Global Bacterial Vaginosis Drug Market Size and Forecast – 2026 To 2033
The global bacterial vaginosis drug market is expected to grow from USD 1.38 Bn in 2026 to USD 2.05 Bn by 2033, registering a compound annual growth rate (CAGR) of 5.8% from 2026 to 2033. The market for bacterial vaginosis drugs is poised for significant expansion, fueled by the soaring burden of sexually transmitted and reproductive tract infections.
According to the World Health Organization, more than 1 million curable sexually transmitted infections (STIs) are acquired every day worldwide, with 374 million new infections reported annually across key STIs such as chlamydia, gonorrhea, syphilis, and trichomoniasis. In addition, the WHO estimates that over 520 million people aged 15–49 are living with herpes simplex virus type 2 (HSV-2), while approximately 8 million adults were infected with syphilis in 2022, highlighting the substantial global burden of genital infections.
(Source: World Health Organization)
Key Takeaways of the Global Bacterial Vaginosis Drug Market
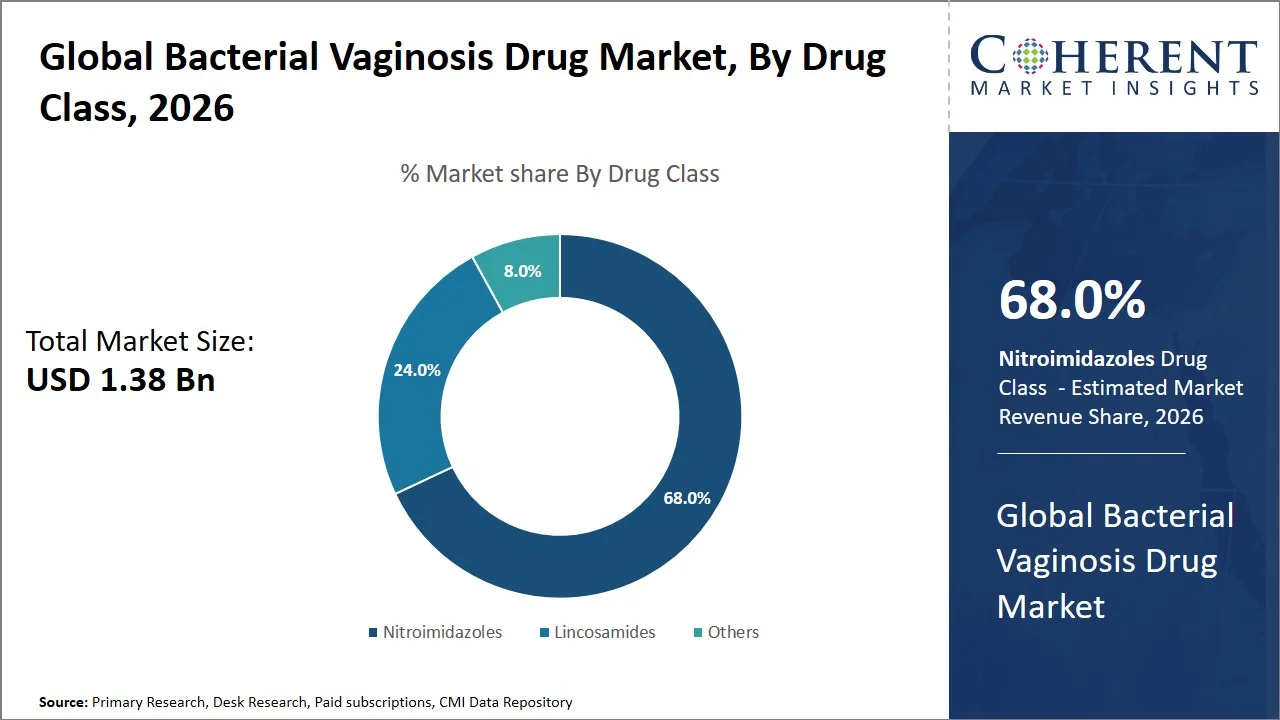
- The nitroimidazoles segment is projected to hold 68.0% of the global bacterial vaginosis drug market share in 2026, making it the dominant drug class segment, especially prevalent in Europe, wherein the existence of centralized clinical guidelines that ensure standardized antibiotic use within member countries. For instance, National Institute for Health and Care Excellence specifically advises metronidazole for the first-line treatment of bacterial vaginosis, thereby supporting standardized prescription practices in public health systems. Such a prescriptive approach would guarantee that nitroimidazoles continue to dominate the market through their continuous prescription. (Source: National Institute for Health and Care Excellence)
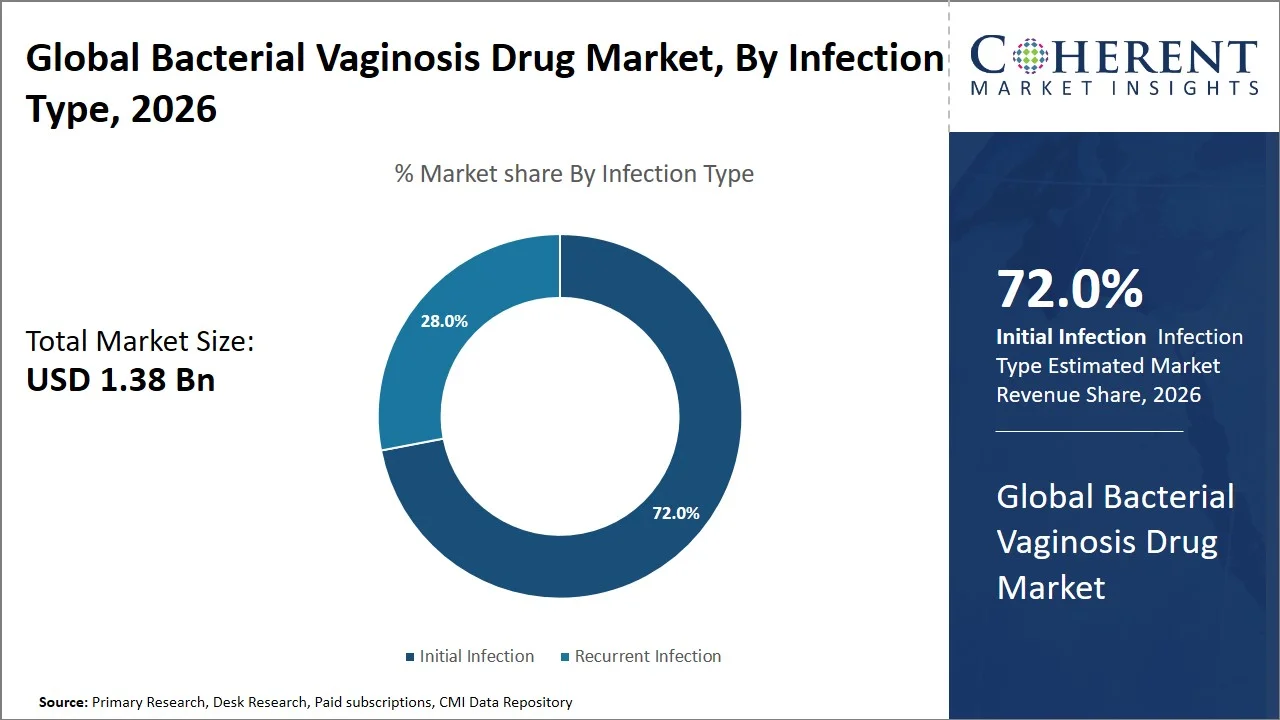
- Initial infection is projected to hold 72.0% of the global bacterial vaginosis drug market share in 2026, making it the dominant infection type segment, with strong prominence in Europe, where sexual health services are organized to guarantee early diagnosis and prompt treatment. For instance, free and confidential testing for sexually transmitted infection/bacterial vaginosis is offered by the National Health Service in Genitourinary Medicine (GUM) clinics to allow quick diagnosis of new infections and provision of immediate treatment, which leads to high treatment numbers. (Source: National Health Services)
- Oral is projected to hold 57.0% of the global bacterial vaginosis drug market share in 2026, making it the dominant route of administration segment, with strong dominance in the Asia Pacific region, especially in China, where formularies at hospitals and prescribing system prefer standardized oral antibiotics for gynecologic infections. For instance, National Essential Medicines List in China, under the purview of the National Health Commission, necessitates the utilization of listed drugs within primary health facilities, where anti-infective medicines are extensively administered, contributing to high prescription rates and availability. (Source: National Library of Medicine)
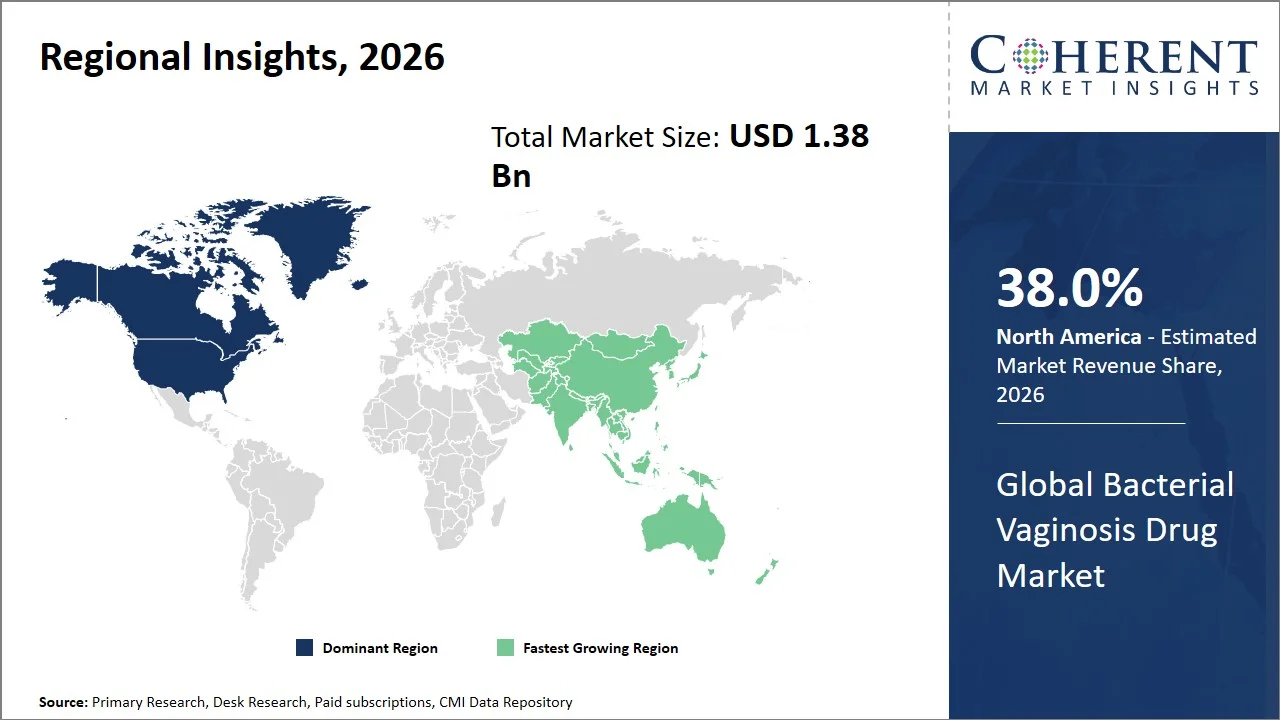
- North America maintains its dominance with an expected share of 38.0% in 2026, supported by formal treatment pathways and insurance coverage for bacterial vaginosis drugs within insured health care systems, ensuring compliance with prescriptions. For instance, Centers for Medicare & Medicaid Services provide outpatient prescription drug benefits in Medicaid and Medicare Part D programs, allowing access to antibiotic medications for the treatment of vaginal infections among low- and middle-income groups. (Source: Centers for Medicare & Medicaid Services) In addition, widespread availability of FDA-approved generics through national retail pharmacy networks guarantees affordability and consistent demand, especially among first-line oral medicines.
- Asia Pacific is expected to exhibit the fastest growth with an estimated contribution of 22.0% share in 2026, due to the expansion of women's health care services and growing involvement in managing reproductive infections in national health care policies. For instance, the diagnosis and management of vaginal infections, such as bacterial vaginosis, is being integrated within the scope of the RMNCH+A strategy implemented by India's Ministry of Health and Family Welfare. As a result, early detection and prescription are becoming more common, especially in the field of public health, which helps boost the market growth. (Source: Ministry of Health and Family Welfare)
- Expansion of prescription digital therapeutics and telehealth-based bacterial vaginosis management: Telemedicine platforms are becoming increasingly integrated, providing avenues for diagnosis and prescription of bacterial vaginosis medication from afar. The adoption of digital health technology is also increasing access to treatment in conditions such as bacterial vaginosis that have stigmatized patient engagement with in-person appointments. Pharmaceutical firms can now benefit from partnerships with digital health organizations to gain access to more patients, especially for oral treatment regimens.
- Increasing focus on adjunctive therapies targeting vaginal microbiome restoration: Apart from traditional antibiotics, there is an increasing need for complementary treatment methods like probiotics and vaginal microbiota regulators that will help restore normal flora following therapy. This is being increasingly adopted due to the high rate of recurrence (around 50-60%) after 6-12 months, thereby making it advisable to employ combination or maintenance treatments. There is a growing market opportunity for companies developing complementary non-antibiotic products and prevention methods.
Why Does the Nitroimidazoles Segment Dominate the Global Bacterial Vaginosis Drug Market?
The nitroimidazoles segment is projected to hold a market share of 68.0% in 2026, owing to their efficacy, wide-spectrum of activity against anaerobic bacteria and consistent endorsement by treatment guidelines. For example, drugs such as metronidazole and tinidazole are recommended as first-line treatments since they have the ability to treat key bacterial vaginosis bacteria such as G. vaginalis and anaerobes. Cure rates of 65% to 90% have been reported by clinical trials, depending on formulation and method of application, with some trials showing metronidazole with cure rates of 91% and tinidazole with 96.5%. (Source: National Library of Medicine)
Additionally, meta-analysis shows that nitroimidazoles have superiority over placebos and are still considered some of the most effective antibiotics available. (Source: Frontiers) Their flexibility in oral and vaginal administration forms, cost efficiency, and years of clinical experience have only added to their supremacy. Moreover, international therapy recommendations, including public health agency recommendations, keep emphasizing the importance of nitroimidazoles in therapy, which ensures continued prescription levels. Although they pose a challenge in treating recurring cases of bacterial vaginosis, their ability to quickly alleviate symptoms and proven effectiveness makes them the drug of choice.
Why does Initial Infection Represent the Largest Infection Type Segment in the Global Bacterial Vaginosis Drug Market?
To learn more about this report, Request Free Sample
Initial infection is projected to hold a market share of 72.0% in 2026, since the onset of the condition is more likely to lead to a clinical pathway involving complete treatment with drugs. Population surveys conducted in the U.S like the National Health and Nutrition Examination Survey (NHANES) show bacterial vaginosis prevalence levels of about 29.2% among women aged between 14 and 49 years, with a significant proportion being new diagnoses. (Source: Centers for Disease Control and Prevention) Moreover, a longitudinal study featured in the Lancet Infectious Diseases reveals that incidents of bacterial vaginosis infections are common among vulnerable groups in the span of months. (Source: The Lancet)
Furthermore, the clinical guidelines recommend prompt treatment after diagnosis to avoid any severe consequences like pelvic inflammatory disease or problems with pregnancy. Consequently, prescription adherence tends to be high at the time of diagnosis. (Source: Centers for Disease Control and Prevention)
In addition, repeat infections tend to move into the observation mode or lifestyle modifications as well as periodic medication administration. Lastly, campaigns for raising awareness on bacterial vaginosis infections (such as, CDC STD Awareness Month, National Women’s Health Week, It’s Time We Talked Campaign by The Eve Appeal, Global STI Awareness Campaign by WHO, Vaginal Health Awareness initiatives by American Sexual Health Association (ASHA)) and regular gynecological checks improve the early diagnosis rates in urban healthcare setups, increasing the number of people undergoing treatment for the first time.
Oral Segment Dominates the Global Bacterial Vaginosis Drug Market
The oral segment is projected to hold a market share of 57.0% in 2026, attributed to their high levels of compliance, efficiency, and preference for guideline-based first-line treatment. Oral antibiotics like Metronidazole are commonly prescribed since they allow for even distribution of medication and the elimination of anaerobic bacteria, especially in moderate-to-severe cases. Guidelines published by the Centers for Disease Control and Prevention emphasize oral regimens as a first-line treatment method, further validating their extensive usage. (Source: Centers for Disease Control and Prevention)
Moreover, research findings show that the oral route of administration is favored by patients and doctors alike because of its convenience and high compliance rates relative to topicals. For instance, based on clinical trials, oral and vaginal treatments are equally effective in terms of curing rates, which usually lie within the range of approximately 70%-80%; nevertheless, oral metronidazole is currently widely used in practice as the gold standard for treatment. (Source: Journal of American Sexually Transmitted Diseases Association) The combination of clinical efficacy, physician preference, and guidelines continues to drive the dominance of the oral segment in the bacterial vaginosis drug market.
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
Regulatory Shift: Updated Clinical Guidelines Recommending Partner Therapy for Recurrent BV (October 2025) |
|
|
Regulatory Advancement: FDA Support for Microbiome-Based & Non-Antibiotic BV Therapies (2025–2026) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
(Source: American College of Obstetricians and Gynecologists, University of Maryland School of Medicine)
Global Bacterial Vaginosis Drug Market Dynamics
To learn more about this report, Request Free Sample
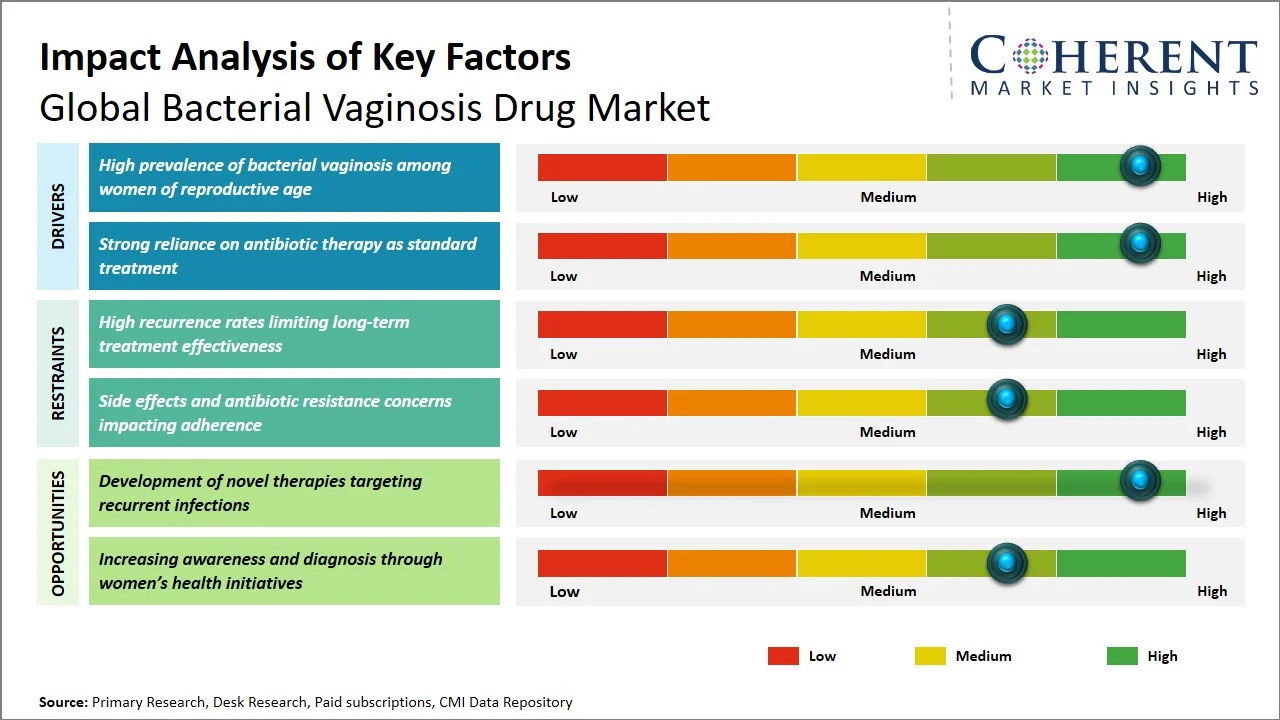
Market Drivers
High prevalence of bacterial vaginosis among women of reproductive age
The high incidence of bacterial vaginosis amongst women in their reproductive age still plays a significant role in the market’s growth, since this translates to ongoing need for treatment. The World Health Organization states that the prevalence of bacterial vaginosis across the globe among women of reproductive age group ranges from 23% to 29%, making it one of the commonest gynecological problems encountered by healthcare professionals. (Source: World Health Organization) In many regions, reports suggesting that around 20-30% of women suffer from bacterial vaginosis, while the prevalence could even be as high as 50% among certain population groups highlighting a substantial patient pool requiring clinical management. (Source: National Library of Medicine)Furthermore, Centers for Disease Control and Prevention states that bacterial vaginosis is the most common vaginal condition in women ages 15-44. (Source: Centers for Disease Control and Prevention) The link between the disease and its complications, including a higher risk of sexually transmitted diseases and negative pregnancy outcomes, calls for prompt management.
Strong reliance on antibiotic therapy as standard treatment
The market growth of bacterial vaginosis drug is highly associated with the continued dependence on antibiotic therapy as the main form of treatment and, thus, there will be continued demand for the drugs in terms of prescriptions. According to guidelines released by the Centers for Disease Control and Prevention, the recommended medications for treating symptoms of bacterial vaginosis include Metronidazole and Clindamycin. These guidelines encourage the continued use of the two drugs in treatment. (Source: Centers for Disease Control and Prevention) Such ingrained clinical dependence ensures repeat prescriptions, especially in recurrent cases, ensuring sustained demand for drugs.
Furthermore, the use of oral and topical antibiotics offers an added advantage due to the flexibility in treatment, thus promoting their acceptance among patients with varied backgrounds.
Emerging Trends
- Shift toward microbiome-restoring therapies: There is a trend toward shifting from conventional antibiotics to the development of new approaches based on microbiome modulation, which aim at restoring healthy vaginal flora and preventing recurrent infections. This trend includes an increasing focus on live biotherapeutics and probiotics with disease management capability over extended periods of time.
- Development of patient-friendly, low-burden treatment formats: There is an increasing trend towards simpler treatment strategies like one-time oral administration or extended release from vaginal applications that have the potential to increase compliance. These developments are specifically for the outpatient care setting as well as telemedicine settings where convenience plays an important role.
Regional Insights
To learn more about this report, Request Free Sample
Why is North America a Strong Market for Bacterial Vaginosis Drugs?
North America leads the global bacterial vaginosis drug market, accounting for an estimated 38.0% share in 2026, attributed to its ability to swiftly turn late-stage clinical advancements into commercial products and incorporate them into standard gynecological treatment procedures. For instance, Daré Bioscience has developed DARE-BV1 (clindamycin phosphate vaginal gel 2%) as a once-dose intravaginal treatment, aimed to improve compliance and ease treatment as opposed to multiple dose treatments, highlighting the relationship between innovation and clinical practice needs. (Source: Daré Bioscience)
The region’s dominance is further supported by the high level of physician adoption for new product formulations in OB-GYN practices, which allows for quick standardization of treatments following the introduction of innovative medicines. Moreover, the availability of commercialization structures tailored to specialties in women’s health provides for effective physician targeting and accelerated adoption of unique products.
Why Does the Asia Pacific Bacterial Vaginosis Drug Market Exhibit High Growth?
The Asia Pacific bacterial vaginosis drug market is expected to exhibit the fastest growth with an estimated contribution of 22.0% share to the market in 2026, attributed to the growing inclusion of the management of bacterial vaginosis into routine gynecological care pathways in hospitals' outpatient department and private clinics, especially those located in urban areas. For instance, hospitals and Obstetrician-Gynecologist clinics in countries such as India and China are using the combined drug and vaginal gel treatments in their treatment protocols as part of their clinical management systems for symptomatic vaginal infections, underscoring a shift toward structured clinical management. (Source: National Library of Medicine)
Moreover, pharmacy-based dispensing and teleconsultation software have been facilitating faster diagnosis-to-treatment processes, particularly in highly populated areas. The rising convergence between clinical practices and evidence-based medicine with better access routes has clearly contributed to rapid market expansion in the Asia Pacific region.
Global Bacterial Vaginosis Drug Market Outlook for Key Countries
Why is the U.S. Leading Innovation and Adoption in the Bacterial Vaginosis Drug Market?
The U.S. has the potential for leadership in innovation and adoption in the field of bacterial vaginosis drug owing to the presence of an active clinical pipeline with regard to microbiome-related and live biotherapeutic products (such as, LACTIN-V, LC-106/LC-115 consortia therapies, TOL-463) especially the ones targeted at preventing the recurrence of the condition. There is also the presence of specialized women’s health organizations (such as, American College of Obstetricians and Gynecologists, Society for Maternal-Fetal Medicine, American Society for Reproductive Medicine) and the highly robust venture-capital supported biotech ecosystem, which helps accelerate the development of vaginal microbiome-related products into commercially viable offerings.
Moreover, the early adoption by physicians of innovative product types such as a single-dose oral administration and prescription vaginal gel is instrumental in ensuring fast market uptake after receiving U.S. FDA approval.
Is U.K. a Favorable Market for Bacterial Vaginosis Drugs?
The U.K. is an ideal market for bacterial vaginosis drug attributed to the presence of the well-established network of sexual health clinics (GUM clinics), which guarantees fast detection and treatment of bacterial vaginosis. The National Health Service provides the guidelines for prescribing the treatment and ensures the consistency of using the recommended bacterial vaginosis medications throughout the country. Moreover, the guidelines of the British Association for Sexual Health and HIV (such as., BASHH Bacterial Vaginosis Treatment Guideline, BASHH Vaginal Discharge Guideline, BASHH STI Management and Testing Guideline) contribute to providing a uniform method of treating patients with bacterial vaginosis.
Is China Emerging as a Key Growth Hub for the Bacterial Vaginosis Drug Market?
Bacterial vaginosis drug market in China is currently experiencing significant development attributed to the rapid development of hospitals' gynecology departments as well as infection management procedures among urban hospitals. Due to the changes introduced by the National Medical Products Administration, it became much easier to receive approval certificates for antiseptics and feminine products; thus, many new medications produced not only in China but worldwide can reach the market faster.
Moreover, local pharma firms (such as, Jiangsu Hengrui Pharmaceuticals, CSPC Pharmaceutical Group, Luye Pharma Group, Shanghai Fosun Pharmaceutical) are aggressively engaged in creating vaginal gels and combination drugs that suit the regional practice of medicine, which improves accessibility. The increasing collaboration between hospital pharmacies and health care IT platforms also helps enhance patients' access to bacterial vaginosis drugs.
Why Does Germany Top the Europe Bacterial Vaginosis Drug Market?
Germany dominates the bacterial vaginosis drug market in Europe owing to the physician-driven prescription practice and significant role of office-based gynecologists in making sure that the condition is diagnosed and treated in time. The Federal Joint Committee (G-BA) is engaged in developing a well-structured approach to the treatment process, as well as taking care of reimbursement issues. Furthermore, the market in Germany enjoys high penetration of brands and generics of vaginal preparations that are easily available through retail pharmacies. The involvement of big pharma companies like Bayer makes the situation even better for products and market maturity.
Is Bacterial Vaginosis Drug Market Developing in India?
The market for bacterial vaginosis drug is currently undergoing development in India owing to the advent of specialized gynecology out-patient clinics and the increased use of vaginal combination therapies. The regulatory oversight by the Central Drugs Standard Control Organization (CDSCO) has made it easier for different types of antibiotics and topicals employed in treating bacterial vaginosis to be made available. Local drug manufacturing firms such as Cipla and Sun Pharmaceutical Industries have expanded their product range within the women’s health area by adding anti-infectives. Moreover, pharmacy-led dispensing and telemedicine services are facilitating the availability of treatment, especially in urban and semi-urban settings.
Regulatory Landscape Governing the Global Bacterial Vaginosis Drug Market
|
Region |
Key Regulatory Bodies |
Approval & Compliance Framework |
Impact on Market |
|
North America |
U.S. Food and Drug Administration, Health Canada |
Drug approvals via NDA/ANDA pathways with strict clinical trial requirements, bioequivalence studies for generics, and pharmacovigilance compliance. BV drugs such as antibiotics must meet safety and efficacy standards under FDA guidelines. |
High regulatory rigor ensures safety and efficacy but increases development timelines and costs. Strong generic approval pathways support market penetration of cost-effective therapies. |
|
Europe |
European Medicines Agency, National Competent Authorities |
Centralized and decentralized procedures for drug approval, with emphasis on quality, safety, and efficacy. Compliance with Good Manufacturing Practice (GMP) and pharmacovigilance regulations is mandatory. |
Harmonized regulations facilitate multi-country access, while stringent compliance requirements encourage high-quality product development and limit entry of substandard drugs. |
|
Asia Pacific |
Pharmaceuticals and Medical Devices Agency, Central Drugs Standard Control Organization, National Medical Products Administration |
Diverse regulatory frameworks with evolving standards. Increasing alignment with global guidelines (ICH), faster approval pathways in some countries, and growing emphasis on local clinical data. |
Regulatory heterogeneity creates challenges for global players but improving frameworks and faster approvals are enhancing market accessibility and growth opportunities. |
|
Latin America |
Agência Nacional de Vigilância Sanitária, Comisión Federal para la Protección contra Riesgos Sanitarios |
Regulatory processes involve local clinical data requirements, import licensing, and GMP compliance. Approval timelines can vary significantly across countries. |
Slower and fragmented regulatory systems may delay product launches, but increasing regulatory modernization is gradually improving market entry conditions. |
|
Middle East & Africa |
Saudi Food and Drug Authority, South African Health Products Regulatory Authority |
Regulatory systems are developing, with reliance models (reference to FDA/EMA approvals) commonly used. Focus on quality standards, import regulations, and pharmacovigilance is increasing. |
Growing regulatory maturity is improving drug availability, though variability across countries can create barriers for consistent market penetration. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
How is the development of novel therapies targeting recurrent infections creating new growth opportunities in the global bacterial vaginosis drug market?
Innovation in the creation of new treatments for recurrent bacterial vaginosis presents great potential for growth through tackling one of the market’s most pressing unsolved problems—relapse rates following conventional antibiotic therapy. New treatments, such as innovative microbiome therapies and prolonged drug treatment, are helping transform the current approach to treating bacterial vaginosis from a limited course of antibiotic therapy to an ongoing strategy to prevent relapses. For instance, the results of phase 1 trials conducted by Mass General Brigham-Probiotic Therapy for bacterial vaginosis showed that a multi-strain probiotics therapy could help restore protective vaginal flora and reduce recurrence rates, with positive effects persisting for several months post-treatment. (Source: Mass General Brigham Incorporate)
Moreover, new live biotherapeutic drugs, such as LACTIN-V, are proving their ability to restore vaginal microbiota containing Lactobacillus species in up to 30% of patients, compared to low placebo efficacy levels. (Source: AZoNetwork) On the other hand, innovations in drug design have led to developments in one-dose or multi-dose antibiotics, including Secnidazole (SOLOSEC), which is used for once-off oral administration and is currently under investigation for bacterial vaginosis recurrence treatment. (Source: HMP Global)
Furthermore, pharmaceutical companies such as Gedea Biotech have introduced new clinical studies (BV2025 study) for the development of non-antibiotic medicines designed for infection control. (Source: Gedea Biotech) In combination, such breakthroughs from microbial ecosystem restoration to new approaches in dosage have opened up new possibilities for earning revenues, thus driving the bacterial vaginosis drug market forward.
Market Players, Key Development, and Competitive Intelligence

To learn more about this report, Request Free Sample
Key Developments
- In September 2025, Gedea Biotech received the CE mark for its product named pHyph which is a new treatment method for bacterial vaginosis that does not involve any antibiotics. The innovation is a new approach for treating bacterial vaginosis as it is an antibiotic-free method of curing infections. It highlights a shift toward innovative, non-antibiotic therapies in the market.
- In May 2025, Evofem Biosciences entered into a license deal with Pharma 1 to develop their bacterial vaginosis treatment drug, SOLOSEC, in the Middle Eastern region. Under the terms of the agreement, there are plans to seek regulatory approval in the United Arab Emirates. This event is indicative of the increased interest in strategic alliances geared towards expanding access to bacterial vaginosis treatments in emerging economies.
Competitive Landscape
The global bacterial vaginosis drug market is moderately competitive due to the involvement of well-established pharmaceutical organizations as well as generic drugs providers, who are providing cost-effective options for treatment of the disease. The competition within the market arises due to the presence of the first-line treatments that include Metronidazole and Clindamycin. Moreover, there has been an emphasis towards improving compliance in patients by using innovative drug forms. Extended-release drugs, combination drugs, vaginal gel, and even oral medication routes are being used. Key focus areas include:
- Development of extended-release and patient-friendly formulations
- Expansion into microbiome-based and probiotic therapies to address recurrence
- Strengthening generic portfolios and cost-competitive offerings
- Increasing distribution through gynecology clinics, retail pharmacies, and online channels
- Strategic collaborations for clinical trials and novel drug development
Market Report Scope
Bacterial Vaginosis Drug Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1.38 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 5.8% | 2033 Value Projection: | USD 2.05 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Pfizer, GlaxoSmithKline, Bayer AG, Sanofi, Novartis AG, Lupin Limited, Teva Pharmaceutical Industries, Padagis, Evofem Biosciences, and Organon & Co. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Analyst Opinion (Expert Opinion)
- In the coming years, the prospects of the bacterial vaginosis drug market will witness a shift towards more specific and effective treatments that are focused on minimizing recurrences and addressing vaginal microbiota imbalances. Since persistent relapses continue to pose challenges for medical professionals, further innovations in the market are likely to include prolonged-release medication, drug combinations, and the use of vaginal microbiota normalization techniques. At the same time, growing awareness regarding women’s health issues and antimicrobial resistance among regulatory authorities will result in the development of new solutions.
- The highest opportunities are anticipated for therapeutics based on the microbiome and those not involving antibiotics in North America, owing to robust clinical research facilities and high rate of diagnosis and acceptance of novel medicines. In particular, the use of prescription vaginal gels and sustained release medicines is estimated to grow significantly due to patient compliance and accurate drug delivery. However, the Asian Pacific countries provide significant untapped opportunities, although short-term sales will primarily be generated from developed countries.
- For gaining any competitive advantage in the market, companies need to concentrate on strategies for pipeline differentiation and life-cycle management. Some of these include innovations around delivery systems as well as combination therapies that could be used against recurrence. Collaboration with research centers for conducting microbiome analysis, fast-tracking of regulations, and creation of real-world evidence would also be key. Moreover, efforts towards educating physicians and creating patient awareness, along with penetration into the outpatient and retail pharmacy sectors, are also imperative.
Market Segmentation
- Drug Class Insights (Revenue, USD Bn, 2021 - 2033)
- Nitroimidazoles
- Lincosamides
- Others
- Infection Type Insights (Revenue, USD Bn, 2021 - 2033)
- Initial Infection
- Recurrent Infection
- Route of Administration Insights (Revenue, USD Bn, 2021 - 2033)
- Oral
- Topical
- Distribution Channel Insights (Revenue, USD Bn, 2021 - 2033)
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
- Regional Insights (Revenue, USD Bn, 2021 - 2033)
- North America
- U.S.
- Canada
- Latin America
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Europe
- Germany
- U.K.
- Spain
- France
- Italy
- Russia
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Australia
- South Korea
- ASEAN
- Rest of Asia Pacific
- Middle East
- GCC Countries
- Israel
- Rest of Middle East
- Africa
- South Africa
- North Africa
- Central Africa
- North America
- Key Players Insights
- Pfizer
- GlaxoSmithKline
- Bayer AG
- Sanofi
- Novartis AG
- Lupin Limited
- Teva Pharmaceutical Industries
- Padagis
- Evofem Biosciences
- Organon & Co.
Sources
Primary Research Interviews
- Gynecologists, obstetricians, and reproductive health specialists from hospitals and women’s health clinics
- Infectious disease specialists involved in the diagnosis and management of vaginal infections
- Pharmaceutical manufacturers specializing in women’s health and anti-infective drug development
- Hospital procurement managers and pharmacists managing antibiotic and gynecological drug supply
- Clinical researchers and investigators involved in microbiome and vaginal health studies
Stakeholders
- Bacterial Vaginosis Drug Manufacturers
- End-use Sectors
- Hospitals and Gynecology Clinics
- Ambulatory Surgical Centers
- Retail and Online Pharmacies
- Regulatory & Health Bodies: U.S. Food and Drug Administration – drug approvals and safety guidelines for BV therapeutics, European Medicines Agency – regulatory framework for anti-infective drugs, World Health Organization – global STI burden and treatment guidelines, Centers for Disease Control and Prevention – STI surveillance and treatment recommendations, Central Drugs Standard Control Organization – pharmaceutical regulations and approvals
Databases
- ClinicalTrials.gov – studies on bacterial vaginosis drugs and microbiome therapies
- World Health Organization Global Health Observatory – STI epidemiology and reproductive health data
- PubMed – research on bacterial vaginosis treatment outcomes and antibiotic resistance
Magazines
- Nature Medicine – research on microbiome therapeutics and women’s health innovations
- Science Magazine – advancements in infectious disease treatment and antimicrobial research
- NIH Research Matters – updates on vaginal microbiome and infection management
Journals
- American Journal of Obstetrics and Gynecology
- The Lancet Infectious Diseases
- Journal of Women’s Health
- Clinical Infectious Diseases
Newspapers
- The New York Times (Health Section) – coverage on women’s health and infection trends
- The Guardian (Health & Science) – reporting on global STI burden
- Financial Times – pharmaceutical market trends and investments
Associations
- American College of Obstetricians and Gynecologists
- Infectious Diseases Society of America
- International Society for Sexually Transmitted Diseases Research
Public Domain Sources
- National Institutes of Health – research on vaginal microbiome and infection treatment
- Centers for Disease Control and Prevention – STI statistics and treatment guidelines
- World Health Organization – global STI and reproductive health data
- National health ministries (e.g., U.S., U.K., India, Australia) – healthcare access and infection burden data
Proprietary Elements
- CMI Data Analytics Tool, Proprietary CMI Existing Repository of information for last 10 years.
Share
Share
About Author
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients