The global peptide therapeutics market is estimated to be valued at USD 53,869.3 Mn in 2026 and is expected to exhibit a compound annual growth rate (CAGR) of 6.8% during the forecast period, reaching USD 85,376.8 Mn in 2033.
The global peptide therapeutics market is projected to grow steadily during the forecast period. This can be attributed to rising prevalence of chronic & metabolic diseases, advancements in peptide drug development technologies, and growing interest in peptide-based therapeutics.
Therapeutic peptides are short chains of amino acids with molecular weights typically between 500 and 5,000 Da. They play an important role in treating various diseases due to their high specificity, safety, and effectiveness.
The development of peptide drugs has grown significantly since the first therapeutic peptide, insulin, was introduced in 1922. Today, more than 80 peptide-based drugs have been approved worldwide, with many others under clinical development. This growing pipeline highlights the increasing importance of peptide therapeutics in the global pharmaceutical industry.
Innovation in peptide therapeutics is breathing new life into the target market. Companies are developing novel peptide-based therapeutics as well as delivery methods to stay ahead of the competition.
For instance, in August 2025, BioMed X and Novo Nordisk partnered to develop novel oral-formulation technologies for therapeutic peptides. The collaboration seeks to develop innovative technologies that improve oral delivery as well as absorption of peptide-based drugs.
|
Current Event |
Description and its Impact |
|
U.S. FDA Regulatory Modernization and Accelerated Approval Pathways |
|
|
Artificial Intelligence and Computational Drug Discovery Revolution |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
As per Coherent Market Insights latest analysis, branded peptide segment is expected to dominate the market, accounting for more than 61% of the market share in 2026. This can be attributed to rising preference for branded peptide therapeutics globally.
Branded peptide therapeutics are popular because they are made and sold by well-known pharmaceutical companies with strong research and development teams. These companies invest heavily in clinical research to make sure their medicines are safe, effective, and approved by major health authorities. Because of this, doctors and patients often choose branded drugs as they trust their quality and reliability.
Furthermore, branded peptide therapeutics usually offer better patient support programs, clear usage instructions, and are more widely available in pharmacies and hospitals. Their strong reputation and reliable results make them the preferred choice for many people. This trust and demand contribute to the large market share held by branded peptide therapeutics.
To learn more about this report, Request Free Sample
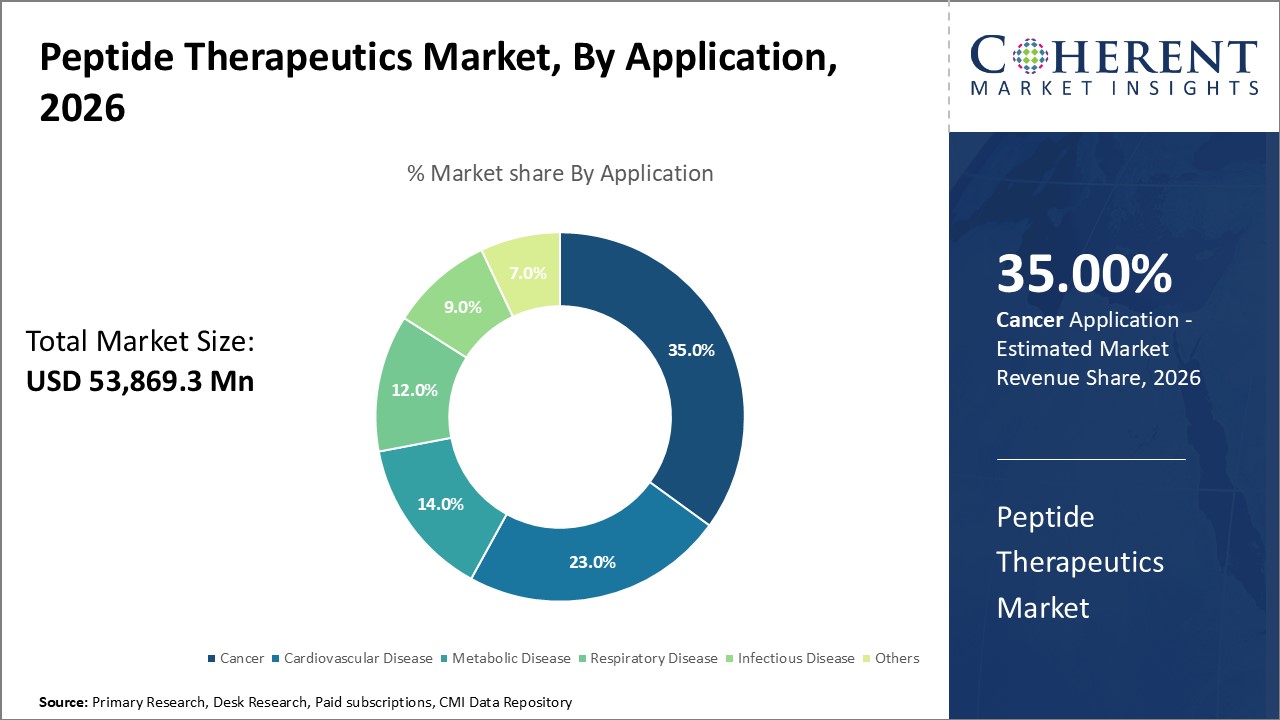
Cancer segment is projected to dominate the market over the forecast period. This is due to increasing usage of peptide therapeutics to treat cancer. Increasing cancer incidence worldwide and rising need for more personalized and targeted therapies are fueling adoption of peptide-based treatments in oncology.
Peptide therapeutics have the tendency to specifically target cancer cells without harming healthy tissue. This precision reduces side effects compared to traditional chemotherapy and improves treatment effectiveness.
Ongoing research and development have broadened the use of peptides in cancer treatment, such as in targeted drug delivery, immunotherapy, and combination therapies. Pharmaceutical companies are investing heavily in peptide-based cancer drugs because of their high success rates in clinical trials and their potential to improve patient outcomes.
To learn more about this report, Request Free Sample
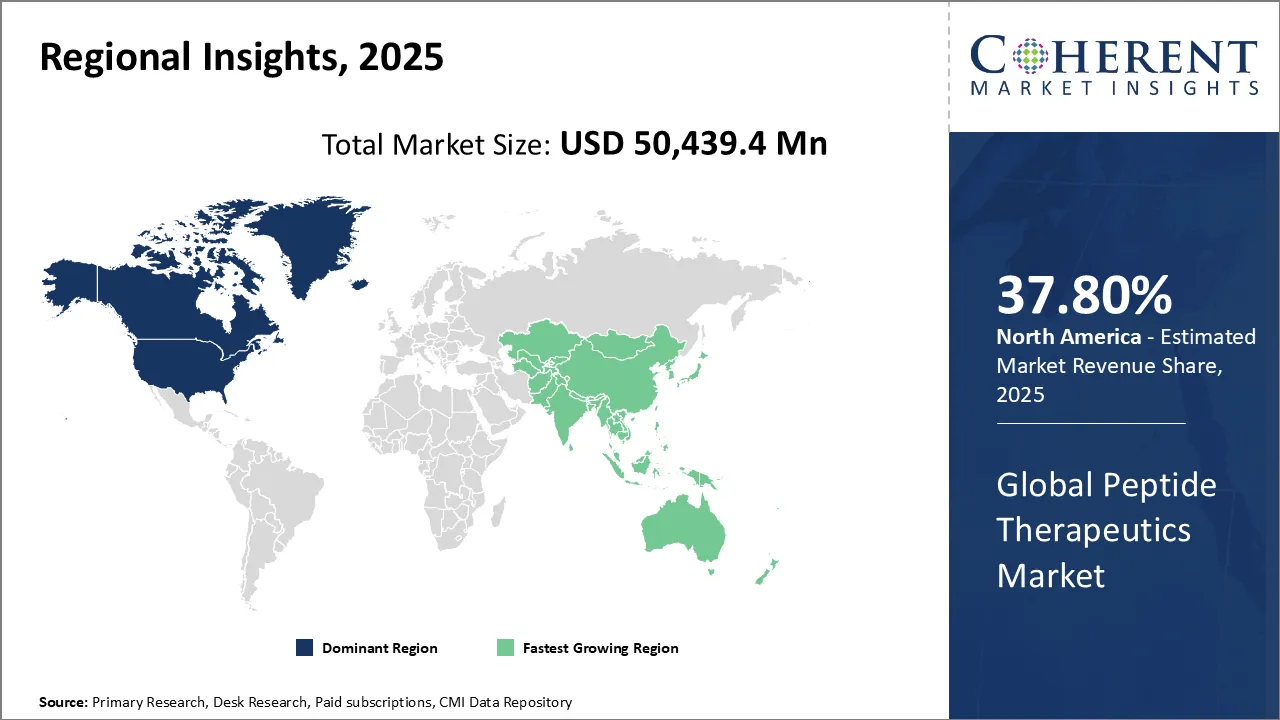
North America is expected to retain its dominance over the global peptide therapeutics market during the forecast period, capturing 37.8% share in 2026. This can be attributed to rising cancer cases, growing demand for advanced therapies, and strong presence of leading players.
North America is home to some of the leading pharmaceutical and biotech companies, including Amgen, Eli Lilly, and Pfizer. These companies are heavily investing in R&D to develop advanced treatments like peptide-based therapeutics.
For instance, in September 2026, Eli Lilly announced plans to construct a $6.5 billion manufacturing plant in Houston, Texas. The new facility will produce active pharmaceutical ingredients for orforglipron and for Lilly's cancer, autoimmune, and other advanced therapies. These developments will likely support market expansion in the region during the forecast period.
Asia Pacific is slated to emerge as the most remunerative market for peptide therapeutics during the assessment period. This is attributable to increasing healthcare investments, expanding pharmaceutical manufacturing, and rising awareness of peptide-based treatments among patients and doctors.
Countries like China, Japan, and India are seeing rapid adoption of peptide therapeutics due to supportive government initiatives as well as a growing patient population. Additionally, lower production costs and a rising number of clinical trials make Asia Pacific a highly attractive market for peptide therapeutics.
Many companies are establishing their facilities across Asia Pacific nations like India and China to boost their sales. For instance, in March 2026, AstraZeneca announced a $2.5 billion investment in a new R&D center, manufacturing facilities, and biotech collaborations in Beijing.
The United States peptide therapeutics industry is expected to register steady growth during the forecast period. This is mostly due to increasing prevalence of chronic diseases, especially cancer, diabetes, and cardiovascular disorders, which require advanced peptide-based treatments.
Strong R&D capabilities and well-established pharmaceutical infrastructure are also boosting market growth in the nation. Similarly, increasing investments by leading biotech companies in peptide development are supporting market expansion.
The U.S. is also benefiting from regulatory support, such as fast-track approvals for innovative therapies, and growing awareness among healthcare professionals and patients regarding the benefits of peptide therapeutics. Increasing investments in personalized medicine and targeted drug delivery are further boosting the adoption of peptide-based treatments in the country.
China is becoming a hotbed for manufacturers of peptide therapeutics. This can be attributed to supportive government initiatives, including subsidies and favorable policies for biotech and pharmaceutical industries. Rapid expansion of domestic pharmaceutical manufacturing and increasing collaborations with global biotech companies are also accelerating market development.
Moreover, rising awareness among doctors and patients about peptide-based treatments, along with a growing population of patients with chronic and lifestyle-related diseases, is driving demand. China’s focus on cost-effective production and scaling up of peptide therapeutics is attracting both domestic and international players to strengthen their presence in the market.
For instance, China has implemented policies like "Made in China 2026" and Circular No. 53 to stimulate transformation of the country's life sciences industry, including the peptide therapeutics sector. These policies are creating a fertile ground for peptide therapeutics market growth in the nation.
Leading peptide therapeutics companies are focusing on strong research and development (R&D) to create innovative and effective treatments. They are investing heavily in discovering new peptides that can target specific diseases such as cancer, diabetes, and cardiovascular disorders.
Many companies are also forming collaborations with universities, research institutes, and other pharmaceutical firms to speed up the development process and share expertise. Similarly, they are using mergers and acquisition to expand their portfolio as well as boost their sales.
For instance, in September 2025, Pfizer acquired Metsera and its next-generation obesity portfolio. The deal aims to strengthen Pfizer’s position in developing innovative treatments for obesity.
Another key strategy is expanding their market presence globally. Companies are entering new regions, especially in Asia Pacific, to reach a larger patient base and benefit from growing healthcare infrastructure. They are also adopting advanced manufacturing technologies to reduce production costs and improve the quality of peptide drugs.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 53,869.3 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 6.8% | 2033 Value Projection: | USD 85,376.8 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Teva Pharmaceutical Industries Ltd, Novo Nordisk A/S, OPKO Health Inc., Lilly, Hanmi Pharm. Co., Sanofi, Circle Pharma, Pfizer Inc., Novartis AG, PeptiDream Inc., Amgen Inc., AstraZeneca, Bachem Holding AG, CordenPharma International, Ipsen Pharma, Merck KGaA, PolyPeptide Group, Bayer AG. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The global cancer burden is rising rapidly. According to the World Health Organization (WHO), the annual number of new cancer cases is expected to exceed 35 million by 2050. This increase is driving demand for peptide therapeutics, which are increasingly used in cancer treatment.
Peptide therapeutics play an important role in developing new drugs and treatments. Thus, increasing research and development activities by leading pharmaceutical and biotech companies to expand their product portfolios are expected to drive market growth over the forecast period. For example, an August 2022 article in Biomedicines highlighted the use of peptide-based drug development for creating new cancer treatments, demonstrating the growing impact of R&D in this field.
There is a rising preference for targeted therapies with improved safety/efficacy profiles in the contemporary world. This is expected to drive demand for peptide therapeutics during the forthcoming period.
Peptides offer high specificity, lower off‐target effects, and lower immunogenicity compared to some biologics/small molecules. This is making them attractive for complex diseases like cancer and metabolic disorders.
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients