Global Chiba Needle Market Size and Forecast – 2026 to 2033
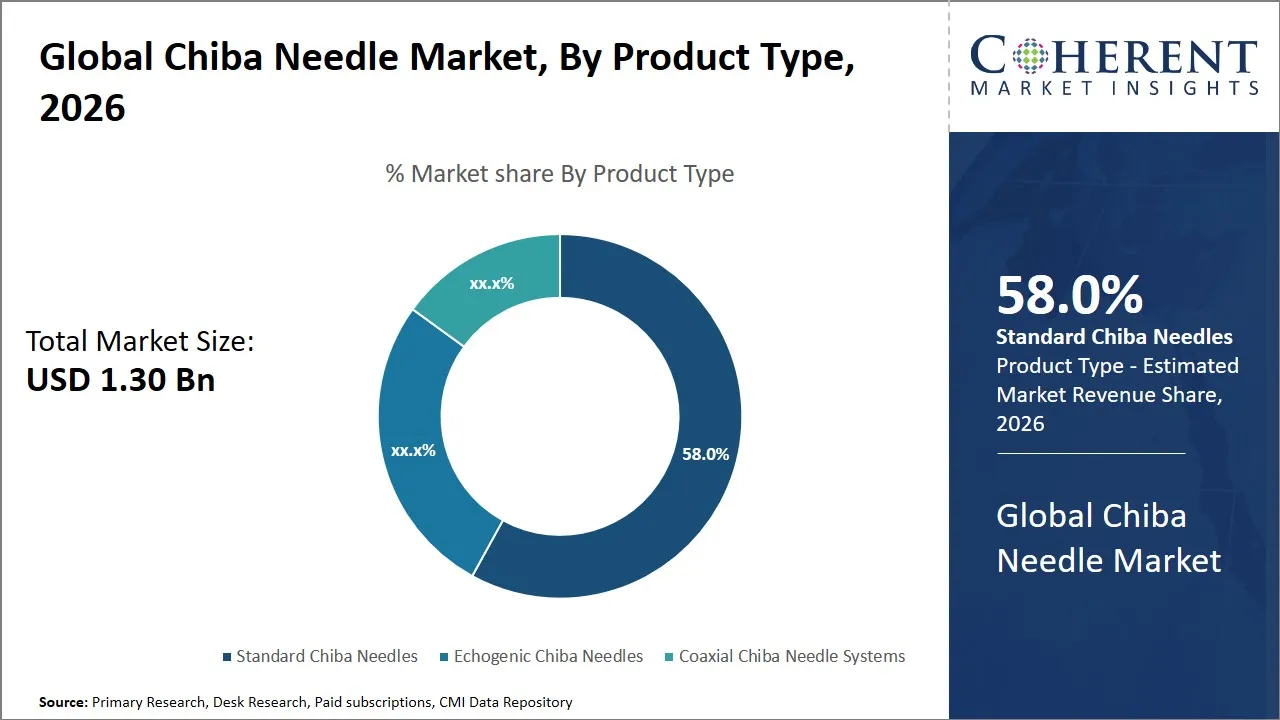
The global Chiba needle market is estimated to be valued at USD 1.30 Bn in 2026 and is expected to reach USD 2.00 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 6.35% from 2026 to 2033. The consistent rise in demand for Chiba needle mirrors their widespread use across healthcare, oncology diagnostics, veterinary medicine, research laboratories, and specialized industrial applications. This trend is fueled by technological progress (such as enhanced needle design, improved imaging compatibility, coatings and materials innovation, safety features, and single-use and sterile innovations) and the broadening of their applications within medical, industrial, and specialized areas.
Key Takeaways of the Global Chiba Needle Market
- The standard Chiba needles segment is expected to dominate the global Chiba needle market by product type, accounting for approximately 58.0% of the total market share in 2026.
- The disposable segment is expected to dominate the global Chiba needle market by usability, accounting for approximately 70.0% of the total market share in 2026.
- The ultrasound-guided procedures segment is expected to dominate the global Chiba needle market by procedure guidance, accounting for approximately 62.0% of the total market share in 2026.
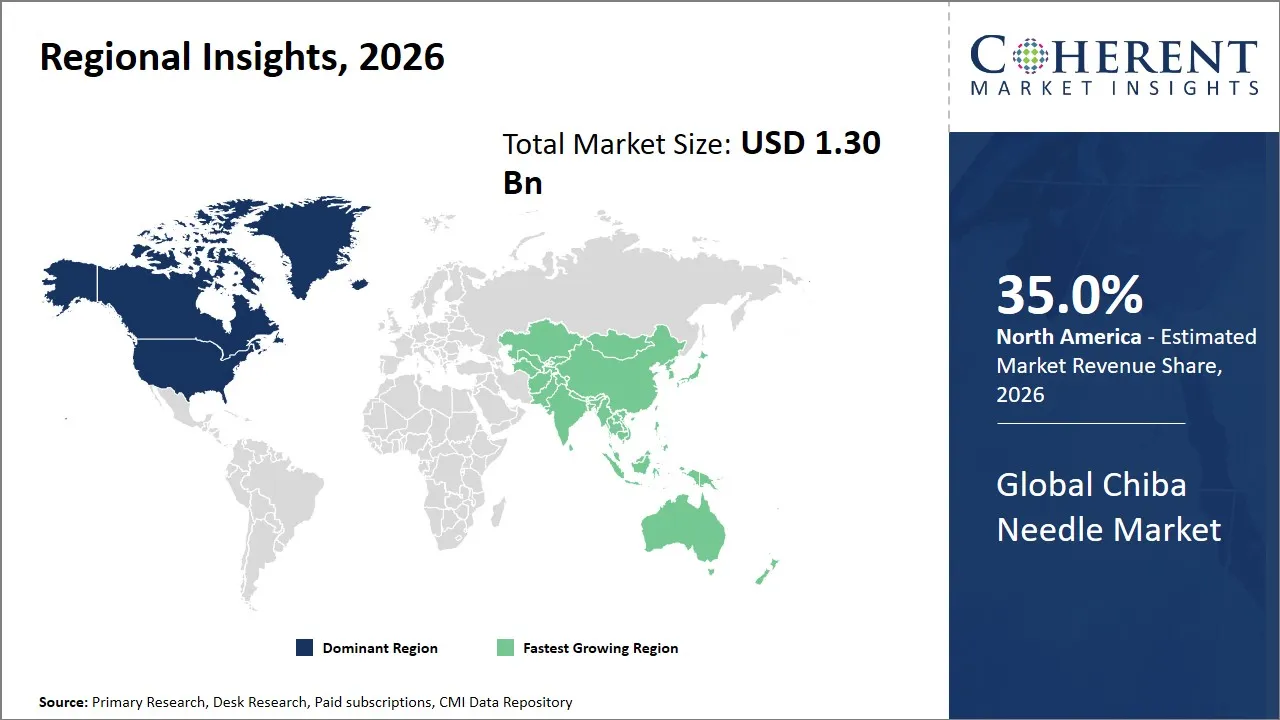
- North America is expected to dominate the global Chiba needle market in 2026, accounting for approximately 35.0% of the total market share.
- Asia Pacific is anticipated to witness the fastest growth, holding a market share of 22.0% in 2026.
Market Overview
- Chiba needles are highly specialized needles used in image-guided diagnostic procedures. These needles are particularly used in fine-needle aspiration and biopsy of body parts such as the liver, lungs, and kidneys. These needles are highly flexible and precise in accessing hard-to-reach areas of the body.
- The market is growing steadily due to an increase in cancer and other chronic disease cases across the world. This is resulting in an increase in biopsy cases. In addition, an increasing preference for early diagnosis is further contributing to the growth of Chiba needle.
- Technological advancements in imaging devices such as ultrasound and CT are contributing to an increase in accuracy in diagnostic procedures. This is further resulting in an increase in the number of Chiba needle used in image-guidance.
- In addition, an increase in healthcare infrastructure and outpatient diagnostic procedures, especially in emerging markets, is also contributing to the Chiba needle market growth.
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
Advancements in Image-Guided Biopsy Technologies |
|
|
Rising Prevalence of Cancer and Chronic Diseases |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Why Does the Standard Chiba Needles Segment Dominate the Global Chiba Needle Market in 2026?
The standard Chiba needles segment is anticipated to hold a market share of 58.0% in 2026, attributed to their versatility, reliability, and clinical acceptance. These are commonly used for routine procedures such as biopsies, fluid drainage, etc., which require precision as well as ease of use. The standard design of Chiba needles reduces the complexity of the procedure, making them the most sought-after product type by clinicians in various healthcare facilities.
Cost-effectiveness is another factor that can be associated with the success of standard Chiba needles in the market, as they are less expensive compared to other advanced product types such as echogenic or coaxial needles. These are particularly useful in developing countries, where healthcare facilities are limited in their budget. The use of standard Chiba needles is possible with various imaging modalities, making them a versatile product type for various clinical procedures.
For instance, in October 2025, Olympus Corporation unveiled its SecureFlex single-use fine needle biopsy device. This new tool is engineered to boost precision and facilitate high-quality tissue sampling during minimally invasive procedures, including endoscopic ultrasound-guided biopsies. The device features an advanced needle design, which aims to improve access to intricate anatomical areas and assist in the accurate diagnosis of ailments such as pancreatic cancer.
Why Does the Disposable Segment Dominate the Global Chiba Needle Market in 2026?
The disposable segment is anticipated to hold a market share of 70.0% in 2026, primarily because of the strict guidelines and regulations regarding the prevention of infections. Disposable needles are the best option in this case, as they do not pose the risk of cross-contamination or hospital-acquired infections. This is particularly true in the case of biopsy and diagnostic tests, where sterility is required at the highest level. As a result, hospitals and clinics are using these types of needles more than the reusable ones. Moreover, the recent developments in the manufacturing process of these needles have improved their performance, which is now at par with the reusable ones.
Another factor that has increased the demand for the disposable type of Chiba needles is the advantages they provide in the process of workflow efficiency. This type of needle has reduced the risk of liability and has increased the confidence of the doctors using them. As the infrastructure is developing in the healthcare sector, the demand for this type of needle is increasing, which is providing the disposable type of needle with a high market share.
Ultrasound-Guided Procedures Segment Dominate the Global Chiba Needle Market
The ultrasound-guided procedures segment is anticipated to hold a market share of 62.0% in 2026. Ultrasound guidance has the potential to provide real-time visualization of soft tissues and vascular structures. This enables precise needle placement in procedures such as biopsies, fluid aspirations, and nerve blocks. This is done without the need to expose the patient to ionizing radiation. Ultrasound guidance is portable and user-friendly, which increases its popularity in hospitals, clinics, and bedside settings. Advances in ultrasound imaging are also helping to improve the accuracy of the procedure and reduce the risk of complications.
Moreover, ultrasound guidance is the most cost-effective option compared to CT or MRI guidance. This is because ultrasound guidance has the lowest equipment investment and operating costs. Additionally, ultrasound guidance has the potential to reduce patient preparation time and eliminate the need to consider precautions related to radiation. As the preference for using minimally invasive techniques increases, ultrasound-guided procedures are becoming the standard of care, which is increasing its share in the procedure guidance segment of the global Chiba needle market.
Regulatory Landscape and Compliance Requirements in the Global Chiba Needle Market
|
Region |
Regulatory Authority |
Key Regulation |
Impact on Chiba Needle Market |
|
North America |
U.S. Food and Drug Administration |
510(k) Clearance Pathway, Quality System Regulation (QSR), Medical Device Labeling Requirements |
Ensures high safety and quality standards for biopsy needles, accelerates adoption through standardized approvals, and builds strong end-user trust |
|
Europe |
European Medicines Agency |
Medical Device Regulation (MDR), CE Marking Requirements, Post-Market Surveillance |
Strengthens product safety, traceability, and compliance, encouraging adoption of high-quality minimally invasive devices |
|
Asia Pacific |
Pharmaceuticals and Medical Devices Agency |
Regional device approval frameworks, Good Manufacturing Practices (GMP), evolving regulatory harmonization |
Supports faster product approvals and promotes adoption of advanced biopsy devices in emerging markets |
|
Middle East & Africa |
Saudi Food and Drug Authority |
Device Registration Guidelines, Reliance Pathways, WHO-aligned standards |
Improves access to safe medical devices and supports gradual regulatory standardization across the region |
|
Latin America |
Brazilian Health Regulatory Agency |
Medical Device Registration, Local Compliance Requirements, Import Regulations |
Facilitates market entry while ensuring product safety, though regulatory complexity may impact timelines |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Regional Insights
To learn more about this report, Request Free Sample
North America Chiba Needle Market Analysis and Trends
The North America region is projected to lead the market with a 35.0% share in 2026, attributed to the well-established medical device sector, the high standard of healthcare facilities, and the high investment in research and development activities. The presence of a well-established supply chain system and the implementation of strict regulations ensure the high quality and safety of the products, which in turn creates trust among the end users.
Moreover, the government's initiatives in the U.S. and Canadian markets are favorable, which accelerates the launch of the products. Companies like Becton Dickinson & Co., Medtronic, and Teleflex Incorporated are some of the key players in the market, who are well-established in the region and are constantly developing the technology and adding more products to their portfolio. Additionally, the region is also focusing on the development of minimally invasive surgical techniques using needles.
For instance, the U.S. Preventive Services Task Force recommends routine cancer screening programs such as lung cancer screening using low-dose computed tomography scans for high-risk populations. These screening programs are expected to enhance the detection of suspicious lesions, which in turn calls for image-guided biopsy procedures. This further calls for the adoption of fine needles such as Chiba needles in the U.S.
Asia Pacific Chiba Needle Market Analysis and Trends
Asia Pacific is anticipated to witness the fastest growth, holding a market share of 22.0% in 2026, attributed to the urbanization, healthcare expenditure, and lifestyle conditions such as chronic diseases, which require minimally invasive medical devices, creating strong growth prospects the market. Countries in Asia Pacific, such as China, Japan, India, and South Korea, have witnessed strong growth in healthcare infrastructure and the adoption of high-tech medical devices. Government initiatives to improve healthcare infrastructure and support local manufacturing are creating an environment conducive to the Chiba needle market.
In addition, healthcare experts in Asia Pacific are also becoming increasingly familiar with advanced needle technology. Some of the key companies in the Chiba needle market are Terumo Corporation, Nipro Corporation, and B. Braun Melsungen AG. These companies are establishing R&D centers in Asia Pacific, which has helped them enter the Chiba needle market. Trade conditions are also favorable in Asia Pacific, with low duties facilitating easy movement of products in Asia Pacific.
Chiba Needle Market Outlook for Key Countries
How is the U.S. Helping in the Growth of the Chiba Needle Market?
The U.S. is a leader in the Chiba needle market on a global scale, attributed to the country’s advanced healthcare system and the demand for minimally invasive surgical procedures. Companies such as Becton Dickinson & Co. and Teleflex have opened research centers and production plants in the country. The country’s focused health programs and the FDA have instilled confidence in the safety and efficacy of the products. The availability of hospitals and clinics provides the demand for the product. The country’s academic institutions have been working with the companies in the development of the product.
For instance, in January 2026, Olympus Corporation unveiled its SecureFlex single use fine needle biopsy device for the market in the U.S. This device is engineered to offer greater precision and adaptability during endoscopic ultrasound-guided procedures. The goal is to elevate the quality of tissue samples, thereby aiding minimally invasive diagnostics. This introduction bolsters Olympus's offerings in advanced biopsy tools and solidifies its position within the U.S. interventional endoscopy sector.
How is Germany Helping in the Growth of the Chiba Needle Market?
Germany is recognized as an important center for medical device engineering and innovation in Europe. Stringent quality requirements and healthcare infrastructure in Germany create demand for high-quality Chiba needles. B. Braun Melsungen AG and Medtronic have an established presence in Germany, contributing to the development of needle technologies and comprehensive product offerings. The developed hospital infrastructure in Germany, coupled with the trend toward minimally invasive techniques, also supports demand. Germany also enjoys an effective export platform, acting as a gateway for medical device exports in Europe.
Key Drivers for the Growth of the China Chiba Needle Market
The Chiba needle market in China is driven by the increasing hospital infrastructure, medical tourism, and government initiatives encouraging the production of medical equipment locally. Companies like Mindray Medical, along with subsidiaries of global players, are increasing their production and R&D capabilities to meet the demand for Chiba needles in cancer and diagnostic treatments.
Regulatory policies are also encouraging the approval process for the entry of products into the market. Furthermore, the emphasis of the China government on providing healthcare facilities in rural areas is likely to create opportunities in the China Chiba needle market. The country is strategically located to benefit from the import-export dynamics.
India Chiba Needle Market Trends
The Chiba needle market in India is showing rapid growth due to the increasing healthcare infrastructure, prevalence of chronic diseases, and the large number of patients who require medical interventions using Chiba needles. The government policies, such as “Make in India,” are also driving the market, with investments from various multinational companies such as Teleflex and Terumo. The increasing number of private hospitals and diagnostic centers is also contributing to the growth of the market.
Additionally, the increasing awareness of healthcare professionals about Chiba needle is also driving the market. The strategic location of the Indian market in the global supply chain, along with the improvements in the regulatory process, are also playing an important role in driving the market.
Healthcare Infrastructure Expansion and Procedure Volume Trends Supporting Growth in the Global Chiba Needle Market
|
Region |
Healthcare Infrastructure Trend |
Procedure Volume Trends |
Key Drivers |
Impact on Market |
|
North America |
Highly developed hospital networks, advanced diagnostic imaging centers, strong outpatient care infrastructure |
High and steadily growing volume of biopsy and interventional radiology procedures |
Aging population, high cancer incidence, advanced diagnostic capabilities |
Sustained demand for high-precision Chiba needles in routine and complex procedures |
|
Europe |
Well-established public healthcare systems with widespread access to diagnostic facilities |
Stable to increasing biopsy procedure volumes supported by early screening programs |
Government healthcare funding, preventive care focus, aging demographics |
Consistent utilization of Chiba needles across hospitals and specialty clinics |
|
Asia Pacific |
Rapid expansion of hospitals, diagnostic centers, and ambulatory care facilities |
Fast-growing procedure volumes, particularly in emerging economies |
Rising healthcare expenditure, improving access, increasing disease burden |
Strong growth opportunity and rising adoption of Chiba needles |
|
Latin America |
Developing healthcare infrastructure with improving access to diagnostic services |
Moderate but increasing biopsy and minimally invasive procedure volumes |
Urbanization, healthcare investments, growing awareness |
Gradual increase in demand for cost-effective Chiba needle solutions |
|
Middle East & Africa |
Expanding healthcare infrastructure, particularly in urban areas and GCC countries |
Emerging but growing procedure volumes |
Government investments, improving healthcare access, rising chronic diseases |
Expanding market potential with increasing adoption of basic biopsy tools |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Market Players, Key Developments, and Competitive Intelligence

To learn more about this report, Request Free Sample
Key Developments- In February 2025, Limaca Medical introduced its Precision-GI automated endoscopic biopsy device to the U.S. market. This launch was backed by both U.S. FDA clearance and CMS reimbursement approval. The device is designed to improve tissue acquisition. It does this via a motorized cutting mechanism, which, in turn, boosts diagnostic accuracy during gastrointestinal procedures.
- In September 2024, Argon Medical Devices introduced the Intara Introducer Sheath and TLAB Transvenous Liver Biopsy System, broadening its offerings in interventional radiology. This new system aims to streamline liver biopsy processes, offering better vascular access and more sophisticated sampling methods. The launch underscores the ongoing progress in biopsy technology, which is key to more accurate diagnoses.
- In May 2024, Cook Medical launched EchoTip ClearCore EUS Biopsy Needle. This new tool aims to improve the precision of gastrointestinal lesion sampling by offering better tissue acquisition. The launch underscores the continuing evolution of biopsy and aspiration needle technologies, which are essential in image-guided procedures. These advancements, seen across the field of interventional radiology, are helping to boost both accuracy and efficiency. Consequently, this is supporting the expansion of the Chiba needle market.
Top Strategies Followed by Global Chiba Needle Market Players
|
Player Type |
Strategic Focus |
Examples |
|
Established Market Leaders |
Established market leaders emphasize innovation and development in terms of product portfolio through increased investments in R&D activities for developing biopsy needles that are highly precise, reliable, and minimally invasive. Their strategy also involves boosting their global footprint by forming strategic alliances and working with original equipment manufacturers and distribution channels in developing markets. |
In July 2025, Teleflex Incorporated announced advancements in its interventional access product portfolio, emphasizing enhanced needle precision and safety features for minimally invasive procedures, strengthening its global market position. |
|
Mid-Level Players |
Mid-level players are currently prioritizing cost-effective manufacturing processes, striving to maintain product quality in response to the needs of increasingly budget-aware consumers. Their strategies are centered on expanding their market reach, all while carefully controlling capital spending. |
In March 2025, Argon Medical Devices expanded its biopsy product offerings through strategic collaborations, focusing on affordable and efficient solutions for image-guided procedures in emerging markets. |
|
Small-Scale Players |
Small-scale players are honing in specialization and customization, leveraging advanced materials and manufacturing technologies. They also focus on improving their presence through strategic collaborations with other players to serve underserved markets. |
In September 2025, SOMATEX Medical Technologies introduced specialized biopsy needle solutions designed for targeted diagnostic applications, strengthening its niche positioning in precision-guided interventions. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Market Report Scope
Chiba Needle Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1.30 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 6.35% | 2033 Value Projection: | USD 2.00 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Cook Medical, Boston Scientific Corporation, BD, Terumo Corporation, Cardinal Health, Medtronic, Argon Medical Devices, Merit Medical Systems, Teleflex Incorporated, and Smith’s Medical |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Chiba Needle Market Dynamics
To learn more about this report, Request Free Sample
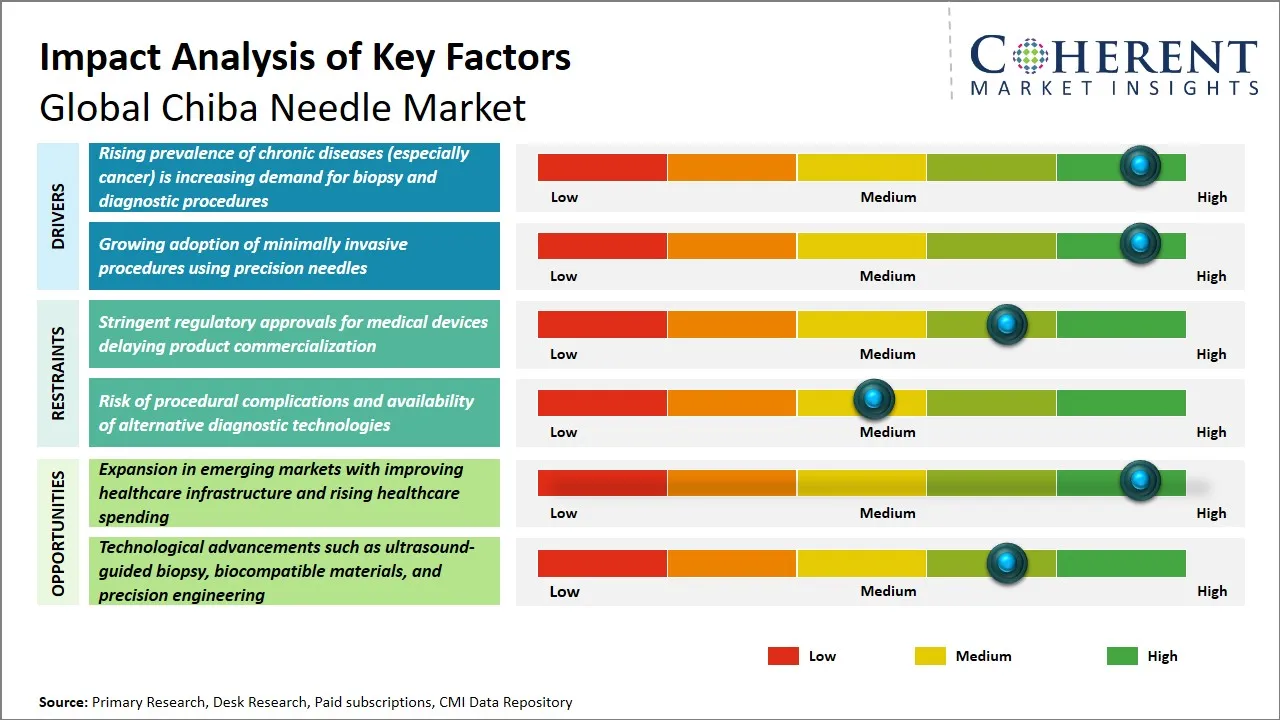
Global Chiba Needle Market Driver – Rising Prevalence of Chronic Diseases (Especially Cancer) is Increasing Demand for Biopsy and Diagnostic Procedures
The rise in the prevalence of chronic health conditions across the globe, such as cancer, has contributed greatly to the demand for advanced diagnostic tools such as Chiba needles. The prevalence of cancer is increasing globally. Consequently, the demand for minimally invasive biopsy procedures that enable the precise diagnosis of cancer and other chronic health conditions is on the rise. Chiba needles are precise and small in gauge. Therefore, they are crucial in the sampling of body tissues in areas that are hard to reach with minimal patient discomfort.
Furthermore, the increased trend in image-guided biopsy devices will also increase the need for these needles, in addition to other chronic illnesses that need tissue diagnosis. This trend is further accentuated by the awareness that healthcare practitioners have about the need for early diagnosis in improving patient outcomes. The rising trend in chronic illnesses is, therefore, an important factor that is contributing to the increased demand for Chiba needles.
For instance, the International Agency for Research on Cancer reports that roughly 20 million new cancer cases were identified worldwide in 2022. Forecasts suggest this number could exceed 35 million by 2050, highlighting a significant rise in the global cancer burden. This escalating incidence is driving the demand for early and accurate diagnostic approaches, which is, in turn, spurring the adoption of less invasive biopsy procedures.
Global Chiba Needle Market Opportunity - Expansion in Emerging Markets with Improving Healthcare Infrastructure and Rising Healthcare Spending
One of the major opportunities that is available in the global Chiba needle market is related to the growing healthcare infrastructure and healthcare expenditure in developing markets, especially in countries in Asia Pacific, Latin America, and parts of Africa. These markets are experiencing healthy growth in terms of overall economic development, which is leading to increased healthcare infrastructure and healthcare expenditure in terms of hospitals and outpatient surgical centers, who are end-users for Chiba needles.
In addition to that, increased government initiatives in terms of improving healthcare accessibility and overall healthcare quality, as well as awareness about minimally invasive diagnostic and therapeutic procedures, is also driving demand for technologically advanced medical devices like Chiba needles. The prevalence of chronic diseases and cancer in these markets is also driving demand for precise biopsy and fluid aspiration devices.
Additionally, as the levels of per capita income increase, and as there are more insurance cover options, patients will be able to access specialized medical care services, and this will result in a larger number of medical procedures that incorporate Chiba needles. The manufacturing partnerships in this region are also evolving, and this will result in the wider accessibility of Chiba needles, and this will increase the penetration of Chiba needles. As the medical infrastructure in developing countries continues to grow, and as more complex medical practices are put in place, there is a chance for Chiba needle manufacturers to penetrate this market through innovations in Chiba needles.
For instance, as per reports published by the World Bank, there is a rise in global healthcare expenditure. However, in emerging markets such as Asia Pacific, Latin America, and Africa, there is a rise in healthcare expenditure, and this is mainly because of the rise in income levels and the increase in government expenditure on healthcare infrastructure. The rise in healthcare expenditure is generating opportunities for hospitals, diagnostic centers, and minimally invasive surgeries, and this is generating opportunities for medical device adoption, i.e., Chiba needles.
Analyst Opinion (Expert Opinion)
- The Chiba needle market is growing due to an increase in cancer and other chronic diseases worldwide. This is leading to an increase in image-guided biopsy and diagnosis procedures.
- Advancements in interventional radiology and imaging technologies are also contributing to the growth of Chiba needles. These imaging technologies include ultrasound and CT imaging, which are enhancing accuracy and safety in image-guided procedures. This is causing an increase in the adoption of Chiba needles in liver, lung, and abdominal procedures.
- The strong trend in minimally invasive medical procedures is also contributing to the growth of Chiba needles. This is due to an emphasis on reducing patient trauma, recovery time, and costs of diagnosis.
- Reimbursement scenarios are also creating an opportunity for Chiba needles in developed countries. In addition, innovations in Chiba needles are also contributing to their growth. Innovations include advancements in needle tip and echogenic features. These are causing an increase in preference among clinicians.
Market Segmentation
- Product Type Insights (Revenue, USD Bn, 2021 - 2033)
- Standard Chiba Needles
- Echogenic Chiba Needles
- Coaxial Chiba Needle Systems
- Usability Insights (Revenue, USD Bn, 2021 - 2033)
- Disposable
- Reusable
- Procedure Guidance Insights (Revenue, USD Bn, 2021 - 2033)
- Ultrasound-Guided Procedures
- CT-Guided Procedures
- Fluoroscopy-Guided Procedures
- MRI-Guided Procedures
- Application Insights (Revenue, USD Bn, 2021 - 2033)
- Tissue Biopsy
- Fluid Aspiration
- Cytological Sampling
- Diagnostic Procedures
- Others
- End User Insights (Revenue, USD Bn, 2021 - 2033)
- Hospitals
- Diagnostic Imaging Centers
- Ambulatory Surgical Centers (ASCs)
- Specialty Clinics
- Others
- Regional Insights (Revenue, USD Bn, 2021 - 2033)
- North America
- U.S.
- Canada
- Latin America
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Europe
- Germany
- U.K.
- Spain
- France
- Italy
- Russia
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Australia
- South Korea
- ASEAN
- Rest of Asia Pacific
- Middle East
- GCC Countries
- Israel
- Rest of Middle East
- Africa
- South Africa
- North Africa
- Central Africa
- North America
- Key Players Insights
- Cook Medical
- Boston Scientific Corporation
- BD
- Terumo Corporation
- Cardinal Health
- Medtronic
- Argon Medical Devices
- Merit Medical Systems
- Teleflex Incorporated
- Smith’s Medical
Sources
Primary Research Interviews
Industry Stakeholders
- Chiba Needle Device Manufacturers
- Medical Device Component Suppliers
- Healthcare Providers (Hospitals & Interventional Radiology Centers)
- Distributors & Service Providers
- Regulatory & Compliance Expert
End Users
- Hospitals & Specialty Clinics
- Interventional Radiology Centers
- Ambulatory Surgical Centers (ASCs)
- Diagnostic Imaging Centers
Government and International Databases
- U.S. Food and Drug Administration – Medical device approvals & safety data
- European Medicines Agency – Medical device and clinical evaluation frameworks
- World Health Organization – Cancer burden & diagnostic insights
Trade Publications
- Medical Device Network
- MedTech Dive
- Fierce Biotech (Medical Devices section)
- Neurotech Reports
Academic Journals
- Journal of Vascular and Interventional Radiology
- European Radiology
- American Journal of Roentgenology
- Diagnostic and Interventional Radiology
Reputable Newspapers
- Financial Times – Global healthcare technology trends
- The Wall Street Journal – Medical technology investments and innovations
Industry Associations
- Advanced Medical Technology Association
- Society of Interventional Radiology
- Radiological Society of North America
Public Domain Resources
- World Health Organization – Disease prevalence and diagnostic guidelines
- Centers for Disease Control and Prevention – Chronic disease and cancer data
- National Institutes of Health – Research on minimally invasive diagnostics
- European Commission – Medical device regulatory frameworks
Proprietary Elements
- CMI Data Analytics Tool: Proprietary analytics tool to analyze real-time market trends, consumer behavior, and technology adoption in market
- Proprietary CMI Existing Repository of Information for Last 10 Years
Share
Share
About Author
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients