The global actinic keratosis market is estimated to be valued at USD 9.67 Bn in 2026 and is expected to reach USD 13.88 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 5.3% from 2026 to 2033.
Discover market dynamics shaping the industry: Request Free Sample
The growth in elderly population suffering from sun damage combined with increasing awareness regarding skin diseases is expected to drive the demand for actinic keratosis treatment. Additionally, technological advancements in treatment modalities and increasing availability of novel therapeutics are further expected to provide opportunities for the growth of actinic keratosis market players. However, lengthy product approval timelines and high costs associated with new drug development may impede market growth to some extent during the forecast period.
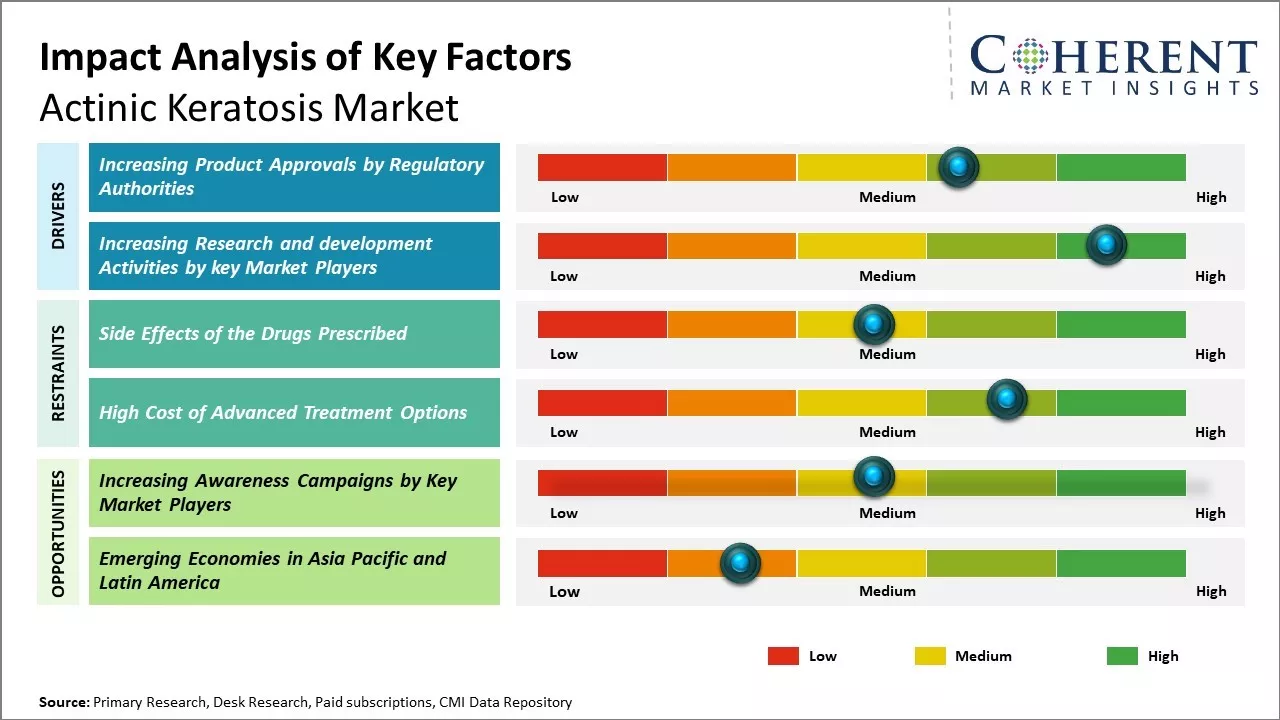
Increasing Product Approvals by Regulatory Authorities
Increasing number of product approvals for the treatment of actinic keratosis from regulatory bodies such as European Commission is expected to drive the growth of the global actinic keratosis market over the forecast period. For instance, in March 2020, Biofrontera AG, a Germany-based pharmaceutical company, announced that it had received approval from the European Commission for the use of its drug, Ameluz, in combination with photodynamic therapy for the treatment of adults suffering from actinic keratosis on the extremities and trunk/neck in Europe.

Get actionable strategies to beat competition: Request Free Sample
Increasing Research and Development Activities by Key Market PlayersIncreasing research and development activities by key market players are expected to drive the global actinic keratosis market during the forecast period. For instance, in January 2023, Bioforntera AG, a biopharmaceutical company, announced that it had initiated Phase 3 clinical trial to evaluate the safety and efficacy of Ameluz in a field-directed treatment of actinic keratosis (AK) on the extremities, neck, and trunk.

To learn more about this report, Request Free Sample
Market Challenges – Side Effects of the Drugs PrescribedGlobal actinic keratosis market growth is expected to be hampered owing to the side effects of the drugs prescribed for actinic keratosis. Some of the common side effects of actinic keratosis drugs are burning or crusting, redness or discoloration, itching and rash, soreness, etc. some of the serious side effects of actinic keratosis drugs are vomiting, severe stomach ache, skin red rashes, etc.
Market Opportunities – Increasing Awareness Campaigns by Key Market Players
Increasing adoption of inorganic growth strategies, such as launch of awareness campaigns, is expected to offer lucrative market growth opportunities. For instance, in June 2022, GALDERMA, a dermatology company, announced that it had launched Face the Facts Campaign to raise awareness about actinic keratosis (AK).
Discover high revenue pocket segments and roadmap to it: Request Free Sample
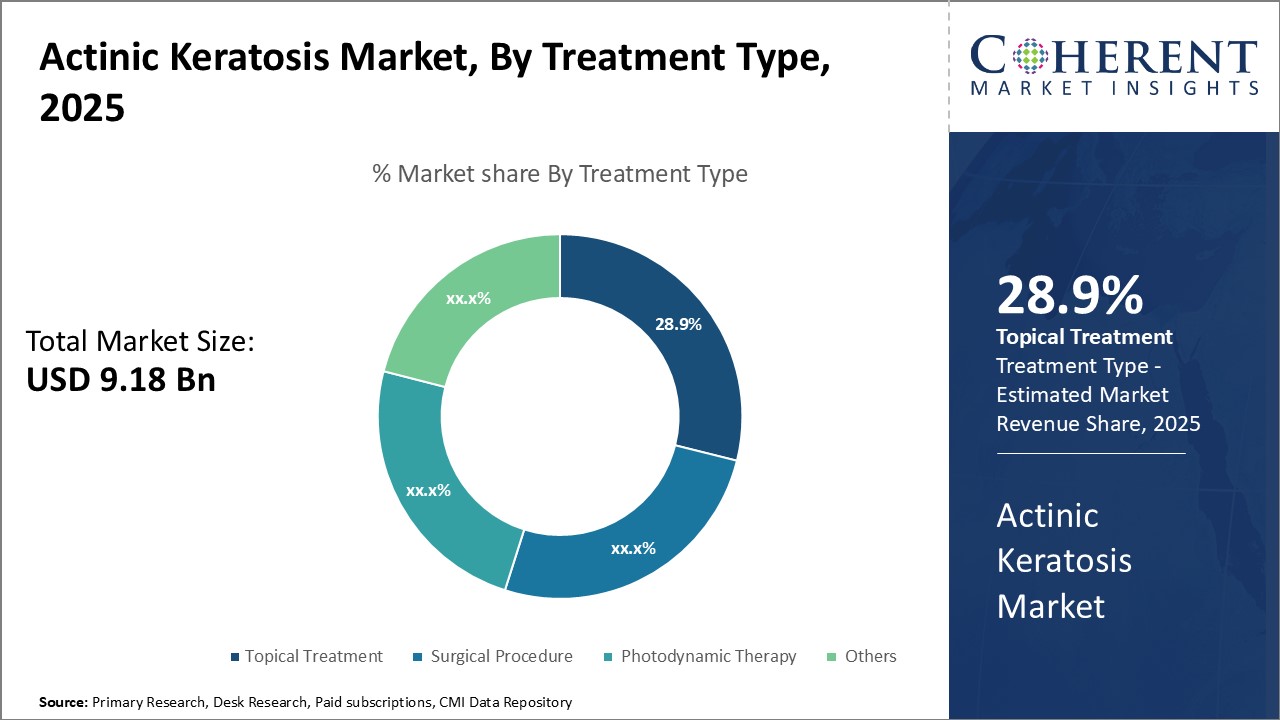
Insights, By Treatment Type: Convenience and Minimal Side EffectsThe treatment type segment includes topical treatment, surgical procedure, photodynamic therapy, and others. The topical treatment sub-segment is estimated to hold 29.9% of the market share in 2026 owing to various advantages. Being a non-invasive option, it is more convenient for patients to apply creams or gels on the affected area as opposed to undergoing certain procedures. This convenience factor has made topical treatment popular especially in the early stages of the disease. Another major driver is the minimal side effects associated with topical treatment. Creams usually have very mild and localized side effects if any. This allows for a safer treatment approach. Certain options are also over-the-counter medicines which increases accessibility. As topical agents spare patients from pain and discomfort of other alternatives, compliance is improved. This has resulted in many topical formulations becoming first line therapies. With ongoing research yielding stronger but gentler topical formulations, popularity of this segment continues to increase. Features like once-daily application further enhance user-friendliness. As dermatologists also consider it appropriate for majority of actinic keratosis cases in early stages, topical treatment maintains its leading position in the global actinic keratosis market.
To learn more about this report, Request Free Sample
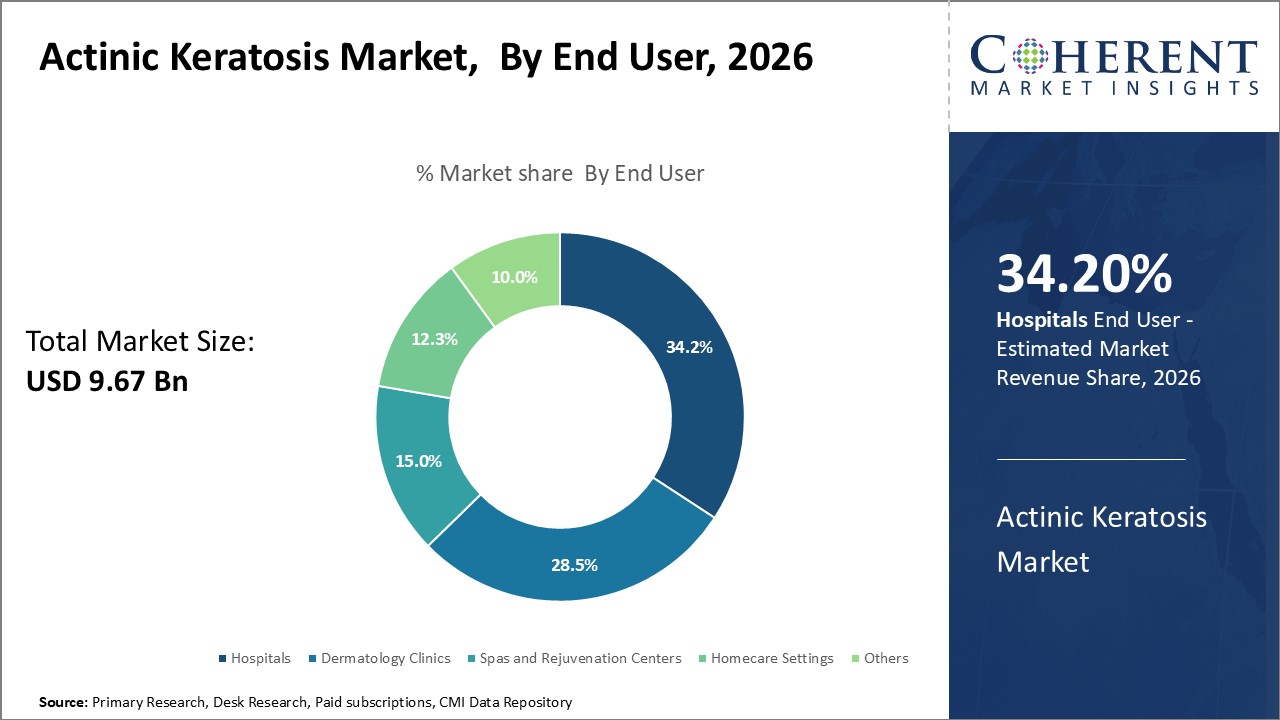
Insights, By End User: Availability of Advanced Diagnostic and Treatment Facilities
The end user segment includes hospitals, dermatology clinics, spas and rejuvenation centers, homecare settings, and others. The hospitals sub-segment is estimated to hold 34.2% of the market share in 2026. This is because hospitals are equipped with better diagnostic and treatment infrastructure to comprehensively manage the condition. Advanced facilities like biopsy, photodynamic therapy machines, etc. allow accuracy in diagnosis and effective treatment management. An integrated team of dermatologists, surgeons, and supporting staff is conducive for efficient care. Hospitals also provide favorable reimbursement for therapeutics as well as procedures carried out. This ensures access to care is not hindered by costs. Referrals from primary physicians also contribute to high patient volume in hospitals.
Need a Different Region or Segment? Request Free Sample
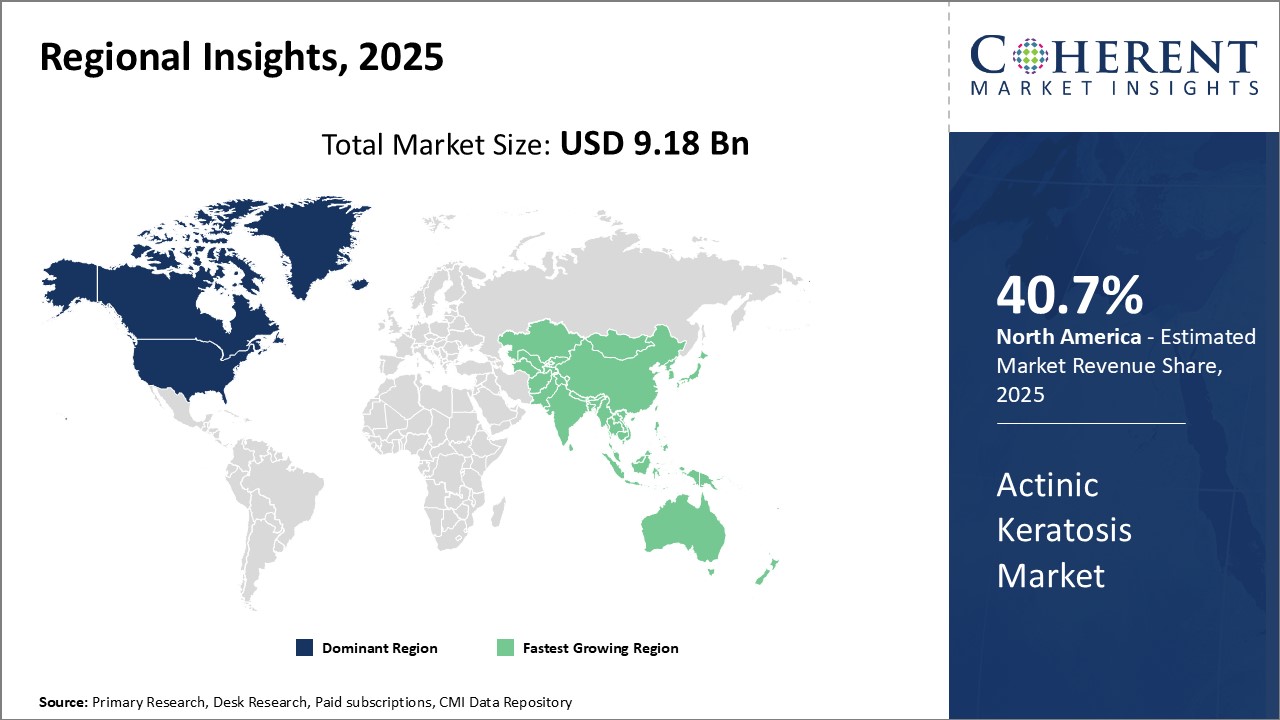
North America remains the dominant region in the global actinic keratosis market and is estimated to hold 40.7% of the market share in 2026 owing to high disease prevalence rates in the region. The U.S. accounts for the largest share primarily due to growing geriatric population that is more prone to the disease. Additionally, increased awareness regarding early diagnosis and available treatment options along with favorable reimbursement policies in the country have supported the market growth over the years. The strong presence of leading pharmaceutical companies in the region engaged in developing novel therapeutic alternatives against actinic keratosis is another major factor contributing to North America's large market size.
Asia Pacific has emerged as the fastest growing regional market for actinic keratosis treatment. Countries such as China, India, and Japan are witnessing rapid growth supported by rising health expenditures, surge in medical tourism, and improving access to healthcare facilities. The rising consumer disposable incomes have also increased willingness to spend on aesthetic procedures. This has positively impacted the demand for therapeutics against actinic keratosis as people are more concerned about early signs of skin damage and aging. In addition, the growing prevalence of the disease due to increasing exposure to UV radiation from sunlight on account of hot and humid climates in Asia Pacific provides immense growth opportunities.
Actinic Keratosis Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 9.67 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 5.3% | 2033 Value Projection: | USD 13.88 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Almirall, S.A, Bausch Health Companies Inc., Perrigo Company plc., Sun Pharmaceutical Industries Ltd., Hill Dermaceuticals, Inc., Tolmar Pharmaceuticals, Inc., Bioforntera AG, Cipher Pharmaceuticals Inc., Vidac, Athenex, Inc., GALDERMA, and LEO Pharma A/S |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
*Definition: Actinic keratosis is a damage caused to skin due to excessive sun exposure. Actinic keratosis is a very common precursor to skin cancer. Actinic keratosis is considered a chronic disorder, where the majority of patients experience recurrent lesions over time. Actinic keratosis increases the risk of developing skin cancer, as it may develop into squamous cell carcinoma (SCC), a common and sometimes invasive skin cancer. Actinic keratosis is an early precursor to squamous cell carcinoma, a malignant skin tumor that develops from the keratinizing cells (keratinocytes) in the epidermis or skin adnexa.
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients