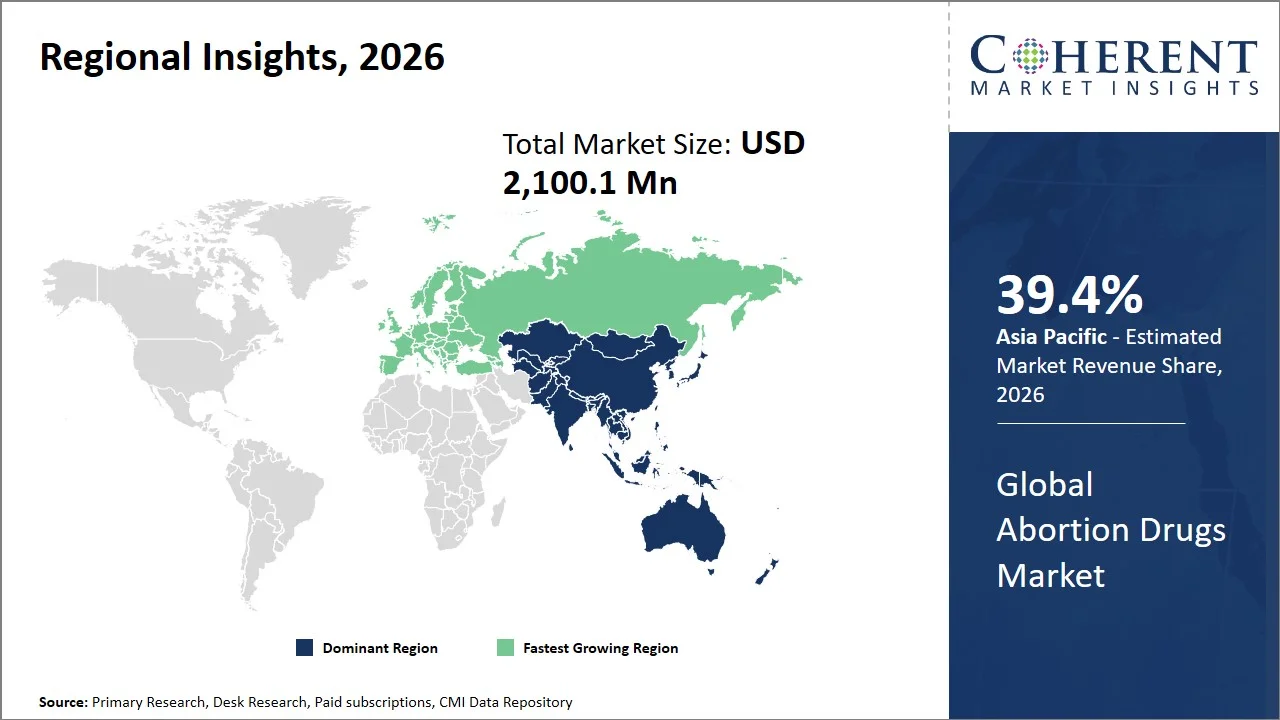
Asia Pacific leads the global abortion drugs market, accounting for an estimated 39.4% share in 2026, owing to the large reproductive-age population, the prevalence of unwanted pregnancies (Asia Pacific accounts for nearly 55–60% of global unintended pregnancies) along with the presence of advanced drug production processes.
India and China are significant in their roles of both consumers and producers because of their advanced pharmaceutical facilities as well as their ease of obtaining medicines. For instance, in India, the law named "The Medical Termination of Pregnancy Act" allows medical abortion by qualified doctors and clinics; therefore, it ensures general popularity of the procedure (Source: Indian Penal Code). Similarly, in China, the provision of abortions is made in its healthcare system under the oversight of the National Health Commission of China, ensuring the widespread availability of medical abortion procedures across hospitals and approved medical institutions. Due to the better education in reproductive issues and popularity of non-surgical abortion, the Asia Pacific region ranks among the most promising market of abortion drugs.
The Europe abortion drugs market is expected to exhibit the fastest growth with an estimated contribution of 24.8% share to the market in 2026, progressive regulatory frameworks (e.g., U.K. telemedicine approval for early medical abortion and France’s authorization for home use of abortion medications up to extended gestational limits), increased availability of telemedicine services, and effective public health care systems. In countries like the U.K. and France, remote consultations as well as the use of abortion medications by individuals at their homes, has been legalized under progressive regulatory frameworks. Additionally, the presence of public health care systems, like the National Health Service, helps perform abortions in an early stage and with the help of abortion drugs. For instance, in England and Wales, taking both abortion medications at home has become the most common method, accounting for 72% of all abortions (around 200,745 cases) in 2023, highlighting the strong adoption of telemedicine-enabled and home-based medical abortion services. (Source: Department of Health and Social Care)
India is one of the key markets for abortion drugs because it has a large population of people of reproductive age coupled with the need for affordable options in reproductive medicine. The well-developed regulatory framework in India, which includes the Medical Termination of Pregnancy Act providing legal permission for using abortion drugs by qualified specialists. Due to the existence of significant local manufacturing in India’s pharmaceutical industry (such as Sun Pharmaceutical Industries Ltd. and Cipla Limited), it becomes possible to get inexpensive generic versions of abortion medications.
Moreover, awareness of family planning techniques combined with government backing in reproductive health campaigns stimulates the application of abortion drugs in India. For example, the government of India has launched various schemes like the National Family Planning Program and Mission Parivar Vikas that focus on making people aware of contraception and birth-spacing methods as well as the safe practice of reproductive health, especially in areas where the fertility rate is higher.
Japan is a developing yet controlled market for abortion drugs. The country has recently approved abortion drugs (Mefeego Pack) highlighting a significant step towards the adoption of medication-based abortion and modernization of reproductive healthcare practices. Nonetheless, there are certain regulations like physician assistance that must be adhered to when purchasing the medicine. The existence of a sophisticated healthcare system and the general knowledge of people about reproductive health adds to the slow progress of adopting such medications. The demographics of Japan, especially its aging population and low birth rate, also affect policies and demand for reproductive health services.
The abortion drugs market is growing rapidly in China due to the high volume of procedures and availability of medical abortion facilities. Abortion is legalized in China and is performed in the healthcare sector managed by the National Health Commission of China. High manufacturing capacities in China will enable companies to produce more abortion drugs at lower costs. Increasing knowledge regarding reproductive health and family planning in China have resulted in a constant need for abortion drugs. For instance, China records a very high number of abortion procedures annually (over 9 million cases reported annually, with estimates reaching up to ~13 million), reflecting widespread accessibility and utilization of abortion services across the country. (Source: National Library of Medicine)
The U.K. is at the forefront of European countries when it comes to the adoption and usage of abortion drugs because of its advanced and progressive regulations (such as, temporary approval of at-home use of both mifepristone and misoprostol, and permanent legalization of telemedicine-based early medical abortion services), and prevalence of abortions using medications (over 85–90% of abortions in England and Wales are conducted using medication-based methods). The country has made it possible for telemedicine services to be used alongside taking these abortion medications at home, thus making the process easier for the patients. The support provided by the public health sector via the National Health Service makes sure that there are consistent and accessible services available for all citizens in terms of reproductive health.
The emergence of France as a promising market for abortion drugs is driven by the government’s gradual policy advancements aimed at expanding access to abortion services. Legal abortions can be conducted until 14 weeks of pregnancy, and state support in providing healthcare services are actively practiced. Telemedicine and self-medicating are becoming increasingly common, allowing conducting an abortion at home using remote consultations with doctors and buying medicines from pharmacies. For example, the France government authorized teleconsultations for medical abortion and permitted home use of abortion pills up to extended gestational limits, improving accessibility and convenience across the country.
|
Region |
Regulatory Environment |
Key Characteristics |
|
North America |
Highly regulated but structured |
In the U.S., abortion drugs such as mifepristone are regulated under strict programs by the U.S. Food and Drug Administration (REMS), with certified providers and pharmacies required. Access varies by state laws despite federal approval. |
|
Europe |
Progressive but country-specific |
Countries like the U.K. and France allow medical abortion with telemedicine and home-use protocols, supported by public healthcare systems. However, regulations differ across Eastern and Southern Europe. |
|
Asia Pacific |
Highly variable and evolving |
Markets such as India permit medical abortion under defined legal frameworks (e.g., MTP Act), while China allows regulated access through hospitals. Some countries maintain restrictive policies, limiting availability. |
|
Latin America |
Restricted but gradually liberalizing |
Countries like Argentina have legalized abortion and expanded access to medication abortion, while others still impose strict legal restrictions, impacting market penetration. |
|
Middle East & Africa |
Highly restrictive |
Most countries maintain strict regulations with limited legal access, allowing abortion only under specific medical conditions, significantly constraining the availability of abortion drugs. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The emerging trend of telemedicine and e-pharmacy distribution systems are providing numerous growth opportunities for the abortion drugs market, as such trends provide better accessibility, privacy, and convenience for patients who live in rural and restricted locations. The presence of distribution channels means that the patient is able to consult their doctor, who gives them a prescription remotely and then sends them their abortion pills without having to go to a clinic. For instance, the U.S. FDA has modified its Risk Evaluation and Mitigation Strategy (REMS) program for mifepristone so that it would be possible to send out the pills using mail services from certified pharmacies (Source: U.S. Food and Drug Administration). Concurrently, nations like the U.K. have embraced telemedicine abortion via the National Health Service (NHS), allowing patients to undergo the procedure from the comfort of their homes. Such developments in regulations and services have considerably enhanced accessibility, facilitating the widespread acceptance of medication abortion across the globe.

To learn more about this report, Request Free Sample
The global abortion drugs market exhibits moderate concentration yet geographical fragmentation with pharmaceutical companies, generic drugs producers, and public health organizations being key participants. The factors influencing competitive pressure within these industries are the need for compliance, competition based on prices, and access, considering that organizations must operate under diverse regulations in various geographical locations. In developed countries, competitive pressure comes from strict rules and few ways to get products to customers. In developing countries, competition is tough because there are so many generic drug makers in the market. Key focus areas include:
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 2,100.1 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7% | 2033 Value Projection: | USD 3,367.7 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Danco Laboratories, LLC, GenBioPro, Inc., Exelgyn S.A., Evita Solutions, Pfizer Inc., Teva Pharmaceuticals, Sun Pharmaceutical Industries Ltd., Cipla Limited, Lupin Limited, and Zydus Lifesciences Ltd. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients