The global distraction osteogenesis devices market size is expected to reach USD 364 Mn in 2026 and is projected to grow at a compound annual growth rate (CAGR) of about 6% during the forecast period (2026-2033), reaching USD 548 Mn by 2033. This growth is attributable to increasing prevalence of craniofacial and orthopedic deformities worldwide and rising adoption of advanced minimally invasive surgical techniques.
The global distraction osteogenesis market is poised to record steady growth during the forecast period. This is primarily due to rising prevalence of orthopedic disorders, growing geriatric population prone to bone-related disorders, and advancements in surgical techniques and medical devices. In addition, innovations in distraction osteogenesis devices and rising need for advanced bone lengthening technologies are supporting market expansion.
Distraction osteogenesis (DO), also called callus distraction, callotasis, osteodistraction, or distraction histogenesis, is a surgical process used to reconstruct skeletal deformities and lengthen bones. Depending on the site of application, DO devices can be classified as mandibular (lower jaw), maxillary (upper jaw), alveolar (tooth-bearing regions of the jaws), palatal (roof of the mouth), or craniofacial (skull and facial bones).
Distraction osteogenesis devices are used in orthopedic, maxillofacial, and oral surgery. They are indicated for treating bone defects, correcting limb length discrepancies, and managing craniofacial anomalies such as cleft palate, craniosynostosis, hemifacial microsomia, and other congenital or acquired skeletal deformities. Thus, rising cases of these disorders are expected to boost growth of distraction osteogenesis devices market during the forecast period.
Growing awareness and acceptance of distraction osteogenesis techniques among orthopedic and craniofacial surgeons are driving sales of distraction osteogenesis devices. These procedures are gaining traction due to their improved functional and aesthetic outcomes compared with traditional methods in selected cases. Customization is becoming increasingly important in the distraction osteogenesis industry. The use of 3D printing, advanced imaging, and surgical planning technologies allows the creation of patient‑specific devices designed for each person's anatomy, which can improve treatment outcomes and encourage wider adoption.
|
Current Event |
Description and its Impact |
|
Technological Advancements and AI Integration in Medical Devices |
|
|
Demographic Shifts and Rising Healthcare Demand |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
By device type, mandibular distraction devices segment is projected to lead the market, generating a revenue share of 34% in 2026. This is mostly due to their extensive use in correcting congenital and acquired mandibular deformities, increasing prevalence of craniofacial anomalies, and rising awareness among surgeons about minimally invasive corrective procedures.
Mandibular distraction devices are favored due to their precision, ease of application, and ability to stimulate bone growth effectively, which enhances both functional and aesthetic outcomes. In addition, technological advancements such as motorized and bioresorbable devices are also driving adoption in clinical settings.
According to Coherent Market Insights’ new distraction osteogenesis devices market analysis, external distractors are expected to account for a market share of 60% in 2026. This is mostly because of their versatility in complex limb-lengthening and multi planar corrections. External distractors are favored worldwide due to their adaptability in treating a variety of craniofacial conditions, including severe micrognathia, hemifacial microsomia, and post-traumatic mandibular defects.
The design of external distractors allows for precise control over bone movement in multiple planes, making them ideal for complex reconstructions. In addition, external devices are often preferred in pediatric and high-risk cases where internal distractors may pose surgical or anatomical limitations, contributing to their strong adoption among craniofacial surgeons globally.
For instance, in clinical series, external distractors were used in approximately 54% of mandibular distraction osteogenesis cases, compared with 46% for internal devices. This indicates a modest preference for external systems in complex or pediatric presentations. As a result, the external distractors segment is likely to retain its market dominance in the coming years.
To learn more about this report, Request Free Sample
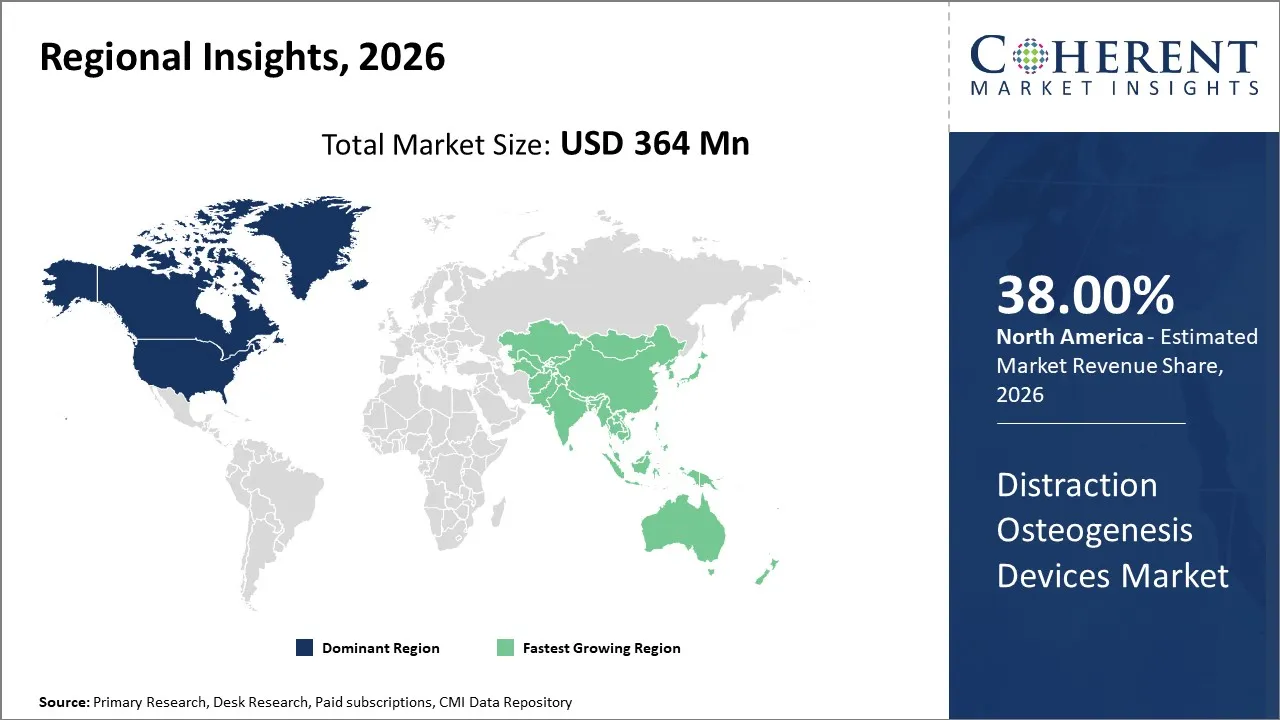
North America is expected to lead the global distraction osteogenesis devices market, capturing a share of 38% in 2026. This is driven by a high volume of reconstructive facial procedures in the United States and the presence of advanced care facilities. According to the American Society of Plastic Surgeons’ 2023 procedural data, there were about 1.02 million reconstructive plastic surgery procedures performed in the U.S., including approximately 52,868 maxillofacial surgeries, a category where distraction osteogenesis is often applied.
Specialized craniofacial centers, such as the Cleft and Craniofacial Anomalies Clinic at American Family Children's Hospital and Children's Hospital of Wisconsin, provide comprehensive treatment for congenital and acquired facial defects, supporting market growth. High procedural volumes, robust clinician expertise, and strong research support drive the adoption of innovative techniques, including magnetically actuated distractors and precision-controlled bone regeneration solutions.
Favorable reimbursement policies and broad insurance coverage further enhance patient access, while ongoing R&D and collaboration between manufacturers and hospitals promote advanced device materials and minimally invasive procedures. Rising use of distraction osteogenesis in orthodontics is also expected to contribute to market expansion in North America during the forecast period.
Asia Pacific is emerging as the fastest growing distraction osteogenesis market, holding a global market share of 22% in 2026. This is mostly due to rapid expansion of healthcare infrastructure, increasing patient awareness of reconstructive surgical options, and rising demand for advanced orthopaedic solutions. Rising incidence of traumatic injuries from accidents and a large population with congenital bone deformities are increasing demand for distraction techniques that can correct limb length discrepancies and skeletal asymmetries.
Another key growth driver in Asia Pacific is the rise of medical tourism and cost-competitive care, attracting international patients to countries such as South Korea and India for distraction osteogenesis treatments at comparatively lower prices. Local manufacturers are also developing affordable distraction devices that meet international quality standards, making these treatments more accessible across urban and semi urban markets. Rising adoption of distraction osteogenesis in dentistry is also fostering revenue growth in the region.
Several global medical technology companies continue to strengthen their presence in the region by establishing local subsidiaries and operations to better serve customers as well as expand geographic reach. For instance, in 2024, Medtronic expanded its investment in the Asia Pacific region by launching its first Robotics Experience Studio in Singapore. It is designed to help hospitals in Southeast Asia use advanced robotics and AI more quickly and to work closely with local healthcare providers and partners.
The U.S. distraction osteogenesis devices market is expected to maintain its leading position globally on account of strong healthcare infrastructure, high procedural volumes, and wide availability of advanced surgical solutions. Increasing demand for corrective bone lengthening procedures, rising incidence of trauma-related fractures, and growing preference for minimally invasive orthopedic and craniofacial surgeries are also supporting market expansion in the region.
A key trend in the U.S. distraction osteogenesis devices market is the fast adoption of advanced distraction systems, such as motorized and precision-controlled internal distractors, which provide better comfort and outcomes for patients. These innovative devices allow surgeons to achieve accurate bone regeneration with fewer complications and shorter recovery times, making them preferred in both specialized hospitals and ambulatory surgical centers across the country.
The distraction osteogenesis devices market in China is projected to grow rapidly during the forecast period. This is mostly due to expanding healthcare access, increasing surgical volumes, and supportive government initiatives for advanced medical care. The rise in road traffic accidents and sports injuries has led to greater demand for effective orthopedic reconstruction techniques, while an expanding middle class is increasingly seeking corrective procedures for congenital deformities and skeletal discrepancies.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 364 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 6% | 2033 Value Projection: | USD 548 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Johnson & Johnson, Zimmer Biomet Holdings, Inc., Stryker Corporation, KLS Martin Group, Acumed LLC, OsteoMed, and Titamed. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Technological Advancements in Distraction Osteogenesis Device Designs Continuous innovation, such as motorized and magnetic distractors, enhanced biocompatible materials, remote adjustment systems, and smart sensor integration, improves precision as well as enhances patient comfort and outcomes. This makes procedures more efficient and appealing to clinicians and patients alike, thereby creating growth opportunities for the distraction osteogenesis devices market.
Expanding Clinical Applications Traditionally distraction osteogenesis techniques were used for limb lengthening. However, today their scope is expanding significantly. They are now being applied in facial reconstruction, spinal deformity correction, bone defect repair, and even dental/maxillofacial surgeries. These expanding applications are expected to open new revenue streams for manufacturers of distraction osteogenesis devices during the assessment period.
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients