the glucagon like peptide 1 analogs market is estimated to be valued at USD 59.01 Bn in 2026 and is expected to reach USD 117.21 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 10.3% from 2026 to 2033. This robust growth reflects increasing prevalence of diabetes and obesity worldwide, driving the demand for innovative therapeutic options. Continued advancements in peptide therapeutics and expanding patient awareness contribute significantly to the market expansion over the forecast period.
Market trends indicate a strong shift towards personalized medicine, with a focus on once-weekly and oral glucagon like peptide 1 analogs formulations enhancing patient compliance and convenience. Additionally, the rising integration of digital health tools and continuous glucose monitoring systems fosters improved treatment management. Collaborations between pharmaceutical companies and technology providers are shaping the landscape, while emerging markets present lucrative opportunities due to growing healthcare infrastructure and rising incidence of metabolic disorders, fueling sustained demand for glucagon like peptide 1 analogs therapies.
|
Current Events |
Description and its impact |
|
Patent Expirations and Generic Market Entry |
|
|
Emergence of Digital Therapeutics
|
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Semaglutide is expected to emerge as the dominant drug type in the glucagon like peptide 1 analogs market with a share of 41.4% in 2026 primarily due to its remarkable efficacy in glycemic control and weight reduction. Its ability to significantly lower HbA1c levels while promoting sustained weight loss positions it as a preferred treatment for patients with type 2 diabetes mellitus (T2DM) and obesity. Semaglutide’s once-weekly injectable formulation, coupled with the availability of an oral version, enhances patient compliance and convenience, factors that are crucial in chronic disease management.
Another key driver of Semaglutide’s dominance is its favorable safety profile, which has been validated across numerous large-scale clinical trials. The drug has demonstrated not only effective glucose regulation but also cardiovascular benefits, making it attractive to healthcare providers who seek comprehensive risk reduction in their diabetic patients. This dual-action characteristic taps into the widening need for multifunctional treatment options in a population with complex comorbidities.
To learn more about this report, Request Free Sample
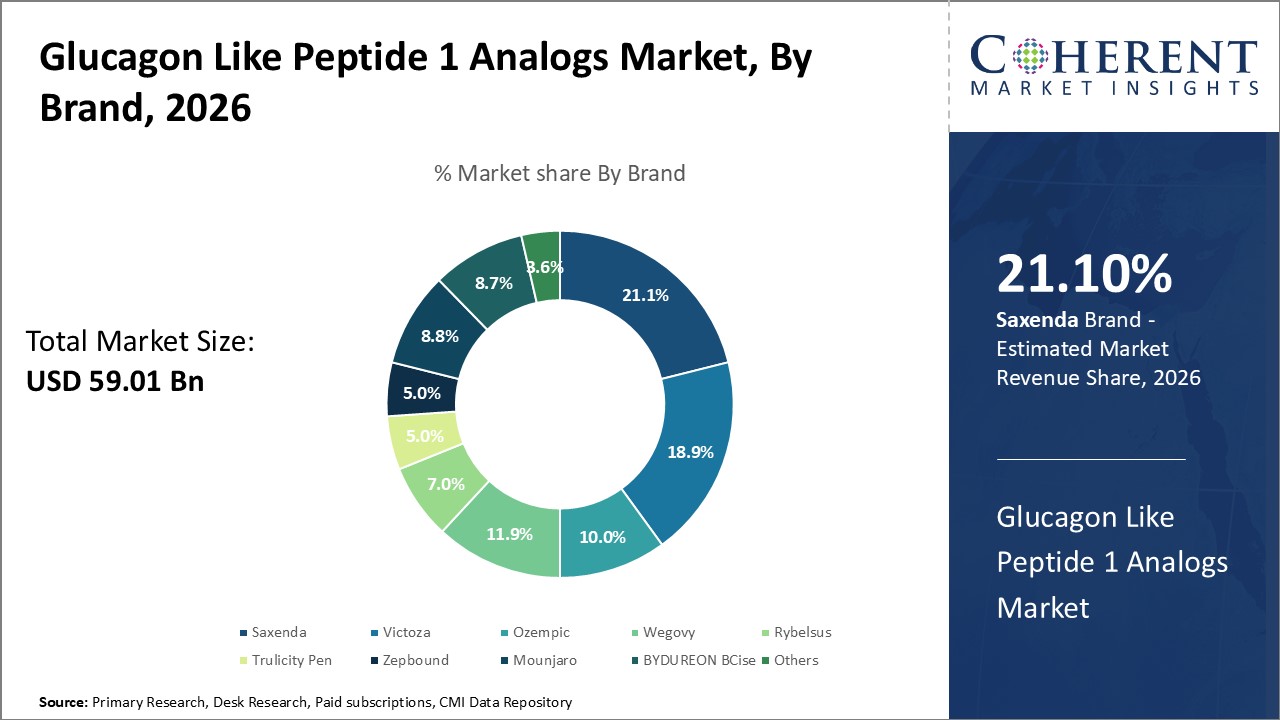
Saxenda is expected to lead the glucagon like peptide 1 analogs market by brand largely because of its established presence as an effective weight management therapy for obesity and overweight adults with share of 21.1% in 2026. Its approval specifically for chronic weight management distinguishes it from many other GLP-1 based medications primarily indicated for diabetes, thereby carving a niche segment focused on the growing global obesity epidemic.
A major growth factor for Saxenda is the increasing awareness and emphasis on obesity as a medical condition requiring pharmacological intervention alongside lifestyle modifications. Healthcare systems and patients are becoming more proactive in seeking treatments that facilitate sustainable weight loss and help reduce obesity-related complications, giving Saxenda a strong advantage as a recognized solution in this domain.
In May 2024, Biocon Limited, one of the global biopharmaceutical companies, entered into a semi-exclusive distribution and supply agreement with Medix, a specialty pharmaceutical company in Mexico, for the commercialization of generic Saxenda (Liraglutide) for chronic weight management. Biocon will handle regulatory approval, manufacturing, and supply, while Medix will manage commercialization in Mexico.
The Type 2 Diabetes Mellitus segment remains the largest application area for glucagon like peptide 1 analogs, driven primarily by the global surge in diabetes prevalence due to lifestyle changes, urbanization, and aging populations with an estimated share of 53.1% in 2026. GLP-1 receptor agonists have become critical in T2DM therapy by addressing key pathophysiological defects including impaired insulin secretion and excess glucagon production, aligning pharmacology directly with disease mechanisms.
This growth is driven by the increasing prevalence of type 2 diabetes and the broad availability of GLP-1 receptor agonists for its treatment. For instance, in November 2024, according to the World Health Organization November 2024 (WHO), over 95% of people globally are affected by diabetes, with type 2 diabetes—once considered an adult-onset, non-insulin-dependent condition—becoming more common among children due to changing eating habits. Currently, several GLP-1 receptor agonists, including Dulaglutide (Trulicity), Exenatide (Byetta), and Liraglutide (Victoza), are used to treat type 2 diabetes. The rising incidence of the disease is expected to further drive demand for glucagon like peptide drugs.
The regulatory landscape for glucagon like peptide 1 analogs, such as Exenatide, Liraglutide, Dulaglutide, and Semaglutide, is closely monitored by health authorities globally to ensure their safety and efficacy for treating Type 2 diabetes.
In all regions, the approval process involves preclinical research, clinical trials, and post-market surveillance to ensure the drugs' long-term safety and effectiveness, addressing the growing global diabetes burden.
To learn more about this report, Request Free Sample
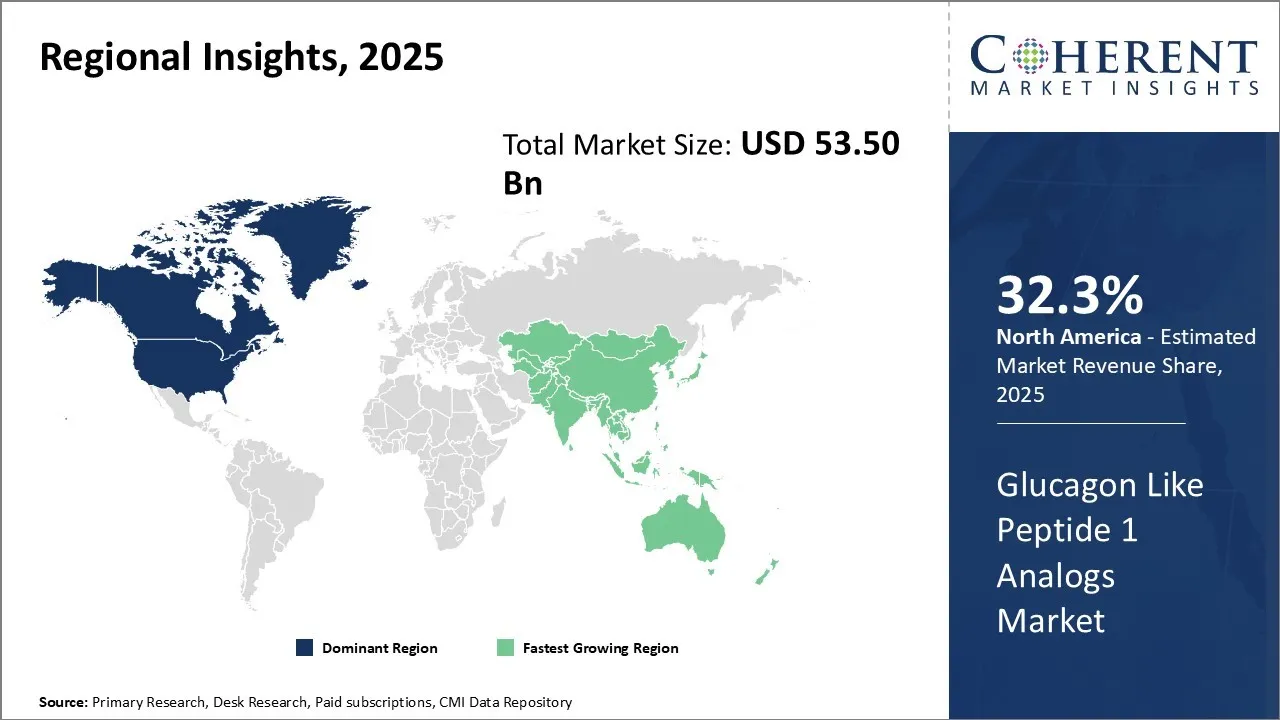
North America's dominance in the glucagon-like peptide 1 analogs market, with an estimated share of 32.3% in 2026, can be attributed to its well-established pharmaceutical ecosystem and robust healthcare infrastructure. These factors have fostered the development and widespread adoption of innovative treatments like glucagon-like peptide 1 analogs. Pharmaceutical giants, such as Eli Lilly and Novo Nordisk, are capitalizing on favorable regulatory policies and reimbursement frameworks to expand access to these therapies, ensuring they are available to a broader population. The collaborations between biotech firms, academic institutions, and healthcare providers, as exemplified by Canada’s diabetes framework development, are accelerating innovation and patient access, reinforcing North America’s position as the leader in the glucagon like peptide 1 analogs market. This newly announced framework will likely further drive market penetration and improve outcomes for individuals with diabetes in the region.
For instance, in October 2022, the government of Canada announced a new framework for diabetes in Canada. The Framework provides common policy direction to help align efforts and to support improved access prevention and treatment for all types of diabetes to ensure better health outcomes. The Framework was developed with input from people living with diabetes, academics, researchers, not-for-profit organizations, provinces and territories, Indigenous communities, and other key partners to ensure it significantly improves the lives of those with diabetes.
Asia Pacific is expected to exhibit the fastest growth in the glucagon like peptide 1 analogs market with a share of 25.2% in 2026, propelled by increasing diabetes prevalence, growing healthcare awareness, and expanding access to advanced treatments. Government initiatives targeting chronic disease management, combined with rising healthcare expenditure, create a fertile environment for market expansion. Countries, such as China, Japan, and India, are witnessing rapid adoption of GLP-1 therapies backed by local manufacturing and import facilitation. Additionally, a growing middle class and improving insurance coverage stimulate demand.
Market players in the glucagon like peptide 1 analogs market are heavily focused on securing regulatory approvals, which is expected to significantly contribute to the growth of the "Others" segment of the market over the forecast period. Regulatory approvals allow for the introduction of new drugs, including innovative formulations and generic versions, thus expanding treatment options and increasing market competition.
For instance, in December 2023, Lupin Limited, one of the global pharmaceutical companies, received tentative approval from the U.S. Food and Drug Administration (FDA) for its Abbreviated New Drug Application (ANDA) for Sitagliptin Tablets USP (25 mg, 50 mg, and 100 mg), a generic version of Januvia Tablets, originally developed by Merck Sharp and Dohme Corp. This approval facilitates Lupin’s entry into the glucagon like peptide 1 analogs market, offering a more affordable alternative to existing therapies, thus enhancing the "others" segment. With increasing competition and a broader range of available treatments, regulatory approvals are expected to play a pivotal role in driving growth in the glucagon like peptide 1 analogs market.
The U.S. glucagon like peptide 1 analogs market remains at the forefront of adoption, with high patient awareness and widespread access to advanced therapies. Leading pharmaceutical companies like Eli Lilly and Novo Nordisk have significantly influenced treatment paradigms with blockbuster drugs such as Trulicity and Ozempic, respectively. These medications are driving the market, supported by strong intellectual property laws, extensive clinical research networks, and comprehensive payer reimbursement systems that facilitate rapid market uptake. Collaborative efforts between healthcare providers and manufacturers also ensure patient-centric care, further solidifying the U.S.'s role as a leader in market innovation.
An example of this ongoing innovation is Pfizer Inc.'s progress in developing Danuglipron, a once-daily modified-release oral GLP-1 receptor agonist. In July 2024, Pfizer Inc., a global pharmaceutical leader, announced the progress of its development of a once-daily modified release formulation for Danuglipron, an oral glucagon-like peptide-1 (GLP-1) receptor agonist. The company has selected its preferred formulation based on encouraging pharmacokinetic data from ongoing studies (NCT06153758). Pfizer Inc. will conduct dose optimization studies in the second half of 2024 to evaluate multiple doses of this formulation, which will inform registration-enabling studies.
Canada glucagon like peptide 1 analogs market is witnessing significant growth, driven by the rising prevalence of type 2 diabetes and obesity. With more Canadians affected by these conditions, the adoption of GLP-1 therapies has increased, particularly in urban areas where access to healthcare is better. The Canadian government has actively supported the adoption of these therapies through initiatives like the Diabetes 360° strategy, which aims to reduce the national diabetes burden by improving access to innovative treatments. Moreover, public healthcare programs and insurance support for chronic disease management have enhanced the affordability and accessibility of glucagon like peptide 1 analogs across diverse populations, including those in remote areas.
For instance, in November 2023, Lilly Canada, a medicine company, announced that Mounjaro (tirzepatide) is now available in Canada. Mounjaro is a new once-weekly GIP (glucose-dependent insulinotropic polypeptide) and GLP-1 (glucagon-like peptide-1) receptor agonist, and is indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes. Mounjaro has not been studied in patients with a history of pancreatitis and is not indicated for use in patients with type 1 diabetes mellitus and is not indicated for weight management.
The U.K. glucagon like peptide 1 analogs market is experiencing significant growth, driven by the rising adoption of drugs like Ozempic and Wegovy for type 2 diabetes and obesity. This growth is fueled by increased access through online pharmacies, where around 500,000 individuals are bypassing NHS (National Health Service) channels. Generic alternatives for drugs like Saxenda and Victoza are expected to lower costs and improve accessibility. Additionally, the NHS is rolling out weight-loss injections in pharmacies to combat obesity, and the MHRA (Medicines and Healthcare products Regulatory Agency) continues to monitor the safety of these drugs due to concerns over side effects like nausea. These trends highlight the growing demand for GLP-1 therapies in the U.K., supported by improved access and regulatory oversight.
For instance, in March 2023, The National Health Service in the U.K. approved Wegovy (semaglutide) by Novo Nordisk A/S, making it available to thousands of people with obesity in England. Wegovy mimics GLP-1 to suppress appetite and support weight loss.
China’s glucagon like peptide 1 analogs market is rapidly evolving due to substantial government focus on diabetes control and expanding healthcare infrastructure across urban and rural regions. The market is influenced by both multinational corporations like Novo Nordisk and growing local pharmaceutical firms investing in biopharmaceutical development. Regulatory bodies in China are actively easing drug approval pathways, which is facilitating the introduction of novel glucagon like peptide 1 analogs tailored to the country's demographic. Factors like increasing urbanization, improved health insurance coverage, and rising disposable incomes are driving the growing adoption of GLP-1 therapies in the region. These changes have created an environment conducive to the swift uptake of new treatments.
On February 7, 2024, Innovent announced that the New Drug Application (NDA) for mazdutide, a glucagon-like peptide-1 receptor (GLP-1R) and glucagon receptor (GCGR) dual agonist, had been accepted by the Center for Drug Evaluation (CDE) of the National Medical Products Administration (NMPA) of China, for chronic weight management in adults with obesity or overweight.
The India glucagon like peptide 1 analogs market is estimated to be valued at USD 1.39 Bn in 2026 and is expected to reach USD 3.01 Bn by 2032, exhibiting a compound annual growth rate (CAGR) of 11.7% from 2026 to 2032.
India’s glucagon like peptide 1 analogs market is steadily growing, driven by a significant diabetic population and increasing investments in healthcare. As awareness about newer treatment options rises among both healthcare professionals and patients, adoption of these therapies continues to expand. The government's efforts to improve healthcare accessibility, along with financial assistance programs, play a crucial role in enabling market growth. Multinational companies like AstraZeneca and Novo Nordisk dominate product availability, but emerging domestic biopharma companies are making strides in the market with biosimilar developments.
For instance, on January 3, 2024, Glenmark Pharmaceuticals Ltd., an Indian multinational pharmaceutical company, announced that it had launched a biosimilar of the anti-diabetic drug, Liraglutide, in India. The drug is being marketed under the brand name Lirafit following the approval from the Drug Controller General of India (DCGI). The drug has been approved globally for the management of type 2 diabetes mellitus in adult patients in the U.S. and the European Union. Moreover, in July 2022, Glenmark Pharmaceuticals Ltd. launched Sitagliptin and its Fixed Dose Combinations (FDCs) for adults with Type 2 diabetes in India.
|
Category |
Details |
|
Generic Name |
Liraglutide, Semaglutide, Dulaglutide, Lixisenatide, Tirzepatide |
|
Brand Names (India) |
Victoza, Saxenda, Ozempic, Rybelsus, Wegovy, Trulicity, Lyxumia, Mounjaro |
|
Therapeutic Class |
Antidiabetic (GLP-1 Receptor Agonist); Anti-Obesity (specific formulations) |
|
Approved Indications |
Type-2 Diabetes Mellitus (all); Weight Management (Liraglutide [Saxenda], Semaglutide [Wegovy]); Obesity with comorbidities (Saxenda) |
|
Dosage Forms (Approved) |
|
|
Key Manufacturers |
Novo Nordisk (Liraglutide, Semaglutide), Eli Lilly (Dulaglutide, Tirzepatide), Sanofi (Lixisenatide) |
|
OTC/Rx Status |
Prescription-only (Rx) |
|
Post-Marketing Requirements |
Pharmacovigilance (PvPI) compliance mandatory; Periodic safety update reports (PSURs); Risk Management Plans (RMPs) for newer agents |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
To learn more about this report, Request Free Sample
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 59.01 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 10.3% | 2033 Value Projection: | USD 117.21 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
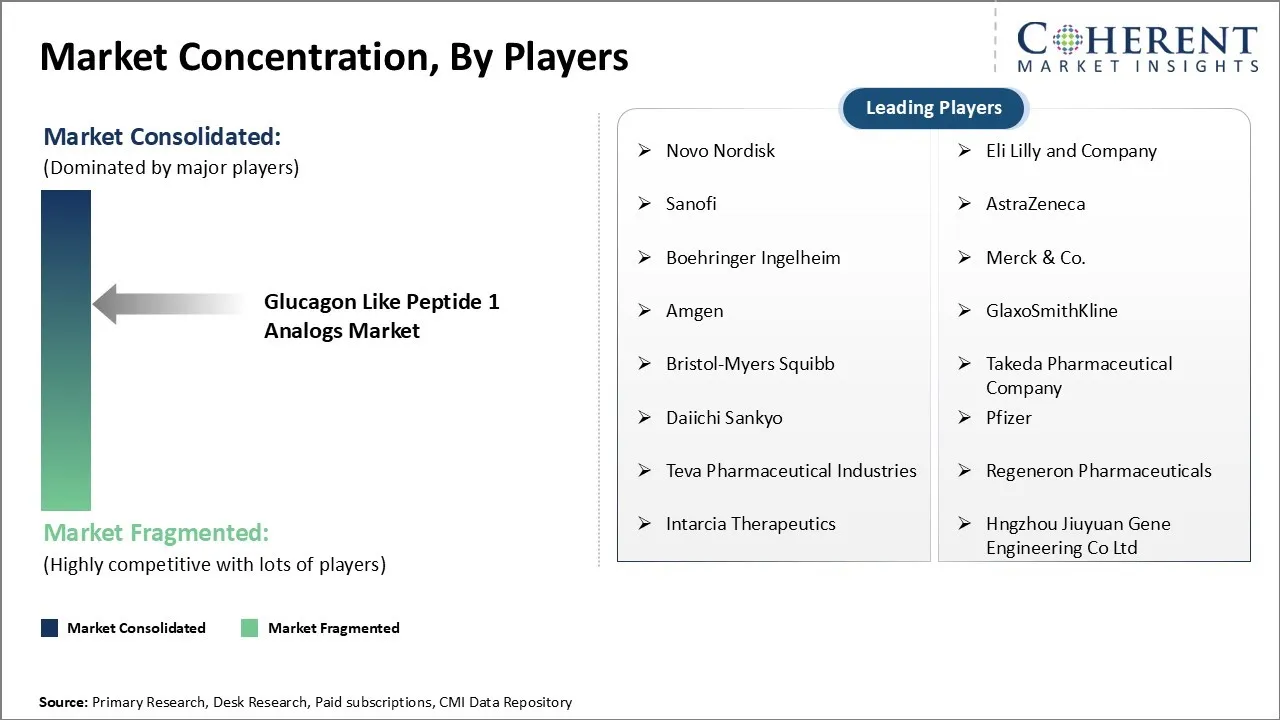
Novo Nordisk, Eli Lilly and Company, Sanofi, AstraZeneca, Boehringer Ingelheim, Merck & Co., Amgen, GlaxoSmithKline, Bristol-Myers Squibb, Takeda Pharmaceutical Company, Daiichi Sankyo, Pfizer, Teva Pharmaceutical Industries, Regeneron Pharmaceuticals, Intarcia Therapeutics, and Hngzhou Jiuyuan Gene Engineering Co Ltd |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
To learn more about this report, Request Free Sample
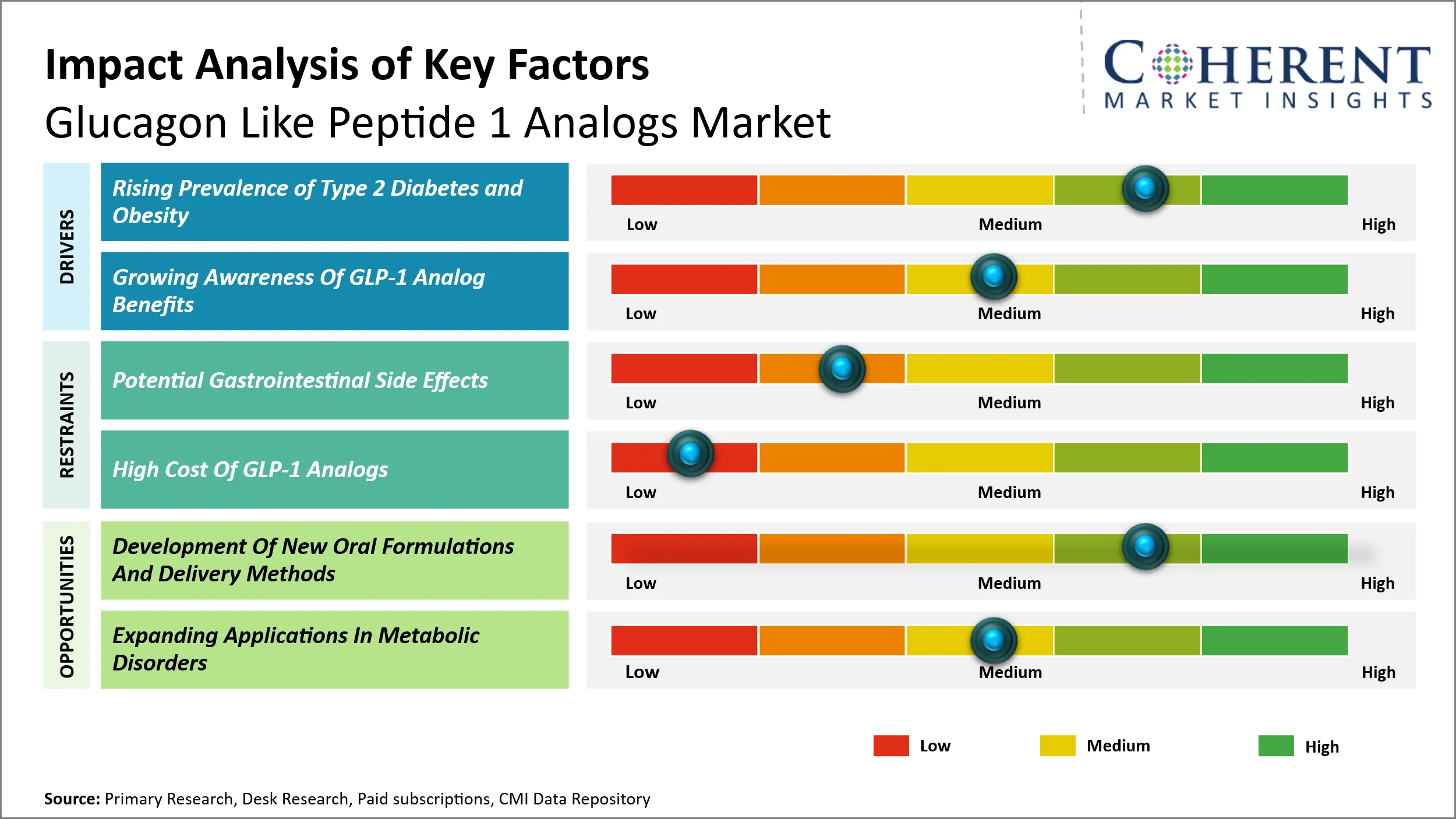
The escalating prevalence of type 2 diabetes and obesity worldwide is a significant factor driving the demand for Glucagon Like Peptide 1 (GLP-1) analogs. As lifestyle changes, sedentary behavior, and unhealthy dietary patterns become increasingly common, the incidence of these metabolic disorders continues to surge. For example, introduction of injectable and oral long-acting formulations has revolutionized the way these drugs are administered. Exenatide extended-release (ER) injection (Byetta, Bydureon), liraglutide ER injection (Victoza, Saxenda), and dulaglutide ER injection (Trulicity) are some of the long-acting injectable incretin mimetics that have been approved by U.S. Food and Drug Administration (FDA) since 2010. These formulations offer several advantages over the short-acting injectable and oral forms, including improved patient compliance, reduced injection frequency, and better glycemic control.
In June 2024, a recent study by the National Library of Medicine, Secular Trends in Diabetes in India (STRiDE-I), revealed a concerning rise in type 2 diabetes (T2DM) prevalence among younger individuals (ages 20–39) in southern India. Over a 10-year period, the prevalence in younger individuals increased by 36%, from 4.5% to 7.8%, significantly outpacing the 11% rise in older individuals (ages 40 and above). The incidence of T2DM in the younger group surged by 120%, highlighting a troubling trend in youth diabetes rates. Key risk factors contributing to this increase were obesity and a family history of diabetes, both of which were more prevalent among younger individuals. The study also found that cardiometabolic risks, including dyslipidemia and hypertension, rose more rapidly in the younger population. These findings underscore the need for targeted interventions to address diabetes risk factors in the youth population.
The glucagon like peptide 1 analogs market stands to benefit significantly from advancements in oral formulations and innovative delivery methods. Traditionally, glucagon like peptide 1 analogs have been administered via subcutaneous injections, which can pose adherence challenges among patients, limiting widespread acceptance and consistent usage. The development of oral glucagon like peptide 1 analogs offers a transformative opportunity to enhance patient convenience, improve compliance, and expand the target demographic, particularly among individuals with needle aversions or injection phobia. Additionally, innovations such as transdermal patches, inhalable formulations, and implantable devices are being explored to provide alternative, less invasive routes of administration. These novel delivery platforms could lower the treatment burden, enhance patient lifestyle compatibility, and facilitate more personalized diabetes and obesity management protocols.
The development of oral semaglutide (Ozempic, Rybelsus) can also drive the market growth. Semaglutide is a once-weekly injectable and a once-daily oral medication that makes it a versatile treatment option for patients with type 2 diabetes. The oral formulation of semaglutide, Rybelsus, was approved by the U.S. FDA in 2019, providing an alternative to injectable treatments for those who prefer oral administration.
For instance, according to a research article published by the National Institutes of Health (NIH) in December 2020, titled ‘Oral Nano Drug Delivery Systems for the Treatment of Type 2 Diabetes Mellitus,’ it was found that oral nano drug delivery systems for phytocompounds aimed at treating T2DM not only retain the benefits of oral administration but also address the limitations of traditional oral drug delivery methods.
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients