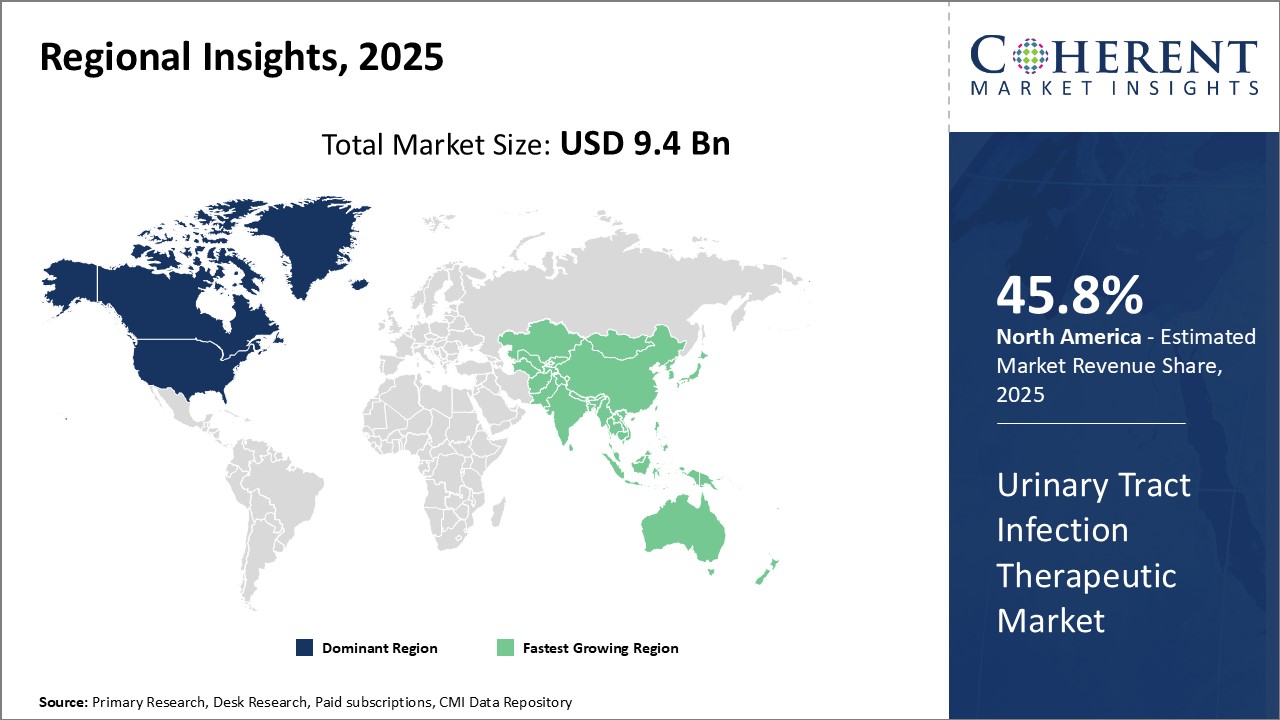
Global Urinary Tract Infection Therapeutic Market is estimated to be valued at USD 9.4 Bn in 2025 and is expected to exhibit a CAGR of 3.20% during the forecast period (2025-2032).
The global urinary tract infection therapeutic market is experiencing strong growth due to the high prevalence of diabetes and kidney stones across the globe. Moreover, increasing prevalence of urinary tract infection (UTI) and growing geriatric population across the globe is expected to boost growth of the market. However, factors such as adverse effects associated with the use of UTI medication and lack of awareness among people are expected to hamper growth of the urinary tract infection therapeutic market.
Global Urinary Tract Infection Therapeutic Market: Regional Insights
Based on geography, the global urinary tract infection therapeutic market is segmented into North America, Latin America, Europe, Asia Pacific, and Middle East & Africa.
Among regions, North America is expected to gain highest share in the market over the forecast period owing to the increasing prevalence of diabetes and kidney stones and increasing geriatric population in this region, especially in the United States. For instance, a urinary tract infection (UTI) is an infection in any part of the urinary system. Diabetics are prone to UTIs or diabetes raises a person's risk for UTIs. According to the American Diabetes Association (ADA), over 37 million people of all ages (11.3.20% of the U.S. population) had diabetes. The percentage of Americans age 65 and older remains high, at 29.2%, or 15.9 million seniors.
Asia Pacific is also expected to witness significant growth in the global urinary tract infection therapeutic market due to the rise in incidence of urinary tract infection, especially in pregnant women, and growing geriatric population in the region. For instance, according to the National Center for Biotechnology Information (NCBI), urinary tract infections (UTIs) are common in pregnant women and pose a great therapeutic challenge, since the risk of serious complications in both the mother and her child is high. The upper UTIs in particular may lead to significant morbidity for both the mother and the fetus.
Figure 1. Global Urinary Tract Infection Therapeutic Market Value (USD Bn), by Region, 2025
To learn more about this report, Download Free Sample
Global Urinary Tract Infection Therapeutic Market Drivers:
High prevalence of urinary tract infection across the globe is expected to augment the growth of the global urinary tract infection therapeutic market over the forecast period. For instance, Urinary tract infection is the infection of bladder, urethra, ureter, and kidneys and occurs due to colonization of microbial pathogen in the bladder or the kidneys. According to the National Center for Biotechnology Information (NCBI), UTIs are among the most common bacterial infections, affecting 150 million people worldwide each year. This in turn expected to increase demand for urinary tract infection therapeutics worldwide.
Increasing prevalence of diabetes and increasing geriatric population is expected to propel the growth of the global urinary tract infection therapeutic market during the forecast period. For instance, urinary tract infections can be a particular problem for people with diabetes as sugar in the urine makes for a fertile breeding ground for bacteria. Diabetes raises a person's risk for urinary tract infections. According to the World Health Organization (WHO), more than 422 million people worldwide have diabetes, majority of them living in low-and middle-income countries, and 1.5 million deaths are directly attributed to diabetes each year.
Global Urinary Tract Infection Therapeutic Market Opportunities:
Increase in focus on the development of novel drugs is expected to provide significant growth opportunities for players in the global urinary tract infection therapeutic market. For instance, with the increase in prevalence of UTIs, the demand for UTI therapeutics is also increasing. In September 2020, CARB-X planned to invest over US$ 7.51 million in GlaxoSmithKline, with an aim to help develop GSK3882347, an orally bioavailable small molecule drug, for both the treatment and prevention of recurrent UTIs caused by E. coli bacteria.
Increase in research and development is expected to provide lucrative growth opportunities for players in the urinary tract infection therapeutic market. For instance, R&D in UTI treatment is expected to offer lucrative growth opportunities for players in the market. In August 2020, researchers at the California Polytechnic State University developed a new class of antibiotics for urinary tract infections (UTIs), which targets bacterial iron acquisition, at the American Chemical Society (ACS) Fall 2020 Virtual Meeting & Expo.
Urinary Tract Infection Therapeutic Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2024 | Market Size in 2025: | USD 9.4 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2025 To 2032 |
| Forecast Period 2025 to 2032 CAGR: | 3.20% | 2032 Value Projection: | USD 11.71 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Pfizer, Novartis AG, Cipla Inc., Bayer AG, AstraZeneca, Shionogi & Co. Ltd., and GlaxoSmithKline PLC, among others. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Urinary Tract Infection Therapeutic Market Trends:
The prevalence of UTIs (complicated UTIs) is expected to increase during the forecast period due to the increase in recurrence rate for UTIs, increasing bacterial resistance in UTI cases, and excessive use of antibiotics. This in turn is expected to increase demand for new urinary tract infection therapeutics (drugs) across the world, and this trend is expected to continue over the forecast period, driving the growth of the market.
Moreover, increase in consumer awareness about the association of diabetes and obesity with bladder dysfunction, increase in focus on the development of novel therapeutics, and the rapid deployment of innovative technologies for UTI treatment are among the key trends driving the overall growth of the urinary tract infection therapeutic market.
Global Urinary Tract Infection Therapeutic Market Restraints:
Adverse effects associated with the use of UTI medication are expected to hinder growth of the global urinary tract infection therapeutic market. For instance, side effects associated with UTI therapeutics like fluoroquinolones, such as variations in blood sugar levels and central nervous system problems such as insomnia, seizures, and anxiety may impede the growth of the urinary tract infection therapeutic market over the forecast period. More serious side effects can include antibiotic-resistant infections or C. diff infection, which causes diarrhea that can lead to severe colon damage and death.
Lack of awareness among people, in developing and underdeveloped countries, is expected to hamper growth of the global urinary tract infection therapeutic market. For instance, urinary tract infections are the second most common type of infection in the body. The infections can affect several parts of the urinary tract. UTIs are very common, if left untreated, bacteria-laden urine can travel to the kidneys and cause serious, even life-threatening complications. Thus, it is important to be aware of urinary tract infection and its therapeutics.
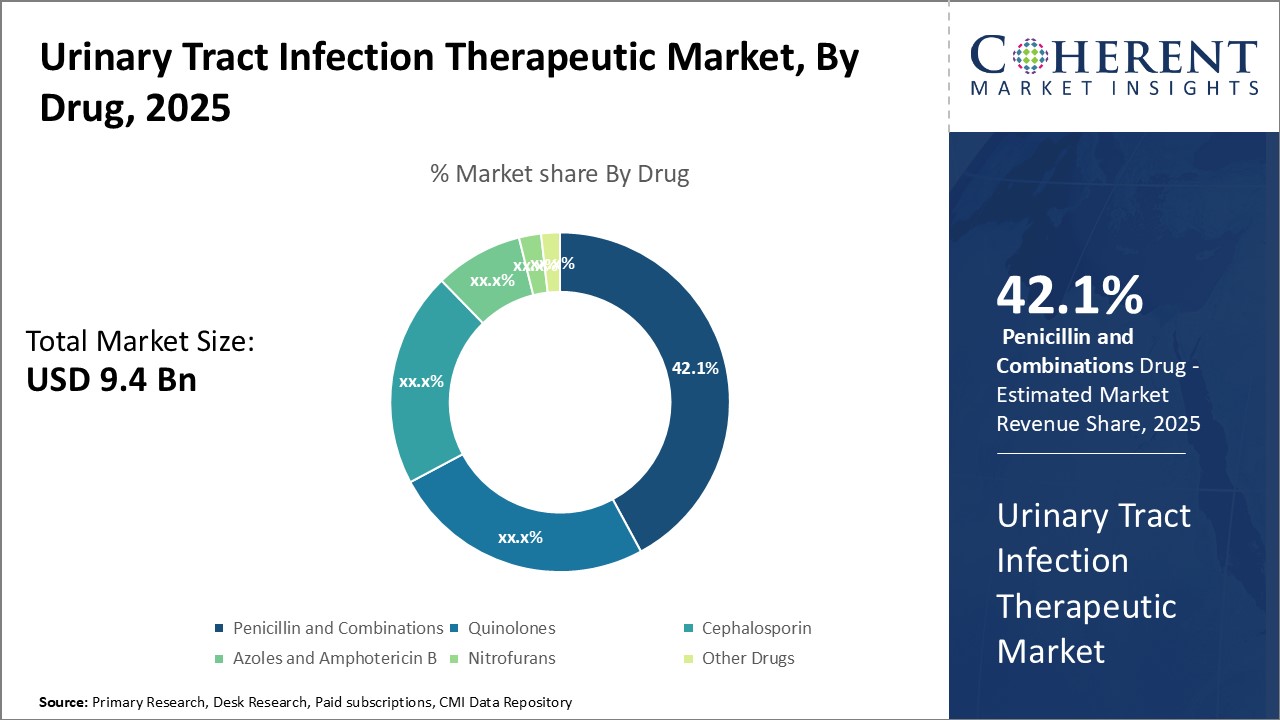
Figure 2. Global Urinary Tract Infection Therapeutic Market Share, By Drug, 2025
To learn more about this report, Download Free Sample
Global Urinary Tract Infection Therapeutic Market Segmentation:
The global urinary tract infection therapeutic market report is segmented into Drug, Indication, and Geography.
Based on Drug, the market is segmented into Penicillin and Combinations, Quinolones, Cephalosporin, Azoles and Amphotericin B, Nitrofurans, and Other Drugs. Out of which, Penicillin and Combinations Segment is expected to dominate the urinary tract infection market during the forecast period and this is attributed to the increase in prevalence of urinary tract infections (UTIs). Most penicillins are widely being used directly and in combinations for treating urinary tract infections (UTIs).
Quinolones Segment is also expected to witness significant growth in the near future and this is owing to the increase in demand for novel UTI therapeutic. Quinolones represent a natural alternative to trimethoprim-sulfamethoxazole for treating urinary tract infections (UTIs) when resistance becomes a potential problem.
Based on Indication, the market is segmented into Complicated UTI, Uncomplicated UTI, and Other Indications. Out of which, Complicated UTI Segment is expected to dominate the market during the forecast period and this is attributed to the rise in prevalence of complicated UTI across the globe. A complicated urinary tract infection (UTI) is a term to describe a UTI that doesn't respond to traditional treatments.
Uncomplicated UTI Segment is also expected to witness significant growth in the near future and this is due to rise in incidence of uncomplicated UTI. A UTI is classified as uncomplicated if there are no functional and/or anatomical anomalies in the urinary tract, no renal functional impairment, and no concomitant disease that would promote the UTI.
Global Urinary Tract Infection Therapeutic Market: Key Developments
On January 23, 2024, Locus Biosciences, Inc., a clinical-stage biotechnology company, announced the release of USD 23.9 million from the Biomedical Advanced Research and Development Authority (BARDA), a part of the Administration for Strategic Preparedness and Response within the U.S. Department of Health and Human Services, to continue the development of Locus Biosciences, Inc.'s Clustered regularly interspaced short palindromic repeats (CRISPR)-enhanced bacteriophage therapy, LBP-EC01, for treating urinary tract infections (UTIs) caused by drug-resistant Escherichia coli.
In April 2023, GSK plc, a multinational pharmaceutical and biotechnology company, reported positive results from the pivotal EAGLE-2 and EAGLE-3 phase III trials of gepotidacin, an investigational, first-in-class oral antibiotic with a novel mechanism of action for uncomplicated urinary tract infections (uUTI) in female adults and adolescents. The findings were presented in an oral presentation at the European Congress of Clinical Microbiology and Infectious Diseases (ECCMID) in Copenhagen, Denmark.
In August 2021, Bayer AG, a Germany-based multinational pharmaceutical and biotechnology company, announced the acquisition of Vividion Therapeutics (Vividion), a U.S.-headquartered biopharmaceutical company using novel discovery technologies to unlock high value, traditionally undruggable targets with precision therapeutics.
In October 2020, Bayer AG, acquired Asklepios BioPharmaceutical, Inc., a gene therapy company. Asklepios BioPharmaceutical, Inc.’s portfolio includes investigational pre-clinical and clinical stage candidates for the treatment of central nervous system, neuromuscular, and cardiovascular and metabolic diseases.
In January 2019, AstraZeneca, a global, science-led biopharmaceutical business, completed the acquisition of Shire, a biopharmaceutical company, bringing together Takeda Pharmaceutical Company Limited, a global biopharmaceutical company and Shire's complementary positions in gastrointestinal and neuroscience to establish leading positions in rare diseases complement Takeda's previously existing strength in oncology and focused efforts in vaccines.
Global Urinary Tract Infection Therapeutic Market: Key Companies Insights
The global urinary tract infection therapeutic market is highly competitive. This is attributed to the increase in prevalence of urinary tract infection and growing geriatric population, as a result, players in the market are focusing on launching novel products in the market.
Some of the key players in the global urinary tract infection therapeutic market are Pfizer, Novartis AG, Cipla Inc., Bayer AG, AstraZeneca, Shionogi & Co. Ltd., and GlaxoSmithKline PLC, among others.
*Definition: A urinary tract infection (UTI) is an infection in any part of the urinary system. The urinary system includes the kidneys, ureters, bladder, and urethra. Women are at greater risk of developing a urinary tract infection compared to men. Antibiotics usually are the first treatment for urinary tract infections.
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients