Global Baby Vitamin D Drops Market Size and Forecast – 2026 to 2033
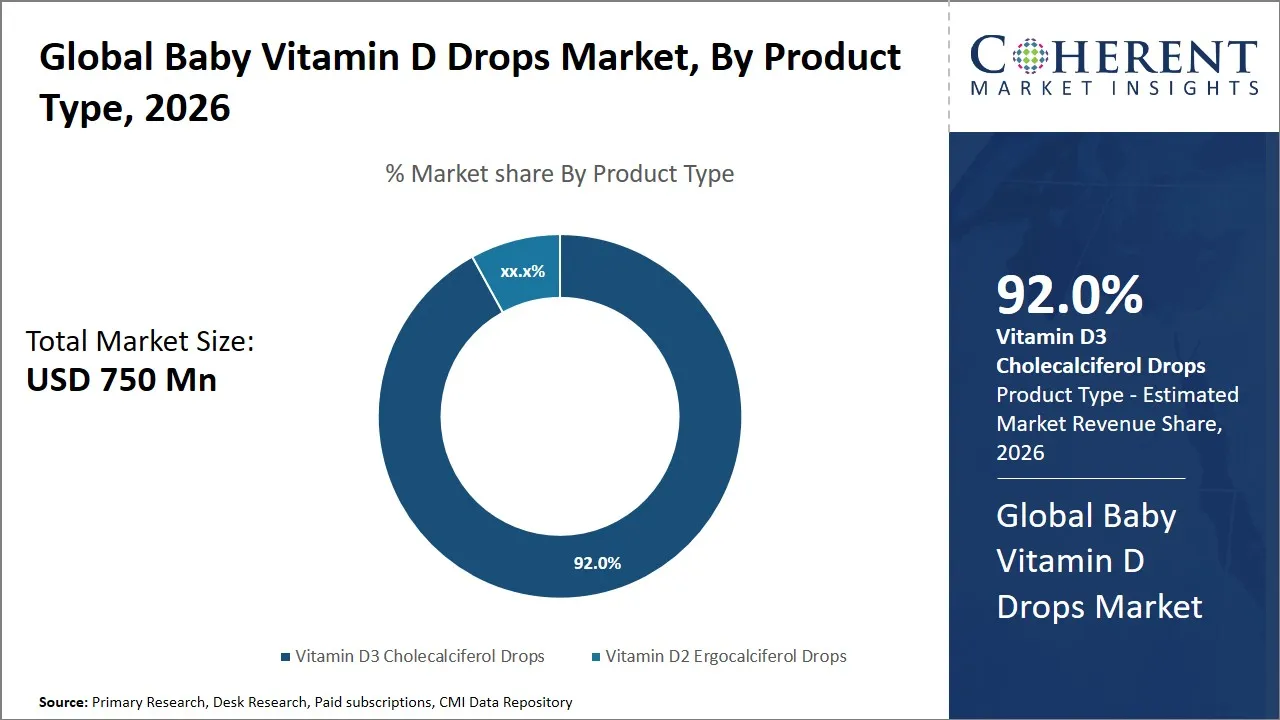
The global baby vitamin D drops market is estimated to be valued at USD 750 Mn in 2026 and is expected to reach USD 1,120 Mn by 2033, exhibiting a compound annual growth rate (CAGR) of 5.9% from 2026 to 2033. This steady rise is a result of enhanced awareness among parents about the role of vitamin D in preventing deficiencies in infants, in addition to increased healthcare expenditures and government initiatives.
Key Takeaways of the Global Baby Vitamin D Drops Market
- The vitamin D3 cholecalciferol drops segment is expected to dominate the global baby vitamin D drops market by product type, accounting for approximately 92.0% of the total market share in 2026.
- The oil-based drops segment is expected to dominate the global baby vitamin D drops market by formulation base, accounting for approximately 64.0% of the total market share in 2026.
- The single vitamin D drops (pure vitamin D) segment is expected to dominate the global baby vitamin D drops market by composition, accounting for approximately 72.0% of the total market share in 2026.
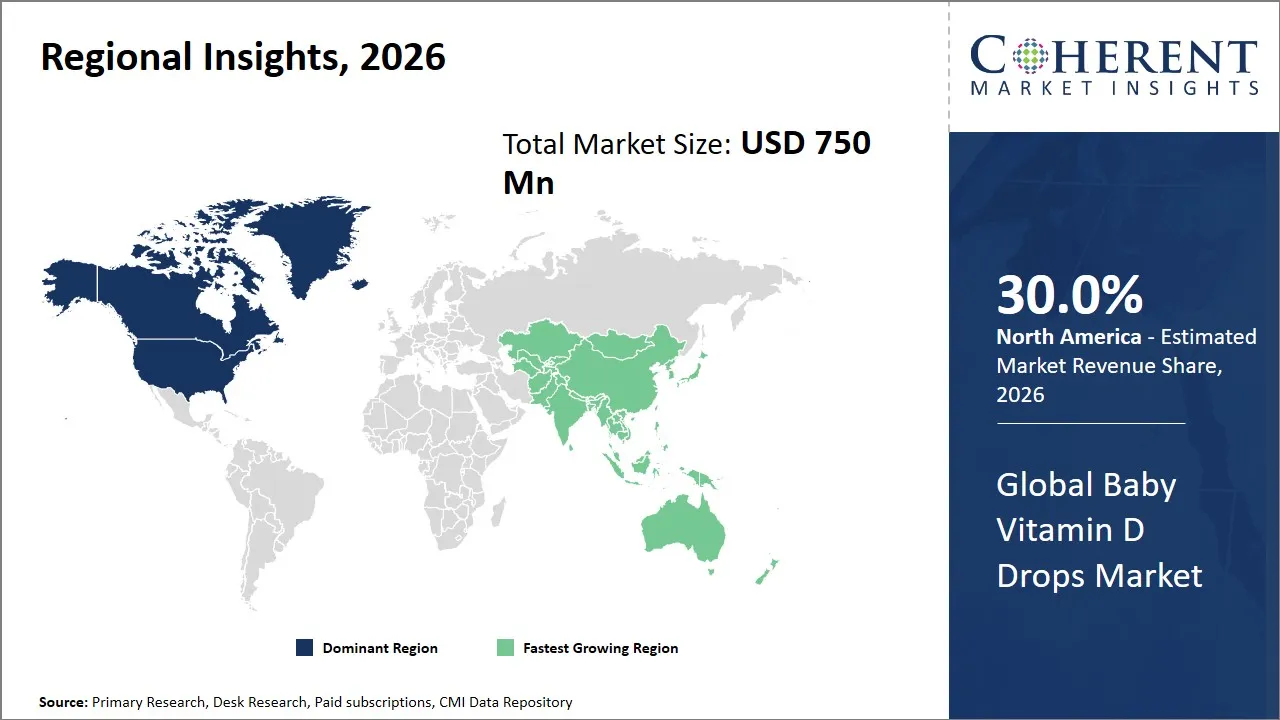
- North America is expected to dominate the global baby vitamin D drops market in 2026, accounting for approximately 30.0% of the total market share.
- Asia Pacific is anticipated to witness the fastest growth, holding a market share of 22.0% in 2026.
Market Overview
- Baby vitamin D drops are a type of dietary supplement in liquid form, used to ensure that infants receive adequate vitamin D, which is very important for calcium absorption, bone development, and overall growth. Since infants, especially those who are breastfed, may not receive adequate vitamin D from natural sources, this has become a very important segment in infant nutrition.
- The market is mainly driven by increasing awareness among parents and caregivers about vitamin D deficiency in infants. Deficiency of vitamin D in early developmental stages may lead to bone-related problems, including rickets, and thus promotes vitamin D supplements in daily care of infants.
- In addition, changing lifestyles such as reduced exposure to the sun and increased urbanization have also reduced the levels of naturally produced vitamin D in infants. As such, the pediatric recommendations for routine supplementation of vitamin D have been well accepted and are likely to maintain the market demand for baby vitamin D drops in the developed and emerging markets.
- Furthermore, the development of tasteless and easy-to-administer liquid drops with controlled dosing has improved the convenience of the product. In the future, the focus on preventive healthcare and baby nutrition is likely to provide opportunities for the company to develop more products.
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
Increasing Research on Infant Nutrition & Micronutrient Deficiency |
|
|
Advancements in Pediatric Supplement Formulations |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Why Does the Vitamin D3 Cholecalciferol Drops Segment Dominate the Global Baby Vitamin D Drops Market in 2026?
The vitamin D3 cholecalciferol drops segment is anticipated to hold a market share of 92.0% in 2026, owing to their higher bioavailability of the product and the clinical preference for the same. This is because vitamin D3 is the natural form of the vitamin that is naturally produced in the human body. It has higher absorption and retention in the body in comparison to vitamin D2. This makes it the first choice of pediatricians for the development of bones in babies and calcium absorption and immune system development in the process. Clinical studies have proven that vitamin D3 is more efficient in the prevention of vitamin D deficiency in babies who are exposed less to the sun.
Moreover, a rise in recommendations by healthcare professionals and pediatric organizations has bolstered the case for cholecalciferol drops to become the norm for infant supplements. Increasing parental knowledge and support through guidelines for neonatal care have fueled the need for vitamin D3 supplements based on their established safety and efficacy in preventing rickets and promoting early child development. This high level of support from healthcare professionals and trust by consumers has ensured that vitamin D3 drops remain at the forefront of this market.
For instance, the American Academy of Pediatrics suggests that all infants should be given 400 IU of vitamin D daily in the form of vitamin D3 or cholecalciferol, as it is more effective in maintaining the levels of vitamin D and preventing conditions associated with vitamin D deficiency.
Why Does the Oil-Based Drops Segment Dominate the Global Baby Vitamin D Drops Market in 2026?
The oil-based drops segment is projected to hold a market share of 64.0% in 2026, primarily due to the high compatibility of the product with the fat-soluble nature of vitamin D, thereby offering the best stability, longer shelf life, and less degradation of the nutrient. Such drops provide the best absorption in the body of the infant, as fat always facilitates the assimilation of the nutrient in the most efficient manner.
Furthermore, the segment of oil-based drops has witnessed a higher preference among parents and healthcare practitioners owing to the ease of consumption and the availability of the product in the form of droppers, thereby facilitating the administration of the drops in the most precise manner. Along with that, the segment has also witnessed a higher preference owing to the presence of a well-established safety profile, thereby offering the best clean label with fewer additives and preservatives in the product.
The Single Vitamin D Drops (pure vitamin D) Segment Dominates the Global Baby Vitamin D Drops Market
The single vitamin D drops (pure vitamin D) segment is projected to hold a market share of 72.0% in 2026, primarily because of the increasing popularity of the use of exact supplements for infants. This allows the doctor to provide the exact amount of a specific vitamin that the infant may be lacking in their body. There exists a high possibility of nutrient imbalance if a multivitamin supplement is taken.
Moreover, a single vitamin D drop has the highest recommendation by pediatric and neonatal guidelines for the treatment of vitamin D deficiency and related conditions. These supplements are increasingly being used by parents as they are considered safe and are not likely to induce any kind of allergic reaction in the infant. They are also easy to administer and are in a user-friendly format, which makes them a major contributing factor in the high market share of this segment as it fulfills the needs of a parent who wants a safe and easy solution for their infant.
Regulatory Guidelines and Compliance Framework Shaping the Global Baby Vitamin D Drops Market
|
Region |
Regulatory Body |
Key Guidelines/Regulations |
|
North America |
Food and Drug Administration |
Vitamin D drops are regulated as dietary supplements, requiring strict adherence to Good Manufacturing Practices (GMP), accurate labeling, ingredient transparency, and safety validation. Additionally, the American Academy of Pediatrics recommends 400 IU/day for infants, which significantly influences product formulation, dosage standards, and marketing claims across the industry. |
|
Europe |
European Food Safety Authority |
The market is governed by the EU Food Supplements Directive, which mandates rigorous safety assessments, controlled nutrient limits, and substantiated health claims. EFSA establishes adequate intake levels for infants, while manufacturers must comply with strict labeling, traceability, and quality assurance standards to ensure consumer safety. |
|
Asia Pacific |
Food Safety and Standards Authority of India |
Baby vitamin D drops are classified under nutraceutical and health supplement regulations, requiring product approval, defined dosage thresholds, and compliance with labeling norms. Increasing regulatory scrutiny around infant nutrition is pushing manufacturers toward higher quality standards and improved transparency in formulations. |
|
Middle East & Africa (MEA) |
Regional authorities such as Saudi Food and Drug Authority and South African Health Products Regulatory Authority |
Regulations vary by country but generally classify vitamin D drops as dietary supplements or health products, requiring registration, ingredient approval, and labeling compliance. Governments are increasingly aligning with international standards (WHO/FAO), with rising focus on infant nutrition and deficiency prevention driving stricter oversight. |
|
Latin America |
Authorities such as ANVISA and COFEPRIS |
Products are regulated as food supplements or health products, requiring product registration, safety validation, and compliance with labeling and advertising standards. Regulatory frameworks are evolving to strengthen quality control and ensure safe infant nutrition products, supporting market formalization. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Regional Insights
To learn more about this report, Request Free Sample
North America Baby Vitamin D Drops Market Analysis and Trends
The North America region is projected to lead the market with a 30.0% share in 2026, owing to its dominance owing to the already established healthcare system and the level of awareness among consumers about the importance of nutrition for babies.
Moreover, the presence of major pharmaceutical and nutraceutical companies such as Abbott Laboratories, Pfizer, and Nestle adds value to the market with the level of research and development in the field. Government initiatives such as the mandatory guidelines for the supplementation of vitamin D in the U.S. and Canada also contribute to the market demand. In terms of the overall market size, North America has already taken the top spot in the market.
For instance, the Centers for Disease Control and Prevention suggest that breastfed and partially breastfed babies should receive 400 IU of vitamin D daily shortly after birth. This again underlines the importance of vitamin D supplementation and increases the demand for baby vitamin D drops.
Asia Pacific Baby Vitamin D Drops Market Analysis and Trends
Asia Pacific is anticipated to witness the fastest growth, holding a market share of 22.0% in 2026, fueled by factors like increased awareness of baby health and a rise in healthcare expenditure in emerging economies like India and China. In addition, factors like a growing population base of the middle class and increased levels of urbanization and a desire for supplements are also contributing factors to the growth rate of this region.
Major companies like DSM, ZYDUS Cadila, and Abbott India are found to be actively involved in localizing their products and making them more affordable in this region. In addition, factors like trade agreements and increased penetration of organized retail and e-commerce are also enhancing the growth rate of this region.
Baby Vitamin D Drops Market Outlook for Key Countries
How is the U.S. Helping in the Growth of the Baby Vitamin D Drops Market?
Baby vitamin D drops market in the U.S. has the added advantage of high standards in the services provided in the healthcare sector, as well as the awareness of children in the country regarding the prevention of vitamin D deficiency. The market in the country also has the added advantage of the quality standards set by the FDA, which generates a sense of trust in the minds of the consumers.
Pfizer and Garden of Life are some of the market players, which are significant contributors to the market in the country. The market players have a wide variety of products to cater to the needs of the consumers. The presence of health insurers is a positive sign for the market growth.
For instance, the U.S. Food and Drug Administration have strict regulations regarding Good Manufacturing Practices and labeling for dietary supplements, including vitamin D drops, to ensure their safety, quality, and consumers’ confidence in the product in the U.S.
How is China Helping in the Growth of the Baby Vitamin D Drops Market?
Baby vitamin D drops market in China has a huge market size with rapid urbanization and rising levels of disposable income, hence the emphasis on preventive infant nutrition. In addition, the China government has implemented various initiatives in the past that have improved the health of the pediatric population and the healthcare infrastructure in the country. This has enabled multinational corporations such as DSM and Abbott Laboratories to partner with local manufacturers in the production of baby food products that meet the taste preferences of the China population.
Key Drivers for the Growth of the Germany Baby Vitamin D Drops Market
Germany is the market leader in the European region in terms of the presence of a sound regulatory environment and the level of consumer confidence in healthcare products. Moreover, the market is also influenced by the European Union’s stringent regulations, which provide baby vitamin D supplements with the best quality and safety. Bayer and Merck are already established in the market and conduct clinical research and development of products to meet the needs of the population in specific age groups. The healthcare infrastructure and insurance also contribute to the regular demand for vitamin supplements by the population.
India Baby Vitamin D Drops Market Trends
The baby vitamin D drops market in India is witnessing rapid growth due to increased awareness about health and rising birth rates in urban centers. Government initiatives such as the National Nutrition Mission also help boost awareness about nutrient deficiencies among young children. Indian players like ZYDUS Cadila and multinational corporations such as Abbott India also contribute to this growth by creating cost-effective products that can cater to a wider audience. In addition to this, e-commerce growth is also contributing to the market's accessibility.
Role of Public Health Programs and Government-Led Supplementation Initiatives in Driving the Baby Vitamin D Drops Market
|
Region |
Program/Authority |
Key Initiatives & Market Impact |
|
North America |
Centers for Disease Control and Prevention / American Academy of Pediatrics |
Public health bodies recommend 400 IU/day vitamin D supplementation for infants, particularly those who are breastfed. While not directly reimbursed, these guidelines strongly influence pediatric prescriptions and parental purchasing behavior, driving consistent market demand. |
|
Europe |
National health systems aligned with European Food Safety Authority |
Several European countries implement government-supported infant supplementation programs, including free or subsidized vitamin D drops through healthcare providers. This ensures high adoption rates and establishes vitamin D supplementation as a routine part of neonatal care. |
|
Asia Pacific |
Ministry of Health and Family Welfare |
Infant nutrition programs and maternal-child health initiatives promote awareness of micronutrient deficiencies. Although direct distribution is limited, increasing integration of supplementation guidance into Anganwadi and pediatric care systems is supporting gradual market penetration. |
|
Middle East & Africa |
World Health Organization supported national programs |
Many countries implement WHO-supported child health programs targeting rickets prevention and micronutrient deficiencies. Free or subsidized supplementation through public healthcare facilities enhances accessibility and drives demand in underserved populations. |
|
Latin America |
National health agencies aligned with Pan American Health Organization |
Governments are increasingly focusing on maternal and child nutrition programs, promoting vitamin supplementation through public hospitals and awareness campaigns. This improves early adoption and supports long-term market growth. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Market Players, Key Developments, and Competitive Intelligence

To learn more about this report, Request Free Sample
Key Developments
- In August 2024, Perrigo voluntarily recalled a specific batch of infant formula because of higher-thanexpected Vitamin D levels. This event underscores the growing regulatory oversight and the vital need for accurate vitamin D dosing in all infant nutrition and supplementation products, including those delivered via drops.
- In February 2024, Nordic Naturals announced a voluntary recall of its Baby’s vitamin D3 liquid. The recall was prompted by a manufacturing problem that resulted in higher-than-expected levels of vitamin D. This situation underscores the vital need for precise dosage and adherence to regulations when it comes to infant supplements.
Top Strategies Followed by Global Baby Vitamin D Drops Market Players
|
Player Type |
Strategic Focus |
Examples |
|
Established Market Leaders |
Established Players are dedicated to growth through innovation, clinicals, and portfolio expansion by utilizing advanced formulation and supplement combinations. These players have a robust presence in the global market, invest significantly in R&D, and are dedicated to premiumization and brand trust to expand their presence in both developed and emerging markets. |
In July 2024, Ddrops received the “Supplement Product of the Year” award for its Baby Vitamin D3 drops, highlighting innovation in single-drop dosing and clean-label formulation. |
|
Mid-Level Players |
Mid-level players are dedicated to value-based positioning in the market by offering affordable, high-quality, and clean-label products. These players are dedicated to securing certifications, ecommerce growth, and strategic manufacturing partnerships to enhance their presence in the market. |
In March 2024, Mommy’s Bliss continued expansion of its organic vitamin D drops portfolio, emphasizing NSF-certified, allergen-free formulations tailored for infants. |
|
Small-Scale Players |
Small-scale players are dedicated to niche targeting through minimal-ingredient, high-bioavailability product formulations and innovative delivery formats such as single drop technology. These players are dedicated to D2C growth, premiumization, and product positioning to attract health-conscious consumers in the market. |
In April 2024, Nordic Naturals promoted its Baby’s Vitamin D3 product line with enhanced purity standards and third-party testing certifications, strengthening its premium positioning in infant nutrition. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Market Report Scope
Baby Vitamin D Drops Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 750 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 5.9% | 2033 Value Projection: | USD 1,120 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Ddrops Company, Carlson Laboratories, Mommy's Bliss, Zarbee's, ChildLife Essentials, Nature's Way, Nordic Naturals, Enfamil Mead Johnson, PediaSure Abbott Laboratories, and Garden of Life |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Baby Vitamin D Drops Market Dynamics
To learn more about this report, Request Free Sample
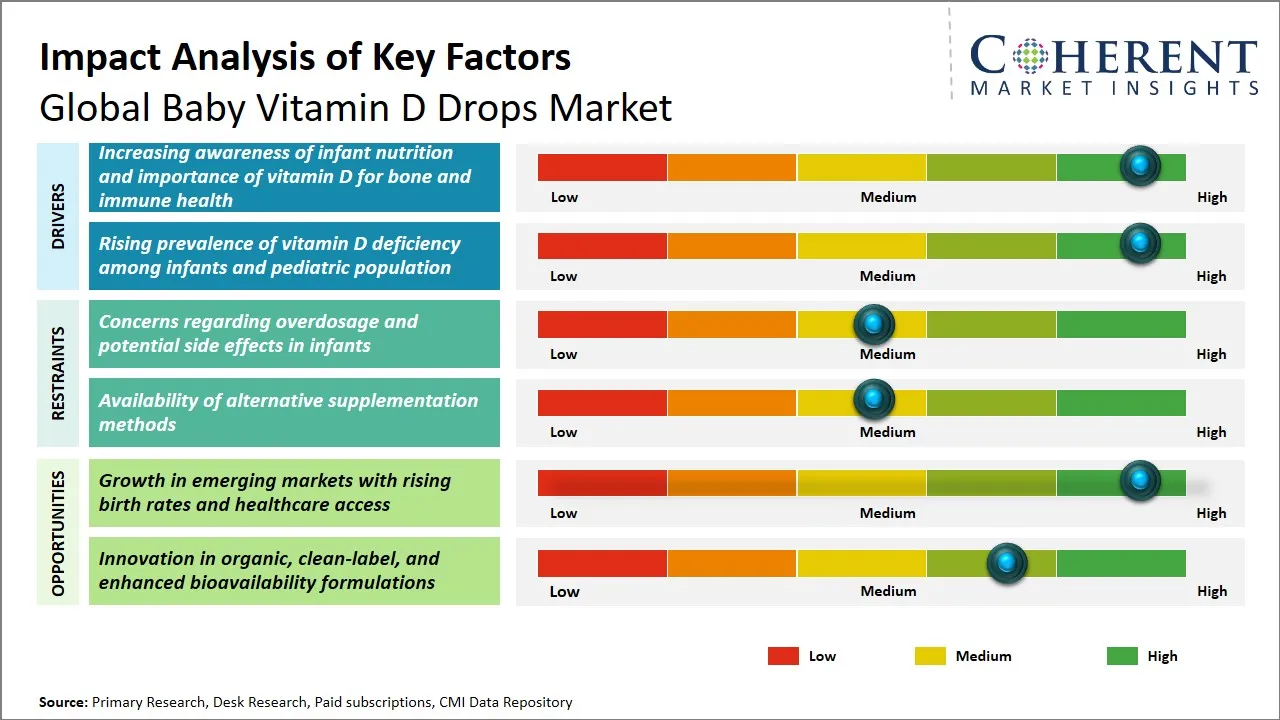
Baby Vitamin D Drops Market Driver - Rising Awareness of Infant Nutrition and Importance of Vitamin D for Bone and Immune Health
The rising awareness about the importance of baby nutrition has been a major contributing factor in the increased demand for baby vitamin D drops. This is because more and more people are becoming aware of the importance of vitamin D in the development of healthy bones and the strengthening of the immune system in babies. This is particularly true in the case of babies because they are more likely to suffer from vitamin D deficiency. This is because they are unable to get enough vitamin D through sun exposure and the food they eat.
As such, more and more people are becoming aware of the importance of baby vitamin D drops in the prevention of rickets in babies. This is further strengthened by the emphasis being placed on the importance of maintaining healthy levels of vitamin D in babies. This has made the product more acceptable and the preferred option in the market. In addition, the increased awareness of the importance of baby vitamin D drops are further being promoted through the increased information being disseminated through various channels.
For instance, the National Institutes of Health emphasizes that vitamin D has a vital role in the absorption of calcium, bone growth, and the immune system in infants, while a deficiency in the vitamin may result in rickets, thereby emphasizing the need for supplementation in infants.
Baby Vitamin D Drops Market Opportunity - Growth in Emerging Markets with Rising Birth Rates and Healthcare Access
The baby vitamin D drops market is likely to experience a boost in terms of market momentum with the entry of emerging markets such as the Asia Pacific, Latin America, and Africa. This is because the birth rate in these countries is higher than in other parts of the world. In addition, the healthcare infrastructure in these markets is improving continuously. Moreover, the government is launching initiatives aimed at reducing infant mortality rates and combating various nutrient deficiencies. This is further adding to the awareness of the importance of various nutrient supplements such as vitamin D drops in the baby vitamin D drops market.
In the same vein, the market is likely to boost because the retail sector is expanding continuously in the baby vitamin D drops market. In addition, the entry of online retail is likely to boost the baby vitamin D drops market in the coming years. This is because the online retail sector is expanding continuously in various markets such as the Asia Pacific, Latin America, and Africa.
For instance, in May 2023, UNICEF stated that the expansion of maternal and child health initiatives in the emerging regions was ongoing, with a focus on improving infant nutrition and reducing micronutrient deficiencies, thereby promoting the consumption of vitamin D drops supplements.
Analyst Opinion (Expert Opinion)
- The emerging market is providing tremendous volume-driven growth opportunities with an improvement in birth rates and an enormous base of underserved infants in which vitamin D supplementation is at a relatively lower penetration level. This is a big white space for manufacturers to gain more market share in consumers.
- Though recommendations from various health organizations such as the World Health Organization and Centers for Disease Control and Prevention are given, the adoption of infant vitamin D supplementation is not uniform.
- Improvement in healthcare infrastructure, an increase in pediatric consultations, and an exponential growth in pharmacy chains and e-commerce websites are making vitamin D supplementation reach the consumers in semi-urban and rural areas. This is further accelerating the market penetration.
- Manufacturers who are focusing on cost-effective strategies, cultural sensitivity in products, and tie-ups with healthcare professionals for campaigns will gain competitive advantage over others. This is because the market is shifting towards preventive healthcare rather than campaigns.
Market Segmentation
- Product Type Insights (Revenue, USD Mn, 2021 - 2033)
- Vitamin D3 Cholecalciferol Drops
- Vitamin D2 Ergocalciferol Drops
- Formulation Base Insights (Revenue, USD Mn, 2021 - 2033)
- Oil-based Drops
- Water-based Drops
- Composition Insights (Revenue, USD Mn, 2021 - 2033)
- Single Vitamin D Drops (pure vitamin D)
- Multivitamin Drops (Vitamin D with A C K etc.)
- Distribution Channel Insights (Revenue, USD Mn, 2021 - 2033)
- Pharmacies and Drug Stores
- Online Retail
- Supermarkets and Hypermarkets
- Pediatric Clinics and Hospitals
- Regional Insights (Revenue, USD Mn, 2021 - 2033)
- North America
- U.S.
- Canada
- Latin America
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Europe
- Germany
- U.K.
- Spain
- France
- Italy
- Russia
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Australia
- South Korea
- ASEAN
- Rest of Asia Pacific
- Middle East
- GCC Countries
- Israel
- Rest of Middle East
- Africa
- South Africa
- North Africa
- Central Africa
- North America
- Key Players Insights
- Ddrops Company
- Carlson Laboratories
- Mommy's Bliss
- Zarbee's
- ChildLife Essentials
- Nature's Way
- Nordic Naturals
- Enfamil Mead Johnson
- PediaSure Abbott Laboratories
- Garden of Life
Sources
Primary Research Interviews
Industry Stakeholders
- Pediatricians and neonatologists
- Infant nutrition specialists and dietitians
- Baby supplement manufacturers and product managers
- Regulatory affairs professionals (infant nutrition)
- Retail distributors and e-commerce category managers
End Users
- Parents and caregivers (infants 0–24 months)
- Hospital pharmacists
- Pediatric clinic professionals
- Maternity and neonatal care centers
Government and International Databases
- World Health Organization – Infant nutrition and micronutrient guidelines
- Centers for Disease Control and Prevention – Vitamin D recommendations for infants
- National Institutes of Health – Office of Dietary Supplements (Vitamin D fact sheets)
- European Food Safety Authority – Nutrient intake recommendations
- Food Safety and Standards Authority of India – Infant nutrition regulations
Trade Publications
- Nutrition Business Journal – Dietary supplements market insights
- Nutraceuticals World – Infant supplement product trends
- FoodNavigator – Pediatric nutrition innovations
- DairyReporter – Infant nutrition product developments
Academic Journals
- Pediatrics (American Academy of Pediatrics) – Infant vitamin D supplementation guidelines
- The American Journal of Clinical Nutrition – Studies on vitamin D deficiency in infants
- Journal of Pediatric Gastroenterology and Nutrition – Infant micronutrient research
- Nutrients (MDPI) – Clinical research on vitamin D bioavailability and absorption
- European Journal of Clinical Nutrition – Pediatric nutrition and supplementation studies
Reputable Newspapers
- The New York Times – Health Section – Infant nutrition and supplementation trends
- The Guardian – Science & Health – Public health coverage on vitamin deficiencies
Industry Associations
- American Academy of Pediatrics
- Infant Nutrition Council
- Council for Responsible Nutrition
- International Alliance of Dietary/Food Supplement Associations
Public Domain Resources
- World Health Organization – Infant nutrition guidelines
- National Institutes of Health – Vitamin D fact sheets
- Centers for Disease Control and Prevention – Infant supplementation recommendations
- European Food Safety Authority – Nutrient intake standards
Proprietary Elements
- CMI Data Analytics Tool: Proprietary analytics tool to analyze real-time market trends, consumer behavior, and technology adoption in market
- Proprietary CMI Existing Repository of Information for Last 10 Years
Share
Share
About Author
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients