The Human Combination Vaccines Market is estimated to be valued at USD 11 Bn in 2026 and is expected to reach USD 20.1 Bn by 2033, growing at a compound annual growth rate (CAGR) of 9% from 2026 to 2033.
This market is being propelled by growing rates of communicable diseases and expanding numbers of children worldwide. Young children face heightened susceptibility to numerous infections during their early developmental years due to their immature immune defenses. This increased vulnerability translates into a greater need for combination vaccination products.
The governmental programs and partnerships aimed at raising public awareness about immunization and ensuring accessible or complimentary vaccine distribution also help in driving growth. Numerous nations have implemented systematic immunization protocols that mandate standardized vaccination timelines for all infants to protect against prevalent infectious conditions. These include hepatitis, diphtheria, polio, and tetanus, which amplifies vaccine demand.
|
Current Event |
Description and the Impact |
|
Technological and Scientific Advancements |
|
|
Public Health and Epidemiological Trends
|
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
AI is shifting the Human Combination Vaccines Market by making it possible to automate, use predictive analytics, and improve vaccine development. AI-powered tools make it easier to identify antigens, predict immune responses, and optimize formulations, which cuts down on the need for manual testing and lowers development costs. Real-time information makes it easier to manage production quality and make decisions. Pharmaceutical companies can develop multi-antigen combination vaccines more efficiently, accurately, and safely by using AI. This is going to enhance immunization coverage and encourage adoption by healthcare providers and public health organizations around the world.
In September 2025, the University of Oxford, through its strategic partnership with the Ellison Institute of Technology (EIT), secured £118 million in research funding to establish a major new vaccine research programme. Led by the Oxford Vaccine Group, the University-based initiative—CoI-AI (Correlates of Immunity–Artificial Intelligence)—will integrate Oxford’s strengths in human challenge studies, immunology, and vaccine development with EIT’s advanced artificial intelligence technologies to improve understanding of immune responses to infection and vaccine-induced protection.
In terms of product type, the inactivated vaccine segment contributes the highest share of 60% in 2026 of the market owing to the proven safety profile and established efficacy that healthcare providers have with inactivated formulations. Using inactivated vaccines allows healthcare systems to give multi-disease protection in fewer doses, which improves vaccination rates and lowers healthcare costs. The expanding national immunization schedules and growing birth rates in the emerging markets are driving higher use of affordable innovative formulations.
For instance, in May 2025, the U.S. Department of Health and Human Services (HHS) and the National Institutes for Health (NIH) announced that they were working on the next-generation, universal vaccine platform called Generation Gold Standard. It would use a beta-propiolactone (BPL)-inactivated, whole-virus platform. The goal of the initiative is to protect against several viruses that are likely to cause pandemics, such as H5N1 avian influenza and coronaviruses like SARS-CoV-2, SARS-CoV-1, and MERS-CoV.
In terms of age group, the children segment contributes the highest share of 74% in 2026 of the market owing to mandatory childhood immunization programs and growing awareness about preventable diseases globally. Vaccinating children allows healthcare systems to give multiple disease protections in single visits, which improves immunization coverage and reduces clinic visits. Rising birth rates in developing regions and growing vaccination schedules recommended by health authorities are driving higher demand for pediatric combination vaccines. This increasing focus on early life disease prevention makes children the target population for combination vaccines.
For instance, in August 2024, the United Nations Children's Fund (UNICEF) delivered 60,000 doses of the PENTA-Hib combined vaccine, with help from the Japanese government. This vaccine safeguards kids during their first year of life from five serious diseases namely diphtheria, tetanus, pertussis, Hepatitis B, and Haemophilus influenzae type b (Hib). It does this by using a whole-cell pertussis component.
In terms of combination type, the DTaP/IPV/Hep B segment contribute the highest share of 40% in 2026 of the market owing to widespread inclusion in national childhood immunization schedules and strong recommendations from global health organizations. They allow healthcare providers to protect against five serious diseases in one shot, which improves vaccine compliance and reduces injection burden. The growing use of pentavalent vaccines in developing countries and increased government funding for routine optimization programs are driving higher demand for this combination. They provide comprehensive protection against diphtheria, pertussis, polio, and hepatitis B, making them the most widely used combination vaccine worldwide.
For instance, in July 2025, with Gavi's assistance, the hexavalent vaccine was introduced in Senegal and Mauritania. This is the first time that low-income nations have received this vital six-in-one vaccine. The six illnesses that the combo vaccine protects against are diphtheria, tetanus, pertussis (whooping cough), hepatitis B, Haemophilus influenzae type B (Hib), and poliomyelitis.
In terms of distribution channel, the hospital pharmacies segment is expected to lead the market with largest share of 47% in 2026, as most childhood vaccinations being given in hospital settings and clinics with proper cold chain facilities. They allow healthcare providers to store and handle vaccines in strict temperature controls, which keeps vaccine quality and ensures patient safety. The growing number of immunization centers in hospitals and increased public trust in hospital-based vaccination programs are driving higher distribution through this channel. They provide trained medical staff and quick access to emergency care if needed, making hospital pharmacies the top distribution point for combination vaccines worldwide.
To learn more about this report, Download Free Sample
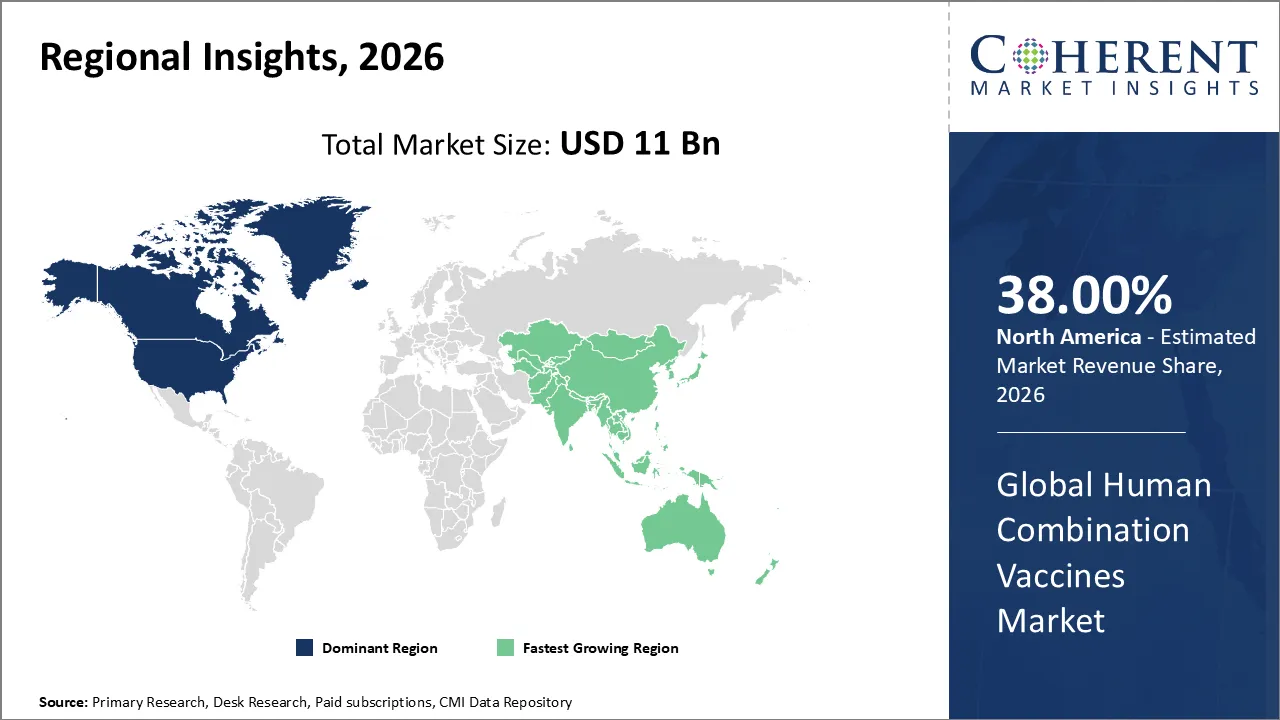
North America has remained the dominant region with 38% in 2026 of the global Human Combination Vaccines Market over the past decade. The growth is attributable to the surging demand for efficient immunization solutions amid rising infectious disease threats. The parents prefer fewer shots for their kids, boosting compliance and cutting healthcare visits. The cutting-edge multivalent formulations help in the protection against multiple pathogens in one single dose. The government programs and high vaccination rates in the U.S. and Canada are resulting in increased adoption of these vaccines. The growth is also backed by strong research and development investments and regulatory approvals that speed up innovation.
The Asia Pacific region has emerged as the fastest growing market for human combination vaccines globally in recent years. The increasing birth rates in countries like India and Indonesia are creating high demand for pediatric immunization programs. The government initiatives aimed at promoting universal vaccination coverage like India’s Mission Indradhanush are also helping in increasing market reach. The increasing spending in the healthcare sector allows for better vaccine accessibility and affordability.
The growing awareness about preventive healthcare and the convenience of fewer injection visits is propelling the parental acceptance of combination vaccines. The region's improving cold chain infrastructure facilitates better vaccine distribution to remote areas. The partnerships between international vaccine manufacturers and local pharmaceutical companies are improving production capabilities and minimizing costs.
In 2026, the market in the U.S. is expected to be strong due to established immunization schedules and high compliance rates. The healthcare providers and parents prefer combination shots to streamline clinic visits and improve vaccination completion amongst children. Comprehensive CDC guidelines and extensive insurance coverage facilitate widespread adoption across the population.
For instance, in January 2023, AstraZeneca acquired Icosavax Inc., a biopharmaceutical firm based in the U.S. that is developing promising vaccines using protein virus-like particle (VLP) technology. Icosavax's leading experimental vaccine candidate, IVX-A12, is expected to enhance AstraZeneca's late-stage pipeline for vaccines and immune therapies, particularly regarding respiratory syncytial virus (RSV). IVX-A12 is the first of its kind combination protein VLP vaccine that is ready for Phase III testing. It is meant to protect against both RSV and hMPV.
In 2026, the market in China is booming owing to expanded government immunization initiatives and increasing healthcare expenditure. The large pediatric population creates consistent demand, while urbanization improves healthcare access in various regions. Also, the rising public awareness of vaccine preventable diseases and the advancement of cold chain infrastructure contribute to greater market penetration in the country.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 11 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 9% | 2033 Value Projection: | USD 20.1 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Sanofi, Pfizer Inc., Cadila Healthcare Ltd., GlaxoSmithKline plc, CSL Ltd., Mitsubishi Tanabe Pharma Corp., Daiichi Sankyo Company, Limited, Merck & Co., Crunchbase Inc., Mass Biologics, Meiji Holdings Co. Ltd., Serum Institute of India, Moderna, AstraZeneca, Influvac, Biological, PaxVax Corporation, Emergent BioSolutions Inc., CNBG, Takeda Pharmaceutical Co. Ltd., and NPS MedicineWise |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The rising prevalence of infectious diseases worldwide is a primary driver for the human combination vaccines industry. As global population grows and urbanization intensifies, disease transmission rates have sped up significantly. Healthcare systems are increasingly burdened by outbreaks of preventable diseases in developing regions with limited medical infrastructure. Combination vaccines provide an efficient solution by providing protection against multiple pathogens in a single formulation, thereby reducing the overall disease burden. This approach proves especially valuable during public health emergencies and routine immunization programs.
The Human Combination Vaccines Market is shaped by strong demand fundamentals supported by expanding immunization programs, improvements in vaccine delivery efficiency, and increasing adoption of multi-antigen formulations across routine vaccination schedules. Combination vaccines reduce the number of injections required per individual, improving adherence rates and simplifying administration within pediatric and adult immunization programs.
Global immunization coverage for core antigens such as diphtheria, tetanus, pertussis, poliomyelitis, and Haemophilus influenzae type b consistently exceeds 80% in many regions, according to international health agencies. This high coverage has reinforced the preference for combination vaccines within national immunization frameworks, particularly under Expanded Programme on Immunization (EPI) models. Procurement data from public health agencies shows sustained growth in demand for pentavalent and hexavalent vaccines, driven by logistical efficiency, reduced cold-chain burden, and lower per-dose handling requirements.
Manufacturing trends indicate capacity expansion among leading vaccine producers, while regulatory filings and clinical trial databases reflect steady growth in the development of advanced combination formulations integrating newer antigens. Clinical development activity for combination vaccines has increased by an estimated 15–20% annually in recent years, highlighting continued innovation.
Health system analyses demonstrate that combination vaccines lower overall immunization program costs by reducing clinic visits and administration complexity. Regionally, mature markets exhibit stable demand supported by established schedules, while emerging economies show rising uptake following inclusion of combination vaccines in national programs, with coverage rates in several countries surpassing 90%.
Definition: The Human Combination Vaccines Market facilitates immunization protection against multiple diseases through single-dose administration. It provides vaccination solutions that allow healthcare providers to protect patients against several pathogens in a safe, effective, and convenient way. Healthcare systems can use these vaccines to reduce the number of injections required, improve vaccination compliance, simplify immunization schedules, and improve patient comfort. Delivery methods include intramuscular injections, subcutaneous administration, and emerging needle-free technologies.
Share
Share
Abhijeet Kale is a results-driven management consultant with five years of specialized experience in the biotech and clinical diagnostics sectors. With a strong background in scientific research and business strategy, Abhijeet helps organizations identify potential revenue pockets, and in turn helping clients with market entry strategies. He assists clients in developing robust strategies for navigating FDA and EMA requirements.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients