Asia Pacific Infertility Gonadotropins Market Size and Forecast: 2026-2033
The Asia Pacific infertility gonadotropins market is expected to grow from USD 1,198.2 Mn in 2026 to USD 2,917.6 Mn by 2033, registering a compound annual growth rate (CAGR) of 13.6% from 2026 to 2033. The Asia Pacific infertility gonadotropins market is fueled by the growing prevalence of infertility.
According to Chinese Center for Disease Control and Prevention, 15% of couples of reproductive ages worldwide experience infertility, with prevalence rates as high as 30% in certain populations. This trend is increasingly reflected across high-burden regions such as Asia Pacific, where delayed parenthood and lifestyle factors are contributing to rising infertility rates. (Source: Chinese Center for Disease Control and Prevention)
Additionally, according to estimates of the Indian Council of Medical Research, about 10% to 15% of the married couples in Asia Pacific suffer from infertility, resulting in a large number of patients who require treatment. (Source: Indian Council of Medical Research)
Key Takeaways of the Asia Pacific Infertility Gonadotropins Market
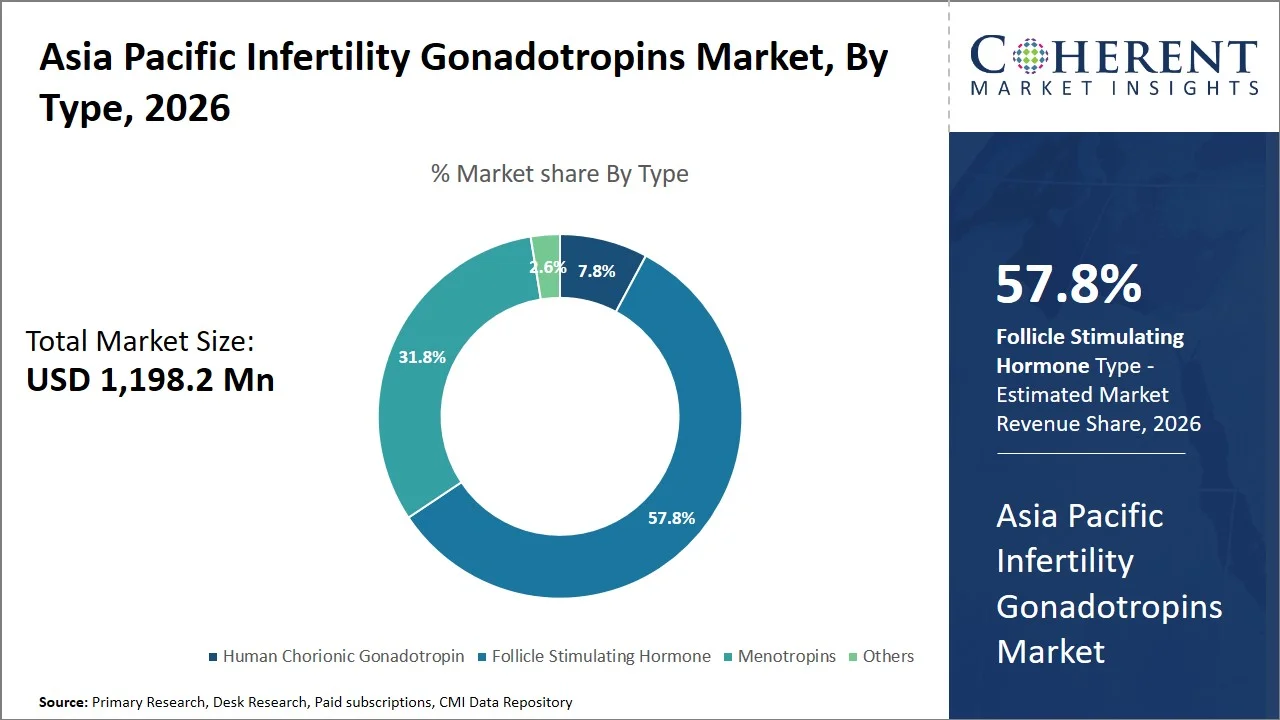
- Follicle stimulating hormone is projected to hold 57.8% of the Asia Pacific infertility gonadotropins market share in 2026, making it the dominant type segment, since it is compulsory during controlled ovarian stimulation treatment for all cycles in accordance with assisted reproductive technology (ART) treatment protocols from the Indian Council of Medical Research. In comparison with human chorionic gonadotropin, and menotropins, follicle stimulating hormone (FSH) treatment spans through the entire stimulation period (8-12 days per cycle), resulting in substantially greater volume and market share per cycle. (Source: Indian Council of Medical Research)
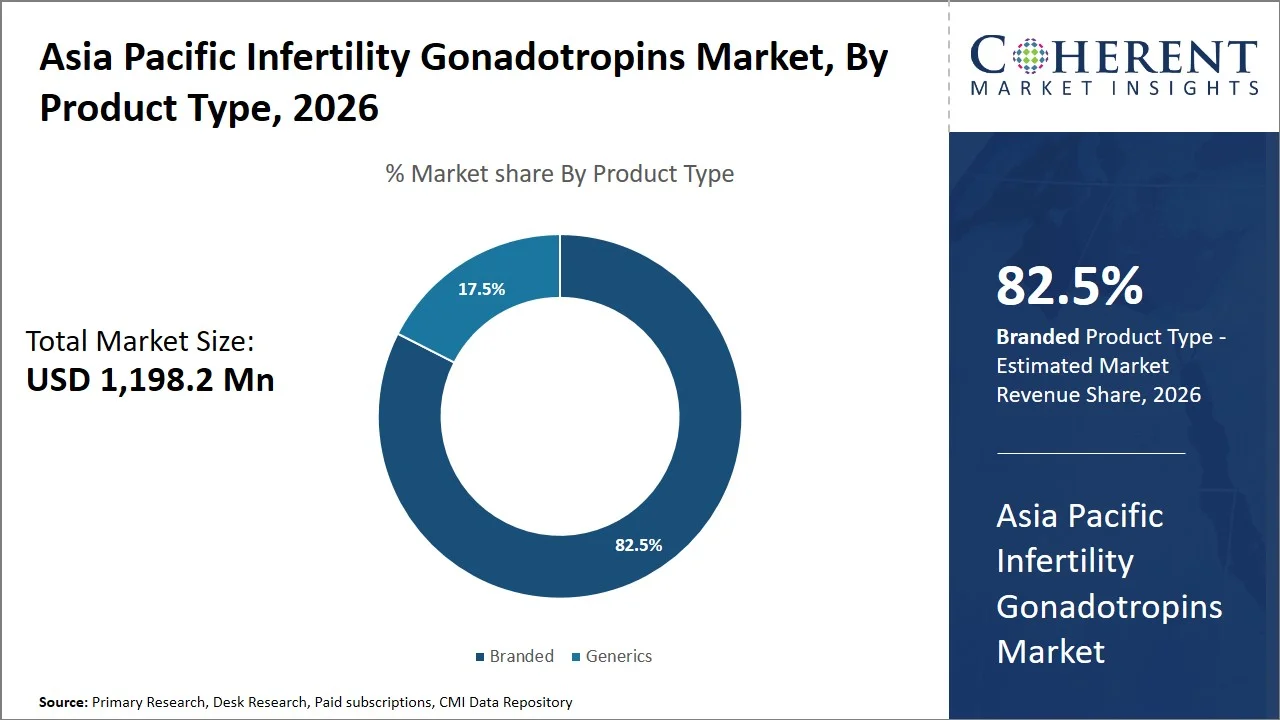
- Branded is projected to hold 82.5% of the Asia Pacific infertility gonadotropins market share in 2026, making it the dominant product type segment. Products in branded form predominate in view of the rigorous regulatory control (such as the National Medical Products Administration (NMPA) in China, Central Drugs Standard Control Organization (CDSCO) in India, and Pharmaceuticals and Medical Devices Agency (PMDA) in Japan), which stipulates the necessity of proving comparability between similar biologics regarding quality, safety, and efficacy. Due to the great sensitivity of IVF procedure to drug consistency, preference is given to the established brand with batch reliability over an unbranded product, supported by evidence that recombinant gonadotropins are preferred due to improved purity and batch-to-batch consistency compared to older preparations, as highlighted in a review published in Endocrine Reviews. (Source: Endocrine Reviews)
- Subcutaneous is projected to hold 65.5% of the Asia Pacific infertility gonadotropins market share in 2026, making it the dominant route of administration segment. The subcutaneous route prevails in view of the growing trend among ART protocols towards outpatient self-administration of multiple doses during a number of treatment cycles.
- Fertility Preservation (Egg Freezing) in Urban Working Women: Increased labor force participation and deferred parenthood have resulted in an increased demand for elective egg banking. This will open up a niche market for gonadotropins, as controlled ovarian hyperstimulation forms the basis of egg retrieval, thereby increasing its usage from merely being limited to infertility treatment.
- Expansion of Government-Regulated ART Clinics in Tier 2/3 Cities: The governments within the Asia Pacific region are increasingly favoring the formation of ART clinics in Tier 2 and Tier 3 cities, improving access to fertility treatments beyond metropolitan areas. Increased penetration of ART has further led to a rise in demand for gonadotropins.
Why Does the Follicle Stimulating Hormone Segment Dominate the Asia Pacific Infertility Gonadotropins Market?
Follicle stimulating hormone is projected to hold a market share of 57.8% in 2026, owing to their main use in stimulating the ovaries during ART cycles that are becoming more widespread across Asia Pacific. Based on the report published by the National Assisted Reproductive Technology (ART) Registry, ICMR, ovarian stimulation using follicle stimulating hormone (FSH) constitutes the initial process during IVF treatments used in all registered clinics, ensuring continued high clinical use of FSH. In addition, the passing of the ART Act by the Ministry of Health and Family Welfare provides for uniform treatment guidelines across all ART clinics that are required to conduct controlled ovarian stimulation. The increase in ART clinic numbers and registered ART treatment procedures ensures sustained demand for FSH as the drug of choice. (Source: National Assisted Reproductive Technology (ART))
Why is Branded the Most Preferred Product Type in the Asia Pacific Infertility Gonadotropins Market?
To learn more about this report, Download Free Sample
Branded is projected to hold 82.5% of the market share in 2026, owing to strict government policies (such as India’s Assisted Reproductive Technology (Regulation) Act, 2021; China’s NMPA regulatory framework for biologics; and Japan’s Pharmaceuticals and Medical Devices Act (PMD Act)), clinical sensitivity related to infertility treatment, and guaranteed effectiveness in assisted reproduction technology (ART) cycles. Under the guidelines laid down in the ART Act enacted by the Ministry of Health & Family Welfare, all ART centers need to abide by certain treatment guidelines and maintain drug traceability in treatment procedures. (Source: Ministry of Health & Family Welfare)
Furthermore, the Central Drugs Standard Control Organization (CDSCO) in India, along with counterparts like National Medical Products Administration (NMPA) and Pharmaceuticals and Medical Devices Agency (PMDA) has established stringent quality control standards for biological products, including gonadotropins, because even small changes in the drug's composition can affect how well the treatment works. For example, according to India's "Guidelines on Similar Biologics" (2016 amendment), manufacturers must demonstrate that their product is comparable to the original drug through thorough analysis and clinical testing, focusing on quality, safety, and effectiveness, to ensure there are no important differences in how the two drugs work. (Source: Central Drugs Standard Control Organization (CDSCO)) Given the significance of ovarian stimulation in achieving a successful IVF cycle rate, doctors usually choose branded drugs after thorough testing because of their batch-to-batch consistency, clinical efficacy, and negligible immunogenicity as compared to generic drugs.
Subcutaneous Segment Dominates the Asia Pacific Infertility Gonadotropins Market
The subcutaneous segment is expected to hold 65.5% of the Asia Pacific infertility gonadotropins market share in 2026, because of better patient convenience, easier self-injection, and clinical efficacy similar to or superior to that of the other methods of drug administration. Subcutaneous injection of gonadotropins are more convenient from a clinical perspective because this route of administration imposes less physical and mental stress on patients owing to self-injection without the need for clinic visits. For instance, according to global evidence from clinical settings, there is a pronounced preference for pen-based gonadotropin therapy owing to greater patient compliance and usability due to its decreased complexity and convenience. This preference is gradually becoming more common in Asian Pacific markets owing to increased adoption of self-administered assisted reproductive technology (ART). (Source: ResearchGate)
Furthermore, scientific studies indicate that giving hormones under the skin leads to similar, or even higher, hormone levels in the blood and follicles compared to injecting them into the muscle. This confirms that gonadotropins work well in in-vitro fertilization (IVF) procedures. For example, in October 2024, study published by ResearchGate titled “Intramuscular (IM) and Subcutaneous (SC) Administration of Gonadotropins Yield Similar In-Vitro Fertilization (IVF) Outcomes in a Propensity-Matched Cohort” indicated that both methods yielded equivalent levels of ovarian reaction and success rate during in-vitro fertilization, with comparable rates of egg harvest, underscoring the practicality of the subcutaneous route for administering the medication. (Source: ResearchGate)
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
China Strengthens Biologics Registration Framework (April, 2025) |
|
|
Regulatory Shift: Implementation of ART Act & National Registry (October 2025) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
(Source: Cisema, Ministry of Health & Family Welfare)
Market Dynamics
To learn more about this report, Download Free Sample
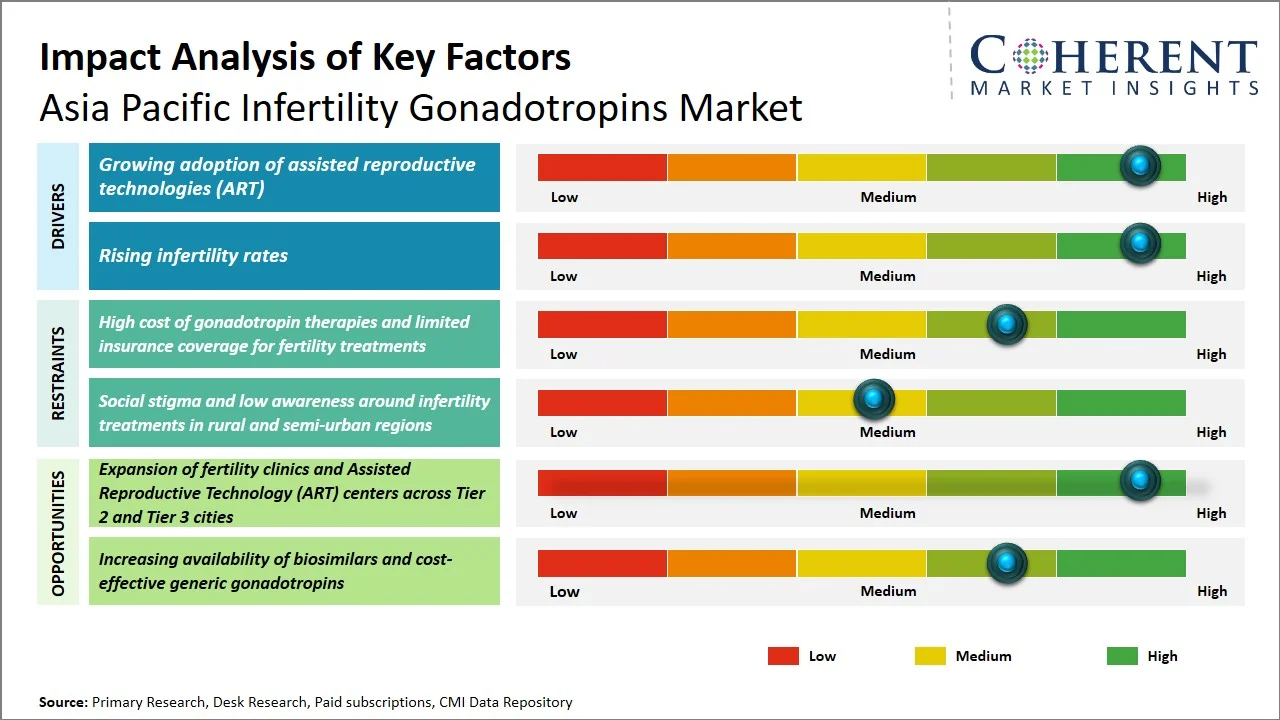
Market Drivers
- Growing Adoption of Assisted Reproductive Technologies (ART): The ARTs techniques have been widely used in Asia Pacific because of the rise in cases of infertility issues and better availability of ARTs in both metro and non-metro cities. According to the International Committee for Monitoring Assisted Reproductive Technologies (ICMART), Asia accounts for approximately half of global ART cycles, indicating that the region performs an estimated 1.5–1.8 million IVF procedures annually. (Source: International Committee for Monitoring Assisted Reproductive Technologies) Furthermore, the growth in fertility centers, increasing knowledge about reproductive health, and favorable regulations (such as India’s Assisted Reproductive Technology (Regulation) Act, 2021; China’s National Health Commission ART regulations; and Japan’s insurance coverage expansion for IVF under its national health system) are fueling the adoption of Assisted Reproductive Technologies (ART), thus boosting the demand for gonadotropin in ovarian stimulation treatment procedures.
- Rising Infertility Rates: Infertility rates in Asia Pacific are climbing, fueled by delayed parenthood, shifting lifestyles, and a rise in reproductive health issues. In October 2024, study in the Indian Journal of Community Medicine found that roughly 15–20 million couples in India are grappling with infertility. The prevalence varies considerably, from 3.9% to 16.8%, depending on the region. (Source: Indian Journal of Community Medicine) Furthermore, a 2023 study available on PubMed Central reports a notable rise in secondary infertility, reaching approximately 28.6%, indicating worsening reproductive health trends. (Source: PubMed Central) An expansion in the patient base indicate that more people require medical attention, leading to an increase in the number of gonadotropins required for both ART and ovulation induction treatments.
Emerging Trends
- Rising Adoption of Recombinant and Biosimilar Gonadotropins: There is an increasing inclination toward recombinant gonadotropins due to their higher efficiency and efficacy compared with the urine-derived products. Meanwhile, biosimilar drugs are being used more frequently in Asia Pacific where cost considerations play a significant role, resulting in higher volumes of gonadotropins usage.
- Integration of Personalized Fertility Treatments and Digital Monitoring: Data-driven practices, treatment planning based on artificial intelligence, and digital monitoring techniques have been gaining popularity among fertility clinics for ovarian stimulation therapy. This helps to improve success rates as well as patient compliance, leading to higher volumes for gonadotropin therapy through assisted reproduction technologies (ART).
Key Regulatory Framework Governing the Asia Pacific Infertility Gonadotropins Market
|
Country
|
Key Regulatory Authority |
Regulatory Framework |
Key Focus Area |
|
Japan |
Pharmaceuticals and Medical Devices Agency (PMDA) |
Pharmaceuticals and Medical Devices Act (PMD Act) |
Approval and lifecycle management of biologics, stringent clinical evaluation, pharmacovigilance |
|
China |
National Medical Products Administration (NMPA) |
Drug Administration Law & Biologics Registration Regulations |
GMP compliance, biologics approval, batch consistency, post-market surveillance |
|
ASEAN |
ASEAN Consultative Committee for Standards and Quality (ACCSQ) – Pharmaceutical Product Working Group |
ASEAN Common Technical Dossier (ACTD) & ASEAN Common Technical Requirements (ACTR) |
Harmonization of drug registration, quality, safety, and efficacy standards across member states |
|
India |
Central Drugs Standard Control Organization (CDSCO) |
Assisted Reproductive Technology (Regulation) Act, 2021 & Guidelines on Similar Biologics |
ART clinic regulation, biologics approval, comparability requirements, safety and efficacy standards |
|
Australia |
Therapeutic Goods Administration (TGA) |
Therapeutic Goods Act, 1989 & Australian Regulatory Guidelines for Biologicals (ARGB) |
Registration of biologics, quality control, risk-based classification, post-market monitoring |
|
Rest of Asia Pacific |
Various national regulatory bodies (e.g., MFDS – South Korea, HSA – Singapore) |
Country-specific biologics and ART regulations |
Drug approval, quality assurance, import controls, and clinical compliance |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
How is the expansion of fertility clinics and assisted reproductive technology (ART) centers across Tier 2 and Tier 3 cities creating new growth opportunities in the Asia Pacific infertility gonadotropins market?
The surge of fertility clinics and assisted reproductive technology (ART) centers into Tier 2 and Tier 3 regions, away from just the big cities, is boosting the demand for gonadotropins in the Asia Pacific infertility gonadotropins market by making advanced reproductive treatments much more accessible. As per the Government of India’s National ART & Surrogacy Registry, there are over 3,138 registered ART clinics in India, reflecting the rapid expansion of fertility infrastructure across the country. (Source: National ART & Surrogacy Registry) Furthermore, a report provided by National Library of Medicine highlights that the number of assisted reproductive technology centers (ART) in India jumped from 500 in 2010 to over 1,500 by 2019 showing an upward trajectory. (Source: National Library of Medicine)
Further, there is an increasing use of the hub-and-spoke concept in fertility chains, which facilitates consultation and diagnosis locally, with advanced procedures centralized. This geographical decentralization translates to higher numbers of IVF cycles, which in turn leads to the increased consumption of gonadotropins used in ovarian stimulation procedures, especially in the context of high penetration levels in semi-urban and developing markets in the Asia Pacific region.
Market Players, Key Development, and Competitive Intelligence

To learn more about this report, Download Free Sample
Key Developments
- In May 2025, Ferring Pharmaceuticals shared findings from its IRIS clinical study conducted in India. The study assessed the use of personalized dosing of recombinant FSH (follitropin delta) in IVF. The results showed better ovarian response and pregnancy rates, bolstering the case for personalized gonadotropin therapies. This news further solidifies the place of advanced fertility treatments within the India infertility gonadotropins market.
- In July 2024, Mankind Pharma revealed its plans to buy Bharat Serums and Vaccines Limited. The deal, valued at roughly USD 1.6 billion, was aimed at broadening Mankind Pharma's reach in women's health and fertility care, specifically in the area of gonadotropins. This move significantly bolsters Mankind Pharma's offerings in infertility treatments, setting the stage for expansion within the fertility sector.
Competitive Landscape
The Asia Pacific infertility gonadotropins market is highly competitive with global pharmaceutical companies competing against regional biosimilar companies and specialist fertility companies. The firms are emphasizing recombinant gonadotropin development and biosimilars to strike a balance between effectiveness and cost-effectiveness. Effective collaboration with IVF facilities and assisted reproductive technology (ART) centers is essential for enhancing the penetration and patient access, especially in developing cities. Other aspects that are influencing competitive dynamics include regulatory compliance and product differentiation to earn the trust of clinicians in a clinical setting. Key focus areas include:
- Recombinant Gonadotropins & Biosimilar Development
- Strategic Partnerships with In Vitro Fertilization (IVF) Clinics & assisted reproductive technology (ART) Centers
- Regulatory Compliance & Quality Differentiation
- Expansion into Tier 2 & Tier 3 Markets
- Cost Optimization & Patient Support Programs
Market Report Scope
Asia Pacific Infertility Gonadotropins Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1,198.2 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 13.6% | 2033 Value Projection: | USD 2,917.6 Mn |
| Geographies covered: |
Japan, China, ASEAN, India, Australia, and Rest of Asia Pacific |
||
| Segments covered: |
|
||
| Companies covered: |
Merck KGaA, Ferring Pharmaceuticals, Organon & Co., IBSA Institut Biochimique SA, Bharat Serums and Vaccines Limited, Intas Pharmaceuticals Ltd., Sun Pharmaceutical Industries Ltd., Zydus Lifesciences Ltd., Lupin Limited, and Dr. Reddy's Laboratories Ltd. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Analyst Opinion (Expert Opinion)
- The Asia Pacific infertility gonadotropins market is well-positioned for strong sustained growth, supported by growing cases of infertility, delayed parenthood, and greater acceptance of assisted reproductive technology (ART). It is anticipated that the industry will gravitate towards value-added and scientifically proven treatments such as recombinant gonadotropins due to increased regulation and the emphasis on efficacy of treatment among physicians. On the other hand, biosimilar penetration in this market will gradually rise due to their cost-effectiveness; however, uptake will be somewhat selective because of the need for consistency of the drugs used in IVF procedures.
- In terms of opportunities, the most promising area of growth can be seen in the branded recombinant gonadotropins market in high growth countries like India and China, due to the rapid growth of volumes in the IVF procedure because of the increasing presence of fertility clinics and greater affordability in tier 2 and 3 cities. Furthermore, Southeast Asian (ASEAN) region will provide a promising opportunity for biosimilar products due to their cost-effectiveness, due to growing middle classes and affordability. Countries like Indonesia, Vietnam, and Thailand have experienced growing acceptance of Assisted Reproductive Technologies (ART).
- It is essential for market players to take into consideration cost optimization, engaging in localized manufacturing, and developing relationships with fertility clinics and hospitals. It will be essential to develop biosimilars at lower costs, expanding their sales networks to include underdeveloped regions, as well as organizing training for physicians. It is also crucial for firms to focus on patient-oriented strategies including payment solutions, communication platforms, and stigma-reduction activities. By employing evidence-based drug regimens and developing connections with ART clinics, firms can create value in a fiercely competitive market setting.
Market Segmentation
- Type Insights (Revenue, USD Mn, 2021 - 2033)
- Human Chorionic Gonadotropin
- Follicle Stimulating Hormone
- Menotropins
- Others
- Product Type Insights (Revenue, USD Mn, 2021 - 2033)
- Branded
- Generics
- Route of Administration Insights (Revenue, USD Mn, 2021 - 2033)
- Intramuscular
- Subcutaneous
- Application Insights (Revenue, USD Mn, 2021 - 2033)
- Female Infertility
- Male Infertility
- Type of Infertility Insights (Revenue, USD Mn, 2021 - 2033)
- Primary Infertility
- Secondary Infertility
- Age Group Insights (Revenue, USD Mn, 2021 - 2033)
- 18–25
- 26–30
- 31–35
- 36 and Above
- Distribution Channel Insights (Revenue, USD Mn, 2021 - 2033)
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
- Government Supply / Public Health Programs
- Country Insights ( Revenue, USD Mn, 2021 - 2033)
- Japan
- China
- ASEAN
- India
- Australia
- Rest of Asia Pacific
- Key Players Insights
- Merck KGaA
- Ferring Pharmaceuticals
- Organon & Co.
- IBSA Institut Biochimique SA
- Bharat Serums and Vaccines Limited
- Intas Pharmaceuticals Ltd.
- Sun Pharmaceutical Industries Ltd.
- Zydus Lifesciences Ltd.
- Lupin Limited
- Reddy's Laboratories Ltd.
Sources
Primary Research Interviews
- Fertility specialists, embryologists, and IVF clinicians involved in ART procedures and ovarian stimulation protocols
- Pharmaceutical executives and product managers from gonadotropin manufacturers
- Endocrinologists and gynecologists specializing in infertility treatment across hospitals and fertility clinics
- CROs and regulatory consultants involved in biologics approval and fertility drug clinical trials
Stakeholders
- Gonadotropin manufacturers & fertility drug providers (branded and biosimilar players)
- End-use Sectors:
- Hospitals and Specialty Fertility Clinics
- Ambulatory Surgical Centers (ASCs)
- Dedicated IVF Centers and Reproductive Health Institutes
- Regulatory & Health Bodies: National Medical Products Administration (NMPA) – biologics approval and ART-related drug regulations in China, Central Drugs Standard Control Organization (CDSCO) – drug approval and ART Act compliance in India, Pharmaceuticals and Medical Devices Agency (PMDA) – clinical evaluation and approval of fertility drugs in Japan, Therapeutic Goods Administration (TGA) – biologics regulation in Australia, World Health Organization (WHO) – infertility prevalence and ART guidelines
Databases
- International Committee for Monitoring Assisted Reproductive Technologies (ICMART) – global and regional ART data
- WHO Global Health Observatory – reproductive health and infertility indicators
- OECD Health Statistics – healthcare infrastructure and fertility treatment access
Magazines
- Fertility and Sterility Reports – ART trends and clinical developments
- BioSpectrum Asia – pharmaceutical and biotech developments in APAC
- PharmaBiz (India) – domestic pharma and fertility drug market updates
- Nikkei Asia – healthcare and fertility market investments
Journals
- Human Reproduction (Oxford Academic)
- Reproductive BioMedicine Online
- Journal of Assisted Reproduction and Genetics
- BMJ Open (fertility and ART-related studies)
Newspapers
- Financial Times – healthcare investments and fertility clinic expansion
- The Wall Street Journal – global pharma and ART developments
- The Economic Times (India) – fertility industry and healthcare policy updates
- China Daily – healthcare reforms and fertility policies in China
Associations
- International Federation of Fertility Societies (IFFS)
- Asia Pacific Initiative on Reproduction (ASPIRE)
- Indian Society for Assisted Reproduction (ISAR)
- Japan Society of Obstetrics and Gynecology (JSOG)
Public Domain Sources
- Ministry of Health & Family Welfare (India) – ART Act, fertility clinic regulations
- National Health Commission (China) – reproductive health policies
- Australian Government Department of Health – ART and IVF guidelines
- Centers for Disease Control and Prevention (CDC) – ART surveillance data (global benchmarking)
Proprietary Elements
- CMI Data Analytics Tool
- Proprietary CMI Existing Repository of information for last 10 years
Share
Share
About Author
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients