Global HbA1c Testing Device Market Size and Forecast – 2026 To 2033
The global HbA1c testing device market is expected to grow from USD 2.45 Bn in 2026 to USD 3.78 Bn by 2033, registering a compound annual growth rate (CAGR) of 7.7%. The market for global HbA1c testing devices is poised for significant expansion, fueled by the increasing need for long-term glycemic monitoring and diabetes management.
According to the World Health Organization, the number of people living with diabetes rose from 200 million in 1990 to 830 million in 2022. The prevalence has been rising more rapidly in low- and middle-income countries than in high-income countries. Moreover, in 2021, diabetes and kidney disease due to diabetes caused over 2 million deaths. In addition, around 11% of cardiovascular deaths were caused by high blood glucose.
(Source: World Health Organization)
Key Takeaways of the Global HbA1c Testing Device Market
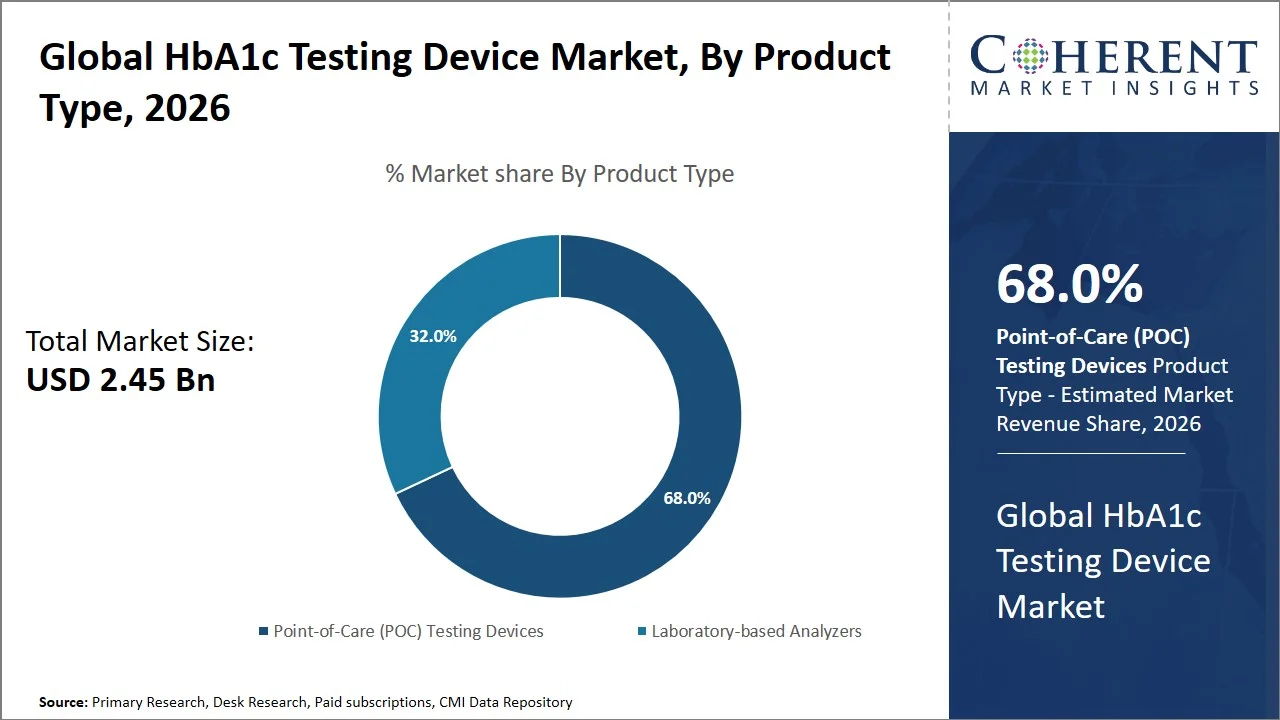
- Point-of-Care (POC) testing devices are projected to hold 0% of the global HbA1c testing device market share in 2026, making it the dominant product type segment, with North America at the forefront attributed to extensive use of point-of-care diagnostics and favorable regulation, supporting Clinical Laboratory Improvement Amendments (CLIA) waived testing (under the CLIA of 1988, regulated by the U.S. Centers for Medicare & Medicaid Services (CMS)). The devices enable prompt decision-making during the clinical visit, leading to improved outcomes in diabetes management. For instance, in the U.S., devices like the Afinion 2 Analyzer have found widespread application in physicians' clinics for delivering results within minutes, facilitating immediate treatment adjustments. (Source: Abbott)
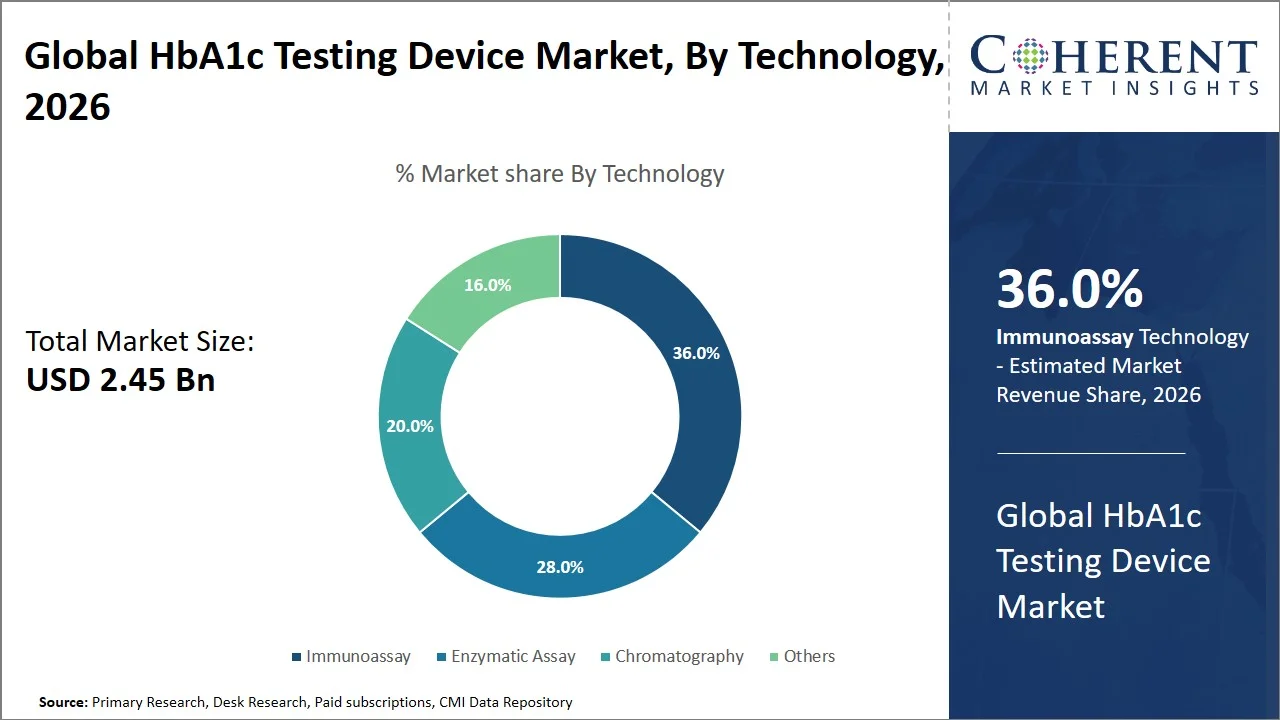
- Immunoassay is projected to hold 0% of the global HbA1c testing device market share in 2026, making it the dominant technology segment, with Europe taking the lead because of its well-developed laboratory network and standard diagnostic procedures. For instance, countries like Germany have structures that are based on statutory health insurance (SHI) Germany, which includes the coverage of diabetes tests like HbA1c, thus facilitating high-throughput laboratory testing and automation through immunoassays systems. (Source: European Observatory on Health Systems and Policies)
- Hospitals are projected to hold 0% of the global HbA1c testing device market share in 2026, making it the dominant end user segment, with Asia Pacific leading due to high patient volumes and expanding national diabetes control programs. For instance, in India, the National Programme for Prevention and Control of Cancer, Diabetes, Cardiovascular Diseases, and Stroke (NPCDCS) provides a comprehensive approach to diabetes screening and treatment at district hospitals, community health centers, and health and wellness centers. According to the NPCDCS, population-based diabetes screening services are provided to people aged 30 and above years of age, assisted by non-communicable disease (NCD) clinics and primary health care facilities. (Source: Ministry of Health and Family Welfare)
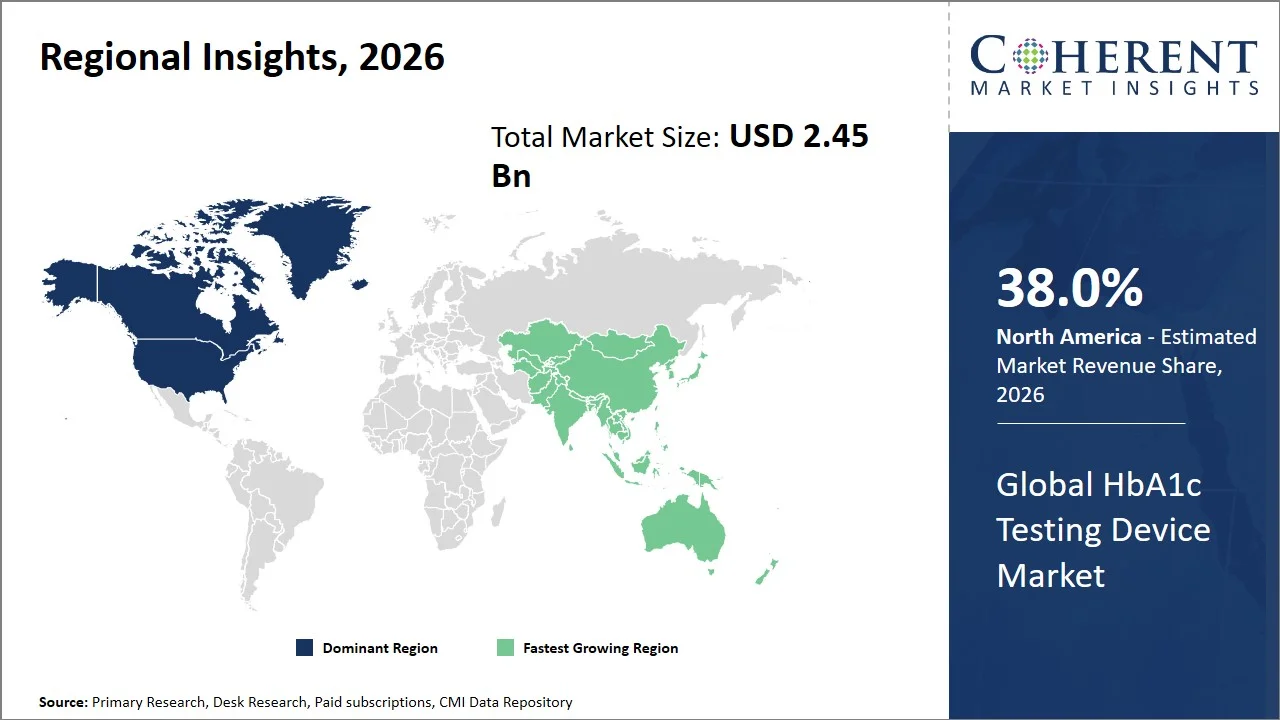
- North America maintains dominance with an expected share of 0% in 2026, supported by effective reimbursement frameworks and large-scale diabetes surveillance programs that facilitate regular testing. For instance, the Centers for Disease Control and Prevention runs the National Diabetes Surveillance System (NDSS), where continuous surveillance of diabetes prevalence is conducted, encouraging the practice of standard surveillance methods within the healthcare system, emphasizing the need for HbA1c testing. (Source: Centers for Disease Control and Prevention)
- Asia Pacific is expected to exhibit the fastest growth with an estimated contribution of 0% share in 2026, driven by regional policy coordination towards the early diagnosis and treatment of non-communicable diseases (NCDs). For instance, World Health Organization South-East Asia Regional Office encourages a structured approach to non-communicable diseases (NCDs), focusing on screening and management models in the member countries, where early detection of diabetes using the primary health care system is prioritized. This trend towards structured screening and monitoring in the region has greatly increased the need for diagnostic tests, including HbA1c. (Source: World Health Organization)
- Expansion of Point-of-Care (POC) HbA1c Testing in Decentralized Settings: Decentralization within the healthcare sector has witnessed the increased use of point-of-care HbA1c analyzers in hospitals, pharmacies, and clinics. These devices provide instant diagnosis through rapid analysis without the need to rely on laboratory results. It is particularly beneficial in developing nations, where laboratory resources are limited. As a result, there is strong demand for compact, user-friendly, and cost-efficient analyzers.
- Growing Use of HbA1c Testing in Preventive and Routine Health Screening: Increased awareness about diabetes and pre-diabetes conditions are encouraging the adoption of HbA1c testing procedures within annual examinations and prevention screening programs. Health practitioners are focusing on early identification and diagnosis so as to minimize complications that may arise as well as reduce the cost of treatment in the long run. This trend is leading to an increased demand for efficient and user-friendly testing machines.
Why Does the Point-of-Care (POC) Testing Devices Segment Dominate the Global HbA1c Testing Device Market?
Point-of-Care (POC) testing devices are projected to hold a market share of 68.0% in 2026, attributed to their ability to deliver rapid, on-the-spot results that enable immediate clinical decision-making, considerably improving diabetes management workflows. In comparison to conventional lab-based testing, Point-of-Care (POC) testing help medical practitioners get their patients' HbA1c test results in less than two minutes by taking a sample through a simple finger prick test such as A1CNow+, delivering results in about 5 minutes, enabling physicians to adjust treatment during the same patient visit, enhancing compliance and outcomes. (Source: PTS Diagnostics)
Furthermore, a clinical study published by Sage Journals indicates that immediate HbA1c feedback can improve glycaemic control because treatment changes will not require further waiting times in lab processing. (Source: Sage Journals) Such advantages have facilitated their widespread adoption within the realm of primary healthcare facilities. In addition, technological advancements associated with the use of Point-of-Care (POC) devices specifically increased accuracy that rival’s laboratory testing, as well as integration into treatment protocols have only enhanced their efficacy and importance, cementing their status as the best available option for regular monitoring, patient empowerment, and disease management.
Why Does Immunoassay Represent the Largest Technology Segment in the HbA1c Testing Device Market?
To learn more about this report, Request Free Sample
Immunoassay is projected to hold a market share of 36.0% in 2026, attributed to its accuracy, flexibility, and adaptability for implementation either in central laboratory or point-of-care use. Immunoassays employ antibody-antigen reactions in order to identify and measure levels of glycated hemoglobin in the sample blood in order to make the test less susceptible to interference and allow ease of application in laboratories. Among the commercially available HbA1c tests, such as the cobas b 101 and DCA Vantage Analyzer, include immunoassay technology in order to perform accurate measurements for HbA1c at hospitals and primary care clinics. (Source: Roche Diagnostics, Siemens Healthineers)
In addition, immunoassay HbA1c testing is aligned with international standardization of HbA1c, including the National Glycohemoglobin Standardization Program, ensuring traceability to reference methods and consistency across laboratories. Continual technological innovations, including automation and multi-parameter integration, will increase efficiency and economy, ensuring the supremacy of immunoassays as the most desirable technique for high-volume and routine HbA1c analysis.
Hospitals Segment Dominates the Global HbA1c Testing Device Market
The hospitals segment is projected to hold a market share of 55.0% in 2026, attributed to their central role in confirming diagnoses, optimizing treatment plans, and conducting regular monitoring, where the process of conducting HbA1c tests is an integral part of the standardized care processes. The leading role played by hospitals are also facilitated by clinical recommendations and well-defined healthcare systems that require the conduct of tests on a regular basis. For instance, the National Health Service makes sure that HbA1c tests become an inherent part of diabetes diagnosis and yearly program evaluation, which ensures the high volume of such tests being conducted by clinics and hospitals. (Source: The British Diabetic Association) Moreover, hospitals are increasingly utilizing care models that integrate data and electronic medical records to monitor patient outcomes and tailor treatment in real time, reinforcing their position as the leading end users in the HbA1c testing device market.
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
EU IVDR Amendment – Regulation (EU) 2024/1860 (2024 - Implementation Ongoing) |
|
|
U.S. FDA Final Rule Aligning QSR with ISO 13485 (February 2024) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global HbA1c Testing Device Market Dynamics
To learn more about this report, Request Free Sample
Market Drivers
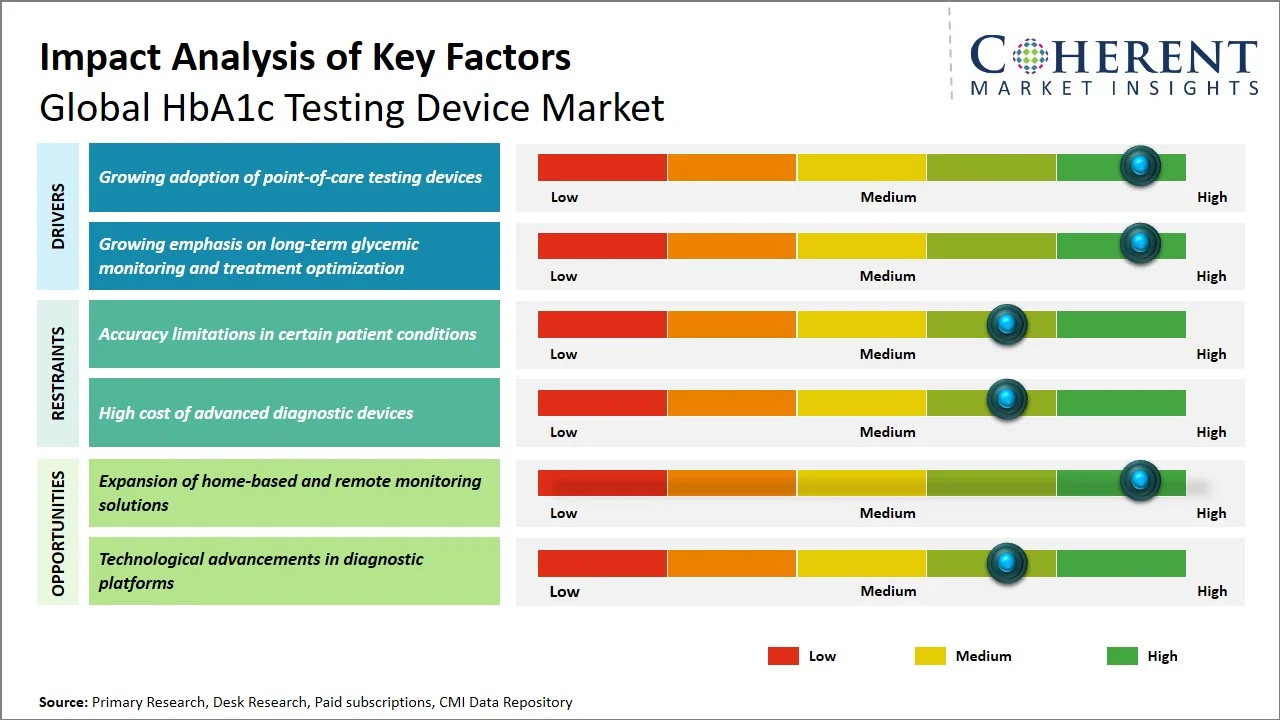
- Growing adoption of point-of-care testing devices: The increasing popularity of point-of-care (POC) testing devices are one of the significant trends that drive the growth of the HbA1c testing device market since medical professionals seek to shorten the time needed to produce results in outpatient facilities. The use of point-of-care testing devices allows obtaining results instantly, which contributes to quicker decision-making and more effective disease management. This trend is strengthened by regulatory and clinical acceptance of point-of-care diagnostics. For example, the U.S. Food and Drug Administration has approved multiple HbA1c point-of-care devices (such as, Afinion HbA1c Dx (Abbott), A1CNow+ (PTS Diagnostics), and Nova Allegro HbA1c assay) for clinical use under streamlined pathways, supporting their deployment beyond traditional laboratories. Moreover, the National Glycohemoglobin Standardization Program provides accreditation for point-of-care HbA1c tests to ensure standardization with reference values, thus enhancing the reliability of such tests. (Source: National Institutes of Diabetes and Digestive and Kidney Diseases) Such innovations, coupled with the rising demand for quick diagnostic devices, have resulted in the rapid growth of point-of-care HbA1c testing devices globally.
- Growing emphasis on long-term glycemic monitoring and treatment optimization: As clinical practice emphasizes trend analysis in measuring blood glucose levels rather than relying on point-of-care glucose testing. HbA1c testing provides a 2–3-month average of blood glucose levels, making it essential for assessing treatment efficacy and adjusting medications in a timely manner. According to clinical guidelines set by the American Diabetes Association (ADA), individuals with diabetes mellitus who have stable blood sugar control and are meeting treatment goals should have their HbA1c (A1C) checked at least twice a year. (Source: National Library of Medicine) Furthermore, health-care institutions are embracing personalized medicine approaches where data from constant monitoring is used for optimizing the effectiveness of medication and minimizing adverse side effects, thus underscoring the significance of HbA1c testing.
Emerging Trends
- Shift Toward Non-Invasive and Minimally Invasive HbA1c Testing Technologies: The growing interest in developing fewer invasive approaches to blood-based tests that require small amounts of blood, like HbA1c testing, is on the rise as manufacturers work to improve patient experience. There has been a shift towards the use of optics and micro-sampling techniques, especially in pediatric and geriatric patients.
- Integration of AI and Data Analytics in HbA1c Monitoring Systems: The use of HbA1c measuring instruments are increasingly becoming aligned with AI-based diagnostic platforms that can predict outcomes and facilitate the management of diabetes on a personal level. Such tools make it possible for practitioners to discern glycemic patterns and design effective therapies accordingly. The combination of diagnostics and digital health is transforming HbA1c analysis.
Regional Insights
To learn more about this report, Request Free Sample
Why is North America a Strong Market for HbA1c Testing Devices?
North America leads the global HbA1c testing device market, accounting for an estimated 38.0% share in 2026, owing to their earlier adoption of innovations and the strong presence of key players in the industry (such as Danaher Corporation, Bio-Rad Laboratories, and Siemens Healthineers), ensuring rapid commercialization of the products in the region. North America experiences regular launch and clearance of products within the HbA1c testing device market. For instance, in October 2025, Tosoh Bioscience obtained 510(k) approval from the U.S. Food and Drug Administration for its HLC-723-GR01 glycohemoglobin analyzer, an innovative system that is capable of providing fast and precise HbA1c analysis along with hemoglobin variant detection. (Source: U.S. Food and Drug Administration) Moreover, the region is characterized by a strong ecosystem of diagnostic device manufacturers and innovation pipelines, thus ensuring that the latest technologically sound and user-friendly devices are integrated quickly, which, in turn, helps to maintain its market leadership position in global HbA1c testing device market.
Why Does Asia Pacific HbA1c Testing Device Market Exhibit High Growth?
The Asia Pacific HbA1c testing device market is expected to exhibit the fastest growth with an estimated contribution of 32.0% share to the market in 2026, attributed to the increasing prevalence of diabetics in the area, the rising incidence rate of early onset diabetes, and large diagnostic requirements. According to the World Health Organization in the South-East Asia region, nearly 246 million people aged 30 years and above are estimated to have diabetes. More than half of people with people with diabetes are unaware of the condition. It is estimated that 68 million people with diabetes are on treatment underlining the treatment gap of 177 million in the region. Diabetes is responsible for around 48,000 deaths annually in the region. (Source: World Health Organization)
Furthermore, a research study published by Frontiers in February 2025, there has been an increased number of early onset type 2 diabetes in some Asia Pacific countries, like China, India, and Indonesia, due to urbanization and aging populations, among other factors. (Source: Frontiers) The regional healthcare system is increasingly focusing on the management of diabetes based on primary care and investing in diagnostic infrastructure, which includes HbA1c tests, to fill any gaps and ensure better management of diabetes. Such trends have contributed to the robust growth of the HbA1c testing device market in the Asia Pacific region.
Global HbA1c Testing Device Market Outlook for Key Countries
Why is the U.S. Leading Innovation and Adoption in the HbA1c Testing Device Market?
The U.S. has the potential for leadership in innovation and adoption in the field of HbA1c testing device due to robust diabetes management systems and clinical standardization, which facilitate the widespread application of HbA1c testing methods. Associations such as American Diabetes Association and National Glycohemoglobin Standardization Program provide an important platform for developing test methodologies and assuring device accuracy. This allows quick uptake of new technology and its adoption on a large scale, which helps boost the market’s growth potential. In addition, the well-integrated testing of HbA1c into primary care and outpatient practice workflows promotes its constant use, while the cooperation of diagnostic companies and health care professionals expedites the implementation of new diagnostic innovations.
Is Japan a Favorable Market for HbA1c Testing Devices?
Japan represents an ideal environment for HbA1c testing device market because of the country’s highly developed health care sector and its rigorous requirements in terms of diagnostic precision and the regular monitoring of disease conditions. Japan features a sophisticated clinical environment wherein HbA1c testing is regularly applied for diagnosing and managing diabetes cases and thus ensuring that testing procedures occur often per patient. Moreover, institutions such as the Japan Diabetes Society promote standardizing testing procedures and aligning them to international standards. In addition to that, Japan is host to prominent diagnostic companies such as Tosoh Corporation, that manufacture highly efficient HbA1c testing machines utilizing the HPLC technique. The presence of advanced technology, along with established testing standards, has made Japan a very reliable and valuable market for HbA1c test devices.
Is China Emerging as a Key Growth Hub for the HbA1c Testing Device Market?
China stands out as one of the most prominent countries with respect to the future growth of the HbA1c testing device market owing to its growing diagnostics base and its government’s emphasis on managing chronic diseases. Initiatives such as “Healthy China 2030” stress the importance of early screening and continuous monitoring of diabetes patients, thus promoting mass adoption of tests. Moreover, higher investments in hospital facilities (China’s healthcare expenditure exceeded USD 1.3 trillion (CNY 9 trillion) in recent years, reflecting sustained investment in hospital facilities and services) and upgrades in laboratories will enhance accessibility to HbA1c testing in both urban and semi-urban centers. In addition, presence of indigenous diagnostics companies in China along with growing healthcare spending will boost the growth prospects of the industry.
Why Does Germany Top the Europe HbA1c Testing Device Market?
Germany is the leader in the Europe market for HbA1c testing devices due to well-established healthcare infrastructure along with the high volume of testing facilitated by universal health care coverages. The well-equipped laboratory system in the country, along with standard procedures for diagnostics, help in ensuring that HbA1c tests are conducted on a continuous basis. Furthermore, the presence of associations such as German Diabetes Association, support diabetes monitoring programs (such as Disease Management Programs (DMPs) for Type 1 and Type 2 diabetes, and national diabetes care guidelines) in the country, reinforcing consistent testing demand.
Is HbA1c Testing Device Market Developing in India?
The market for HbA1c testing devices in India is undergoing transformation attributed to an enhanced accessibility of diagnostic tests and heightened monitoring of diabetes cases. The Research Society for the Study of Diabetes in India plays a role in supporting the clinical benchmarks for the treatment of diabetes, which involves HbA1c level tests. There has also been a tremendous increase in the number of private laboratories (with over 300,000 diagnostic labs across India, a majority being privately operated), has led to the development in test process. With a growing interest in preventive medicine and diagnosis, it appears that India will soon become an attractive emerging market for HbA1c testing equipment.
Regulatory Landscape Governing the Global HbA1c Testing Device Market
|
Region
|
Key Regulatory Bodies |
Framework |
Impact on Market |
|
North America |
U.S. Food and Drug Administration (FDA), Health Canada |
FDA 21 CFR (e.g., 862.1373 for HbA1c), 510(k) pathway, CLIA; ISO 13485 |
Strong regulatory oversight ensures high accuracy and reliability, but increases approval timelines and costs; CLIA-waived pathways enable expansion into POC settings |
|
Europe |
European Commission, Notified Bodies |
IVDR (EU 2017/746); CE Marking; ISO standards |
IVDR significantly increases compliance burden and certification complexity, leading to consolidation among manufacturers but improving patient safety and traceability |
|
Asia Pacific |
NMPA (China), CDSCO (India), PMDA (Japan), TGA (Australia) |
Country-specific IVD regulations (e.g., India Medical Device Rules 2017) |
Regulatory fragmentation creates entry barriers, but increasing harmonization and government support are boosting market expansion |
|
Latin America |
ANVISA (Brazil), COFEPRIS (Mexico), INVIMA (Colombia) |
National frameworks aligned with FDA/EU IVD standards |
Improving regulatory maturity enhances market standardization, though approval timelines and bureaucracy may delay product launches |
|
Middle East & Africa |
SFDA (Saudi Arabia), SAHPRA (South Africa), National MoH bodies |
Hybrid frameworks relying on FDA/CE approvals + local registration |
Faster approvals via reliance pathways support market accessibility, but limited local manufacturing keeps dependency on imports high |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
How is expansion of home-based and remote monitoring solutions creating new growth opportunities in the global HbA1c testing device market?
Remote and at-home monitoring technologies have created huge growth prospects for the HbA1c testing device market since they allow for decentralized diabetes management away from clinical settings. The proliferation of portable HbA1c kits has made it easy for people to carry out HbA1c tests at home, hence making it easier to avoid laboratory visits. For instance, devices such as OBM rapid A1c home testing can be used to carry out high-quality laboratory-grade HbA1c tests within minutes using only a finger prick sample, which enables at-home HbA1c testing. (Source: ME+DI Qmed) Likewise, other products such as A1CNow Self Check monitor can be used for quick HbA1c tests at the point of care since they do not require any kind of lab analysis. (Source: Healthline) Telemedicine technology has also played a role in the development of this trend since at-home HbA1c test kits as well as remote glycemic index monitors have shown strong concordance with point-of-care testing, making them ideal for telehealth applications . This has led to an increased demand for user-friendly HbA1c testing devices that facilitate remote monitoring.
Market Players, Key Development, and Competitive Intelligence

To learn more about this report, Request Free Sample
Key Developments
- In January 2026, Avricore Health Inc. launched the expansion of its HealthTab technology across North East London, involving nearly 70 pharmacies within the community. This is part of its strategy following NHS diagnostic initiatives and focuses on improving accessibility to point-of-care diagnostics in retail health care facilities. This move demonstrates the emerging trend towards decentralization of diagnostics, enabling more rapid test options like HbA1c outside traditional medical facilities.
- In May 2025, Tosoh Bioscience Diagnostics EMEA released a new generation HbA1c analyzer called HLC-723 GR01 which is capable of offering high levels of accuracy and dependability. This new model is based on modern HPLC technology and is ideal for laboratories that process large numbers of samples due to its efficiency and effectiveness. With its superior accuracy of HbA1c measurements, the new analyzer will facilitate better clinical decisions.
Competitive Landscape
The global HbA1c testing device market will likely stay highly competitive characterized by constant innovations in diagnostics technology and rising demands for improved diabetes testing solutions. Key players including Abbott Laboratories, Roche Diagnostics, and Siemens Healthineers aim to improve the accuracy, mobility, and processing time of their products to maintain their competitive advantage. Besides, manufacturers are adopting point-of-care kits and automated analyzers to serve the requirements of decentralized and centralized facilities. The introduction of collaborations and new products further adds to the fierce competition within the industry, while the penetration of new markets becomes an important growth tactic. Key focus areas include:
- Advancement in point-of-care (POC) and portable HbA1c devices
- Enhancing accuracy and standardization of testing methods
- Integration of digital health and data connectivity
- Expansion into emerging markets with cost-effective and scalable diagnostic solutions
- Strategic collaborations and product innovation
Market Report Scope
HbA1c Testing Device Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 2.45 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7.7% | 2033 Value Projection: | USD 3.78 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Abbott Laboratories, F. Hoffmann-La Roche Ltd., Siemens Healthineers, Danaher Corporation, Bio-Rad Laboratories, ARKRAY Inc., Trinity Biotech plc, EKF Diagnostics Holdings plc, A. Menarini Diagnostics, and Tosoh Corporation |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Analyst Opinion (Expert Opinion)
- In the coming years, the prospects of the HbA1c testing device market will become one defined by a diagnostics approach that is more decentralized, more interconnected, and more oriented towards patients than before, as it will be possible to perform diagnostics not only in a lab but also through primary care practices, from home, and even in a pharmacy. Digital medicine, cloud computing for storing patient data, and telemedicine could facilitate continuous assessment of patient results.
- The biggest opportunity would come in the point-of-care testing (POC) segment in India and China because of the rising demands from under-served populations, urbanization, and growth in the number of primary care facilities. In addition, Tier II and III cities will offer immense opportunities, considering the continuous investments made by both the government and the private sector in screening and decentralized diagnostics.
- For gaining any competitive advantage in the market, companies need to concentrate on developing a balance between efficiency and accuracy. This should be achieved through investments in small, portable gadgets and fast tests that are compatible with electronic health records and mHealth applications. Moreover, collaborations should be established with hospitals, diagnostic centers, and public health campaigns.
Market Segmentation
- Product Type Insights (Revenue, USD Bn, 2021 - 2033)
- Point-of-Care (POC) Testing Devices
- Laboratory-based Analyzers
- Technology Insights (Revenue, USD Bn, 2021 - 2033)
- Immunoassay
- Enzymatic Assay
- Chromatography
- Others
- End User Insights (Revenue, USD Bn, 2021 - 2033)
- Hospitals
- Diagnostic Laboratories
- Clinics
- Homecare Settings
- Others
- Regional Insights (Revenue, USD Bn, 2021 - 2033)
- North America
- U.S.
- Canada
- Latin America
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Europe
- Germany
- U.K.
- Spain
- France
- Italy
- Russia
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Australia
- South Korea
- ASEAN
- Rest of Asia Pacific
- Middle East
- GCC Countries
- Israel
- Rest of Middle East
- Africa
- South Africa
- North Africa
- Central Africa
- North America
Sources
Primary Research Interviews
- Diagnostic laboratory directors and clinical biochemists specializing in diabetes testing from accredited labs and hospitals
- Medical device executives and product managers from HbA1c analyzer manufacturers
- Endocrinologists and diabetologists using HbA1c testing for long-term glycemic monitoring
- CROs and regulatory consultants involved in IVD device approvals and clinical validation
Stakeholders
- HbA1c Testing Device Manufacturers & IVD Solution Providers
- End-use Sectors
- Hospitals & Diagnostic Laboratories
- Diabetes Clinics & Specialty Centers
- Home Healthcare & Self-monitoring Users
- Academic & Research Institute
- Regulatory & Health Bodies: U.S. Food and Drug Administration – classification and approval of HbA1c test systems (21 CFR 862.1373), European Commission – IVDR (EU 2017/746) for IVD devices, World Health Organization – diabetes diagnostics guidelines and global disease monitoring, Centers for Disease Control and Prevention – diabetes surveillance and testing recommendations
Databases
- FDA Product Classification Database – HbA1c test system classification (Class II, 510(k))
- EUDAMED – EU database for IVD device registration and tracking
- WHO Global Health Observatory – diabetes prevalence and diagnostic access data
- OECD Health Statistics – healthcare expenditure and diagnostic adoption trends
Magazines
- Clinical Laboratory Products (CLP) – HbA1c testing advancements and lab diagnostics
- Medical Device Network – IVD regulatory updates and diagnostic innovations
- Diagnostics World News – developments in diabetes testing technologies
Journals
- Diabetes Care
- Clinical Chemistry
- Journal of Diabetes Science and Technology
- Clinica Chimica Acta
Newspapers
- Financial Times – coverage on global diagnostics and healthcare investments
- The Wall Street Journal – reporting on diagnostic companies and innovation
- The Economic Times (India) – diabetes burden and diagnostics expansion in India
- Business Standard (India) – healthcare policy and diagnostic market developments
Associations
- American Diabetes Association
- International Diabetes Federation
- Clinical and Forensic Toxicology Association
- Association for Diagnostics & Laboratory Medicine
Public Domain Sources
- National Institutes of Health – diabetes research and biomarker studies
- Centers for Disease Control and Prevention – diabetes statistics and screening guidelines
- European Commission – funding and regulatory initiatives in IVDs
- Ministry of Health and Family Welfare – national diabetes programs and diagnostic initiatives
Proprietary Elements
- CMI Data Analytics Tool
- Proprietary CMI Existing Repository of information for last 10 years.
Share
Share
About Author
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients