Global Burette Set Market Size and Forecast: 2026-2033
The global burette set market is expected to grow from USD 620 Mn in 2026 to USD 1,020 Mn by 2033, registering a compound annual growth rate (CAGR) of 7.4%. The global market for burette set is poised for significant expansion, fueled by increasing emphasis on patient safety during infusion therapy, especially in pediatrics and neonatology, where accurate fluid administration is imperative.
For instance, the World Health Organization emphasizes the vulnerability of children to medication errors due to weight-based dosing requirements, which increase the risk of calculation and administration errors thereby necessitating precise infusion control systems such as burette sets.
(Source: World Health Organization)
Key Takeaways of the Global Burette Set Market
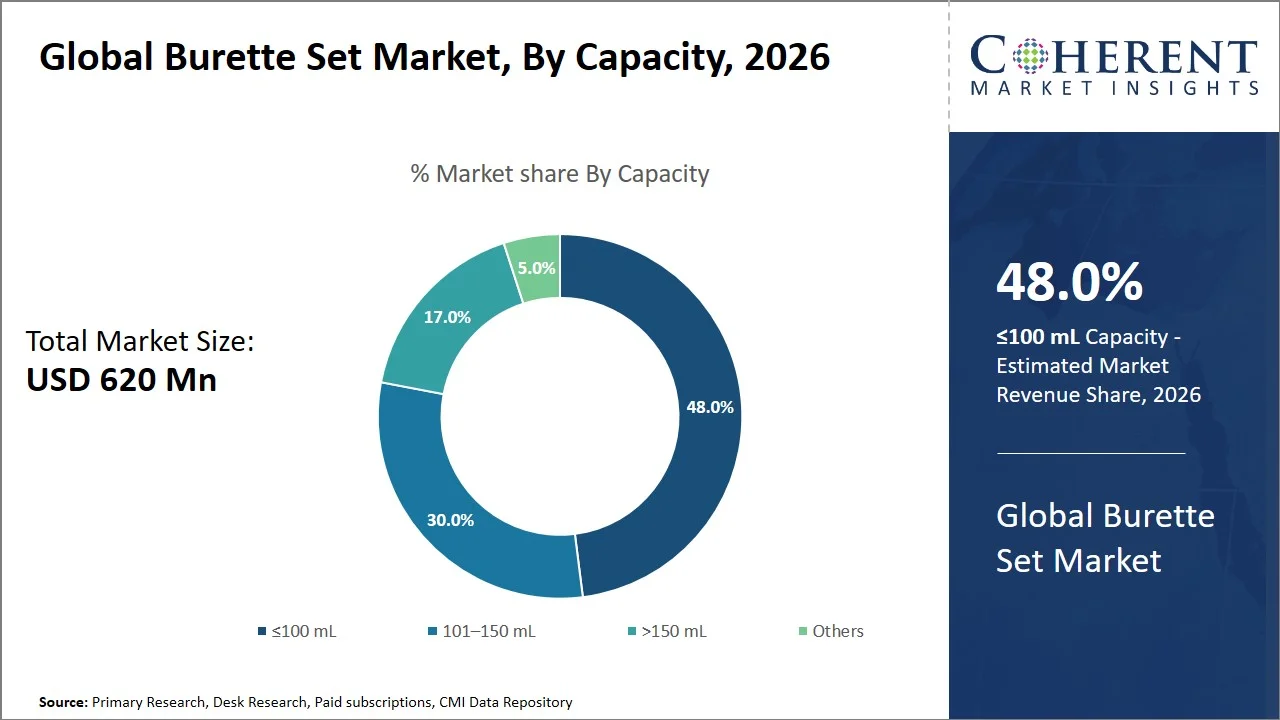
- ≤100 mL is projected to hold 48.0% of the global burette set market share in 2026, making it the dominant capacity segment, with strong penetration in Asia Pacific, where the requirement for pediatric and neonatal care services are growing at a quick pace. For instance, countries like India are scaling Neonatal Intensive Care Units (NICU) infrastructure through various national initiatives, such as Facility Based Newborn Care (FBNC), that aim at setting up specialized centers like Special Newborn Care Units (SNCUs), Newborn Stabilization Units (NBSUs), and Newborn Care Corners (NBCCs), across public health facilities to improve neonatal outcomes. (Source: Ministry of Health and Family Welfare) In clinical practice, neonates require micropoint volume IV infusion, whereby any mistake in dosing will have serious repercussions. Research indicates that the frequency of medication errors in children can go up to 11.1%, particularly within hospital environments, emphasizing the need for controlled, small-volume delivery systems. (Source: National Library of Medicine)
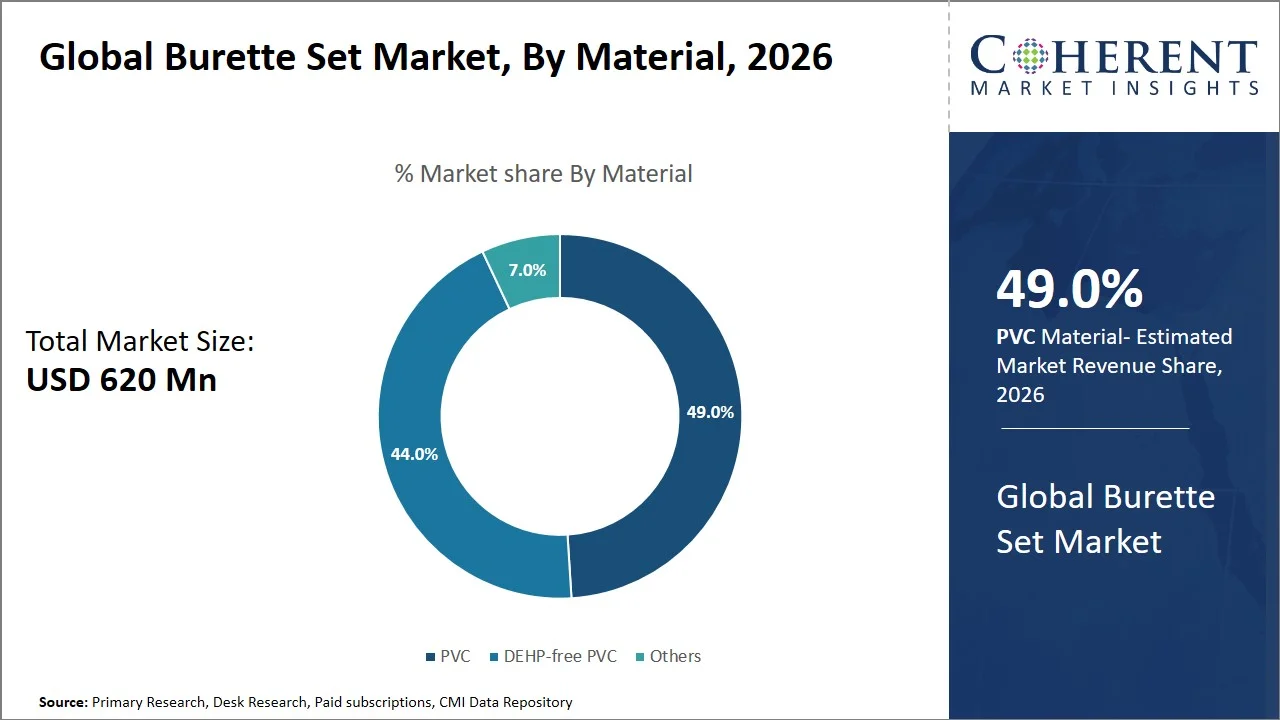
- PVC is projected to hold 49.0% of the global burette set market share in 2026, making it the dominant material segment, especially in price-sensitive regions like Asia Pacific and Latin America, where public hospitals use large numbers of infusion consumables through volume purchases. The government focuses on providing affordable and disposable infusion consumables to handle more patients. Even though there is an increasing trend of using DEHP-free materials in advanced economies, the use of PVC persists because of its inexpensive nature and flexibility. With hospitals using tens of thousands of infusions sets daily, it is cheaper to use PVC than other materials. For instance, in India, under the National Health Mission, public hospitals purchase medical consumables, including infusion-related products, using procurement and tender-based systems that emphasize bulk purchasing at lower costs to ensure constant supply at high volume hospitals. (Source: Department of Health and Family Welfare)
- Pediatric / Neonatal is projected to hold 58.0% of the global burette set market share in 2026, making it the dominant patient type segment, with strong potential in North America and Europe attributed stringent safety requirements and clinical practices that mandate precision infusion systems (such as Infusion Nurses Society standards, National Institute for Health and Care Excellence guidelines, and European Society for Paediatric Research recommendations). For instance, according to the Centers for Disease Control and Prevention, about 1 out of 10 babies born in the U.S. are born prematurely. (Source: Centers for Disease Control and Prevention) These babies often need to be admitted in the Neonatal Intensive Care Units (NICU), where they receive intensive treatment and precise management of their intravenous fluid intake. This reliance on accurate and low-volume delivery of fluids is one of the reasons behind the widespread use of burette set.
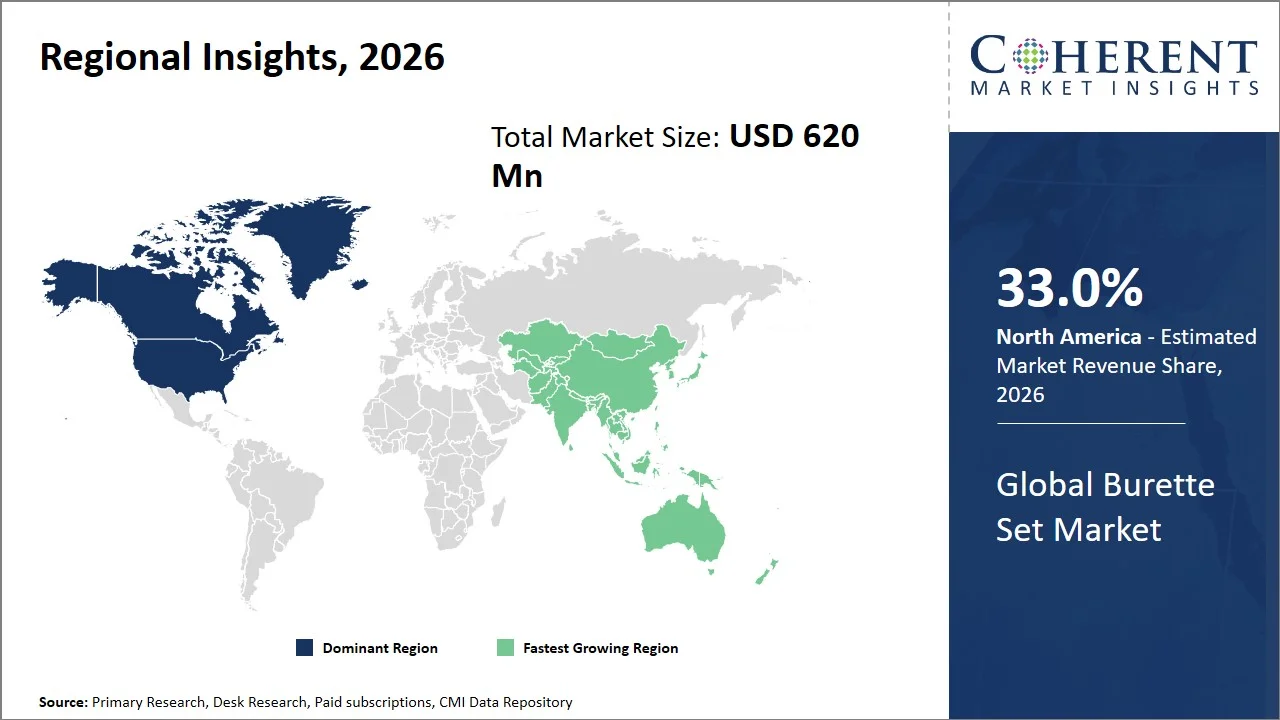
- North America maintains dominance with an expected share of 33.0%in 2026, backed by robust procurement processes in hospitals and stringent compliance regulations that encourage the use of clinically proven infusion disposables (such as U.S. Food and Drug Administration (FDA) Quality System Regulation – 21 CFR Part 820, Health Canada Medical Devices Regulations (SOR/98-282), Centers for Disease Control and Prevention (CDC) infusion safety guidelines, and the Joint Commission standards for medication and infusion safety). The region is known for having a high adoption rate of standardized infusion practices and high ICU bed capacity, hence frequent utilization of controlled infusion devices. For instance, the American Hospital Association reports that the U.S. has over 6,100 hospitals, reflecting a large and structured care network with consistent demand for infusion devices. (Source: American Hospital Association)
- Asia Pacific is expected to exhibit the fastest growth with an estimated contribution of 26.0% share in 2026, stimulated by the fast growth of the hospitals' physical infrastructure, as well as the number of patients in densely populated countries like India and China. The region’s government is spending heavily on healthcare capacity, especially secondary and tertiary healthcare providers, which is driving up the demand for infusion disposables. For instance, data from the World Bank reveals that East Asia & Pacific have more than 4 hospital beds per 1,000 people, indicating an increasing base of inpatient services that directly boosts the demand for IV drug delivery systems. (Source: World Bank Group)
- Increasing Adoption in Neonatal and Pediatric Intensive Care Units (NICUs/PICUs): The increasing development of Neonatal Intensive Care Units (NICU) and Pediatric Intensive Care Units (PICU) infrastructure, especially in developing countries, is helping in the creation of niche yet valuable demand for burette sets. In such setups, there is a need for microvolume and precise infusion, which requires accurate administration of medications because any mistake may cause severe consequences. As the government and other private organizations invest in setting up specialized pediatric units, demand for burette set will continue growing.
- Growing Preference in Low-Resource and Cost-Sensitive Healthcare Settings: In several developing countries, there is increasing adoption of burette set as a more affordable substitute for expensive infusion devices, particularly in secondary health institutions. The reason for their suitability in such settings are that they offer precise fluid delivery without dependence on electricity or any sophisticated equipment. Therefore, there exists a high possibility for product manufacturers to grow by offering affordable products in large quantities.
Why Does ≤100 mL Segment Dominate the Global Burette Set Market?
≤100 mL is projected to hold the market share of 48.0% in 2026, due to their correlation with the clinical need for low volume and high accuracy infusion, especially in the case of pediatric and neonatal treatments, where patients have a low volume tolerance. These patients typically require low volumes of fluid infusion as their tiny physical structures limit the amount of fluid that can be administered safely. It means that patients require microvolume or low flow rates as large volumes would be counterproductive to their condition (Source: Baishideng Publishing Group Inc). In such situations, low-volume burette sets, in which the chamber volume is usually 100mL, play a critical role by allowing the fluid delivery to occur safely without triggering any risks of volume overload.
Moreover, the clinical literature emphasizes the importance of accurate delivery of small volumes in pediatric infusion therapy, thus favoring low-volume systems (Source: National Library of Medicine). For example, measured volume infusion devices are developed particularly for accurate dosing during neonatal and pediatric uses, where accurate dosing is crucial to prevent any mistakes. (Source: Lars Medicare)
Why Does PVC Represent the Largest Material Segment in the Burette Set Market?
To learn more about this report, Request Free Sample
PVC is projected to hold a market share of 49.0% in 2026, attributed to its cost efficiency, flexibility, and long-established clinical reliability when administering fluid infusions through burette sets. PVC material is extensively used in medical tubes and equipment due to its flexibility, visibility during monitoring of fluid flow, and suitability for sterilization making it ideal for disposable hospital consumables (Source: RAUMEDIC AG). Moreover, PVC is the least expensive material in large-scale applications in hospitals, particularly when the hospital uses thousands of infusions sets each day. The ease of production of the material and availability through its supply chain make PVC more prevalent in both mature and emerging markets (Source: kohope.com). For example, PVC is still regarded as the ideal material for IV bags and infusion systems globally because of its remarkable strength, flexibility, and sterility, which play an important role in fluid administration in medical facilities. (Source: ScienceDirect)
Pediatric / Neonatal Segment Dominates the Global Burette Set Market
The pediatric / neonatal segment is projected to hold a market share of 58.0% in 2026, owing to the vital need for precise and low-volume infusion in these patients wherein even small differences in the dose administration may cause grave consequences. Usage of burette sets are common in Neonatal and Pediatric Intensive Care Units (NICUs/PICUs) for administering controlled doses of intravenous medicines as the use of such devices ensures that there is no over-infusion of drugs or other fluids. According to the National Institute for Health and Care Excellence (NICE) guidelines, precise fluid administration in children is highly recommended as the risk of complications related to imbalances are considerably high in this group. For instance, according to data presented by UNICEF- children face the highest risk of dying in their first month of life at an average global rate of 17.2 deaths per 1,000 live births in 2024. Globally, 2.3 million children died in the first month of life in 2024 – approximately 6,200 neonatal deaths every day. (Source: UNICEF)
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
U.S. FDA Quality Management System Regulation (QMSR) Final Rule (Effective 2026) |
|
|
EU MDR/IVDR Amendment – Regulation (EU) 2024/1860 (2025 Implementation Phase) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
(Source: Food and Drug Administration, EMERGO))
Global Burette Set Market Dynamics
To learn more about this report, Request Free Sample
Market Drivers
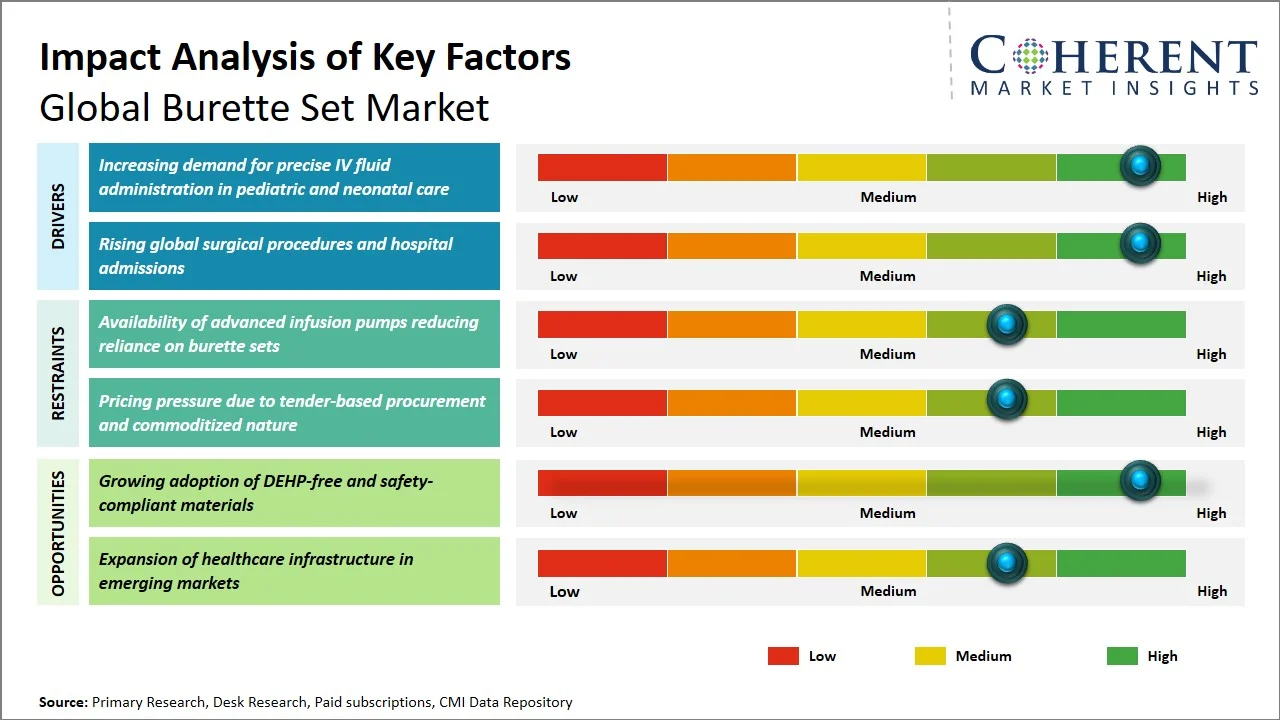
- Increasing demand for precise IV fluid administration in pediatric and neonatal care: There is a need for accuracy and control of fluid flow into patients during IV fluid delivery in the pediatric and neonatal environment due to the potential adverse effects caused by even slight changes in dose amounts that may result in complications like fluid volume overload or electrolyte imbalance. The infants and neonates need dosage based on their body weight and low volume infusion that cannot be provided by the conventional IV fluid delivery sets. International health organizations like the World Health Organization advocate for the practice of safe medicine and infusion techniques in order to minimize mistakes in doses, especially among at-risk patients (Source: World Health Organization). Furthermore, the Centers for Disease Control and Prevention (CDC) advises the use of accurate IV delivery and infection-free infusion systems in hospitals (Source: Centers for Disease Control and Prevention). With an increase in the number of neonatal intensive care units around the world and increased emphasis on infusion accuracy in the clinical environment, the adoption of burette sets is expected to rise steadily.
- Rising global surgical procedures and hospital admissions: An increasing number of surgeries and hospitalizations are the primary factors supporting the growth of the burette sets market due to the necessity of infusion therapy before, during, and after the surgery. Globally, around 313 million
surgeries are carried out each year, additionally, an extra 143 million surgeries are needed in low-income and middle-income countries (LMICs) each year to save lives and prevent disability (Source: The Lancet). Moreover, according to the National Committee for Quality Assurance, about 33.7 million hospital admissions occur every year in the U.S., indicating the large patient base requiring surgical and clinical interventions (Source: National Committee for Quality Assurance). These surgical procedures will call for a meticulous intravenous administration of fluids, electrolytes, and drugs, whereby regulated flow mechanisms are employed to avoid mistakes. With the continuing growth in the number of admissions and operating capabilities at hospitals globally, the need for reliable infusion tools like burette set will rise steadily
Emerging Trends
- Shift Toward DEHP-free and Safer Material Composition: The emergence of health problems owing to the chemicals that leach into the patients' body has witnessed improvements in DEHP (Di(2-ethylhexyl) phthalate)) free PVC and alternative materials in the healthcare sector. The improvement has seen the birth of differentiation in the commodities market due to innovations made to satisfy global safety standards.
- Integration with Advanced Infusion Safety Practices: Burette sets are being increasingly used together with advanced methods of ensuring safety in drug infusion and advanced infusion equipment to cut down on medication mistakes and ensure proper administration of drugs. This trend is quite notable especially in critical care facilities and pediatric care units where accurate drug infusion is very important.
Regional Insights
To learn more about this report, Request Free Sample
Why is North America a Strong Market for Burette Set?
North America leads the global burette set market, accounting for an estimated 33.0% share in 2026, attributed to region’s stringent infection control and compliance culture, characterized by the use of disposable infusion system protocols due to the need for infection control. According to the Centers for Disease Control and Prevention's (CDC) clinical guidelines, all intravenous administration sets and fluid systems should only be used for one patient at a time before being discarded immediately; hence, the steady stream of infusion product consumption in all the hospitals in North America. (Source: Centers for Disease Control and Prevention)
Meanwhile, the Infusion Nurses Society continues to revise its standards for the year 2024 concerning infusion therapy practices. These are protocols for ensuring safe and effective infusion therapies in health care facilities. The constant revision in the catheter infection prevention guidelines regarding the use of IV catheters also requires strict handling procedures, thus ensuring consistent use of infusion devices (Source: Infusion Nurses Society). Altogether, regulatory and clinical developments establish an atmosphere of high compliance and high utilization, where precision infusion devices like burette sets are commonly favored for compliance with safety standards, traceability, and care quality.
Why Does Asia Pacific Burette Set Market Exhibit High Growth?
The Asia Pacific burette set market is expected to exhibit the fastest growth with an estimated contribution of 26.0% share to the market in 2026, owing to improved healthcare facilities, hospitalization, and increasing focus on safe infusion practices across emerging economies such as India and China. The development of healthcare initiatives backed by government policies like Ayushman Bharat has greatly facilitated hospitalization and intravenous therapies, leading to higher demands for regulated infusions.
In terms of supply, there has been rapid growth in the capacity of manufacturers to supply products domestically through policy measures such as production linked incentive schemes and medical device parks which are strengthening local availability of consumables (Source: Ministry of Chemicals and Fertilizers). For instance, Poly Medicure Limited is working to enhance its manufacturing capacities and invest in advanced infusion and critical care devices, including neonatal specific infusion devices. (Source: Trade Brains)
Global Burette Set Market Outlook for Key Countries
Why is the U.S. Leading Innovation and Adoption in the Burette Set Market?
The U.S. is at the forefront of development and adoption in the burette set market because of the strictness of its regulatory framework, which includes the U.S. Food and Drug Administration. Infusion sets fall under Class II devices that must undergo thorough 510(k) approval processes, adhere to the quality system requirements, and undergo ongoing post-market monitoring, thus contributing to the development of advanced products (Source: Operon Strategist). The nation’s healthcare system integrates smart infusion solutions, which incorporate administration sets together with pumps and electronic medical records for reducing the risks of dosing mistakes and improving accuracy in IV therapy.
Moreover, robust manufacturing capacities coupled with swift U.S. FDA approval for innovation further reinforces continuous product upgrades and faster adoption across hospitals and outpatient care settings.
Is Japan a Favorable Market for Burette Set?
Japan represents an ideal environment for burette set market because of the country’s strict regulations that govern the country’s medical devices industry through the Pharmaceuticals and Medical Devices Agency (such as, compliance with the Pharmaceuticals and Medical Devices Act, adherence to Japanese Industrial Standards, and alignment with ISO 13485). With its advanced acute care system, Japan is always looking forward to accurate and error-free delivery systems, especially those used in low volume infusions in geriatrics and intensive care wards. As such, the country's procurement policies favor high-end quality devices (such as., Central Social Insurance Medical Council reimbursement system, Diagnosis Procedure Combination payment system, and national health insurance listing standards under the Ministry of Health Labor and Welfare).
Is China Emerging as a Key Growth Hub for the Burette Set Market?
The emergence of China as a significant growth market for the burette set can be attributed to the rapid expansion of hospitals and robust local manufacturing capabilities of low-cost infusion disposables. The quality control of products through the National Medical Products Administration is improving, thus forcing companies to adopt safe and compliant design features. In addition, centralized purchasing programs are leading to high-volume adoption of standard infusion devices at public hospitals (such as, Volume-Based Procurement program, National Centralized Drug Procurement Program, and provincial-level tendering systems under the National Healthcare Security Administration).
Why Does Germany Top the Europe Burette Set Market?
Germany is the leader in the Europe market for burette set due to high level of standardization of the procurement process in German hospitals and the need for strict adherence to EU medical devices rules under the Medical Device Regulation. Germany’s robust production process for medical devices and focus on safety certification of disposable devices are some of the factors that fuel the usage of burette sets in the country. Moreover, there is established clinical practice in ICU and surgical environments that leads to regular use of controlled IV administration devices, like burette set.
Is the Burette Set Market Developing in India?
The market for burette set in India is undergoing transformation attributed to the development and growing nature of the healthcare sector in India, along with government incentives towards producing local medical equipment (such as, Production Linked Incentive Scheme, Scheme for Promotion of Medical Device Parks, and Make in India). Regulations from the Central Drugs Standard Control Organization have necessitated proper licensing and quality of infusion sets, making them more efficient and widely used. However, Production Linked Incentive Scheme and medical device parks are being established in order to manufacture more devices within the country and reduce import dependency. Moreover, expansion in hospital infrastructure and rising demands for IV therapies in urban and tier-2/3 hospitals are contributing to the adoption of burette sets, thus making India a promising growth market.
|
Region
|
Key Regulatory Bodies |
Framework |
Impact on Market |
|
North America |
U.S. Food and Drug Administration, Health Canada |
Class II medical devices under 510(k); Medical Devices Regulations (Canada) |
High compliance standards ensure product safety and quality; longer approval timelines but strong market trust |
|
Europe |
European Commission |
Medical Device Regulation (EU) 2017/745 (MDR); CE marking |
Strict clinical evaluation and post-market surveillance increase entry barriers but enhance product reliability |
|
Asia Pacific |
Central Drugs Standard Control Organization, National Medical Products Administration, Pharmaceuticals and Medical Devices Agency |
Medical Devices Rules (India), NMPA registration (China), PMDA approval (Japan) |
Diverse regulatory frameworks; improving harmonization supports faster approvals and regional manufacturing growth |
|
Latin America |
ANVISA, COFEPRIS |
Device registration and local compliance standards |
Moderate regulatory complexity; growing standardization improving market entry and imports |
|
Middle East & Africa |
Saudi Food and Drug Authority, South African Health Products Regulatory Authority |
Import-based approvals; reliance on EU/US certifications |
Developing frameworks; dependence on international approvals enables faster product entry but limited local manufacturing |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
How is growing adoption of DEHP-free and safety-compliant materials creating new growth opportunities in the global burette set market?
The use of DEHP-free and conformable products will open up new avenues for expansion within the burette set market, owing to the fact that more products are aligned to comply with regulatory policies aimed at ensuring patient safety. DEHP, which is a common component used in making PVC IV tubing and infusion sets, has come under scrutiny owing to toxicity issues and the potential for migration as well as being considered a reproductive toxin. This has led to health authorities around the world seeking to reduce and substitute DEHP in infusion products. For instance, under the European regulatory framework, DEHP usage is restricted in certain medical devices, and efforts are being made to replace its usage with other options by 2030, necessitating changes in infusion disposable products, including burette sets. (Source: EcoMundo)
Similarly, healthcare providers are gradually leaning towards using non-DEHP and non-PVC infusions to minimize patient exposure risks, especially in vulnerable groups such as neonates and chronically ill patients (Source: Lars Medicare). These trends are encouraging innovations in material sciences (such as alternative polymers, co-extruded tubing), which have led to higher-end product positioning, allowing new income streams for burette manufacturers that could provide regulatory-compliant, safer, and unique burette sets.
Market Players, Key Development, and Competitive Intelligence

To learn more about this report, Request Free Sample
Key Developments
- In December 2025, ICU Medical, Inc. issued a recall for their IV Gravity Burette Sets after detecting performance problems, with the regulators considering this incident to be one of the most severe cases because it poses a threat to life or health. It is apparent that there are increased regulations on IV infusion sets, motivating hospitals to purchase burette sets that have reliability and compliance with safety standards.
- In September 2025, BD released a warning about performance problems in some infusion sets, the problem was classified by the regulatory bodies as one of the most severe recalls because of the likelihood of harm to individuals using the product. This event illustrates the tightening of regulatory requirements on quality control in intravenous infusion systems.
Competitive Landscape
The global burette set market will likely stay highly competitive yet fragmented comprising both leading brands that offer infusion therapy products worldwide along with many regional and local players offering their own versions. Competition is mainly influenced by price factors, reliability, and clinical safety of the products. The market leaders are trying to develop product variants that offer better flow control, reduced risks of contamination, ease of use, while maintaining their competitive edge when it comes to prices, which is crucial for mass consumption. The competition is also evident in public procurement processes based on tenders, especially in emerging nations, where the domestic manufacturing units have an upper hand due to their competitive pricing. Key focus areas include:
- Safety-enhanced designs (anti-reflux valves, air-elimination filters, needle-free systems)
- Cost optimization and large-scale manufacturing for tender-based supply
- Expansion in emerging markets (India, Southeast Asia, Africa)
- Integration with smart infusion and digital monitoring systems
- Strategic collaborations with hospitals and distributors for volume growth
Market Report Scope
Burette Set Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 620 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7.4% | 2033 Value Projection: | USD 1,020 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
B. Braun, Baxter International, Inc., PL Medical, Wellmed International Industries Pvt. Ltd., Merck, Thermo Fisher Scientific, Global Medikit Ltd., Angiplast Pvt. Ltd., APEX Medical Devices, and BQ Plus Medical Co. Ltd. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Analyst Opinion (Expert Opinion)
- In the coming years, the prospects of the burette set market is anticipated to continue steady and innovative, backed by the growing focus on safety and accuracy in administration as well as the treatment of pediatric patients. Although the conventional gravity flow burette sets will continue to gain traction among the cost-sensitive healthcare markets, the trend is gradually moving towards more efficient systems that include closed system IV sets, valveless designs, and smart infusion pumps. The developing nations will continue to play an important role in driving market growth.
- The biggest opportunity would come in the ≤100 mL capacity range, especially in the pediatric and neonatal segments in emerging economies like India and China where a high birth rate, improvements in Neonatal Intensive Care Unit (NICU) equipment, and government-led expansion in health care will create significant demand for precise low-volume infusion devices. Also, public hospitals and healthcare facilities in tier-2 and tier-3 towns have considerable possibilities because of bulk purchasing and reduction in medicine dosage errors.
- For gaining any competitive advantage in the market, companies need to emphasize innovations and product differentiation, through the creation of anti-reflux valves, precise flow regulators, and safety systems. From a cost-based competitive perspective, firms will need to optimize their activities from a cost efficiency standpoint and manufacturing process viewpoint. The collaboration with hospitals and group purchasing organizations (GPOs), local manufacturing capability, regulatory convergence, as well as product bundling in the broader IV Therapy product range will further enhance market positioning.
Market Segmentation
- Capacity Insights (Revenue, USD Mn, 2021 - 2033)
- ≤100 mL
- 101–150 mL
- >150 mL
- Others
- Material Insights (Revenue, USD Mn, 2021 - 2033)
- PVC
- DEHP-free PVC
- Others
- Patient Type Insights (Revenue, USD Mn, 2021 - 2033)
- Pediatric / Neonatal
- Adult
- End User Insights (Revenue, USD Mn, 2021 - 2033)
- Hospitals
- Ambulatory Surgical Centers (ASCs)
- Specialty Clinics
- Others
- Distribution Channel Insights (Revenue, USD Mn, 2021 - 2033)
- Direct / Tender-based Procurement
- Distributors and Wholesalers
- Others
- Regional Insights (Revenue, USD Mn, 2021 - 2033)
- North America
- U.S.
- Canada
- Latin America
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Europe
- Germany
- U.K.
- Spain
- France
- Italy
- Russia
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Australia
- South Korea
- ASEAN
- Rest of Asia Pacific
- Middle East
- GCC Countries
- Israel
- Rest of Middle East
- Africa
- South Africa
- North Africa
- Central Africa
- North America
- Key Players Insights
- Braun
- Baxter International, Inc.
- PL Medical
- Wellmed International Industries Pvt. Ltd.
- Merck
- Thermo Fisher Scientific
- Global Medikit Ltd.
- Angiplast Pvt. Ltd.
- APEX Medical Devices
- BQ Plus Medical Co. Ltd.
Sources
Primary Research Interviews
- Hospital procurement managers and ICU clinicians involved in infusion therapy and critical care fluid administration
- Medical device executives and product managers from infusion set manufacturers
- Pediatricians and neonatologists using burette sets for controlled IV fluid delivery in neonatal and pediatric care
- CROs and regulatory consultants involved in medical device approvals and infusion device validation
Stakeholders
- Burette Set Manufacturers & Infusion Therapy Device Providers (e.g., infusion set manufacturers, disposable medical device companies)
- End-use Sectors:
- Hospitals and Clinics
- Ambulatory Surgical Centers (ASCs)
- Specialty and Pediatric Care Centers
- Home Healthcare Settings
- Regulatory & Health Bodies: U.S. Food and Drug Administration (FDA) – classification and approval of infusion therapy devices (Class II medical devices), European Commission – Medical Device Regulation (EU MDR 2017/745) for infusion and disposable devices, World Health Organization – guidelines on injection safety and infusion practices, Centers for Disease Control and Prevention – infection control and IV therapy safety guidelines
Databases
- FDA Medical Device Databases – infusion set and IV administration device classification
- EUDAMED – EU database for medical device registration and compliance tracking
- WHO Global Health Observatory – hospital infrastructure and patient safety indicators
- OECD Health Statistics – healthcare expenditure and hospital utilization trends
Magazines
- Medical Device Network – infusion device innovations and regulatory updates
- Healthcare Packaging – trends in sterile and single-use medical devices
- MedTech Dive – developments in infusion systems and hospital devices
Journals
- Journal of Infusion Nursing
- Clinical Nursing Research
- International Journal of Medical Devices
Newspapers
- Financial Times – healthcare infrastructure and medical device investments
- The Wall Street Journal – hospital procurement and medtech developments
- The Economic Times (India) – domestic medical device manufacturing and exports
- Business Standard (India) – healthcare policy and device market trends
Associations
- Infusion Nurses Society (INS)
- Association for the Advancement of Medical Instrumentation (AAMI)
- International Federation of Medical and Biological Engineering (IFMBE)
- Advanced Medical Technology Association (AdvaMed)
Public Domain Sources
- National Institutes of Health (NIH) – infusion therapy and clinical safety studies
- Centers for Disease Control and Prevention (CDC) – IV safety and infection prevention data
- European Commission – medical device regulations and healthcare initiatives
- Ministry of Health and Family Welfare (India) – hospital infrastructure and medical device policies
Proprietary Elements
- CMI Data Analytics Tool
- Proprietary CMI Existing Repository of information for last 10 years.
Share
Share
About Author
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients