Global Heparin Sodium Market Size and Forecast – 2026 To 2033
The global heparin sodium market is expected to grow from USD 8.42 Bn in 2026 to USD 12.96 Bn by 2033, registering a compound annual growth rate (CAGR) of 6.4% from 2026 to 2033. The market for heparin sodium is poised for significant expansion, fueled by the rising number of surgical procedures and the growing need for perioperative anticoagulation around the world.
According to the World Health Organization, over 300 million surgical procedures are performed each year worldwide, while an additional 143 million surgical procedures are needed each year to save lives and prevent disability.
(Source: World Health Organization, World Health Organization)
Key Takeaways of the Global Heparin Sodium Market
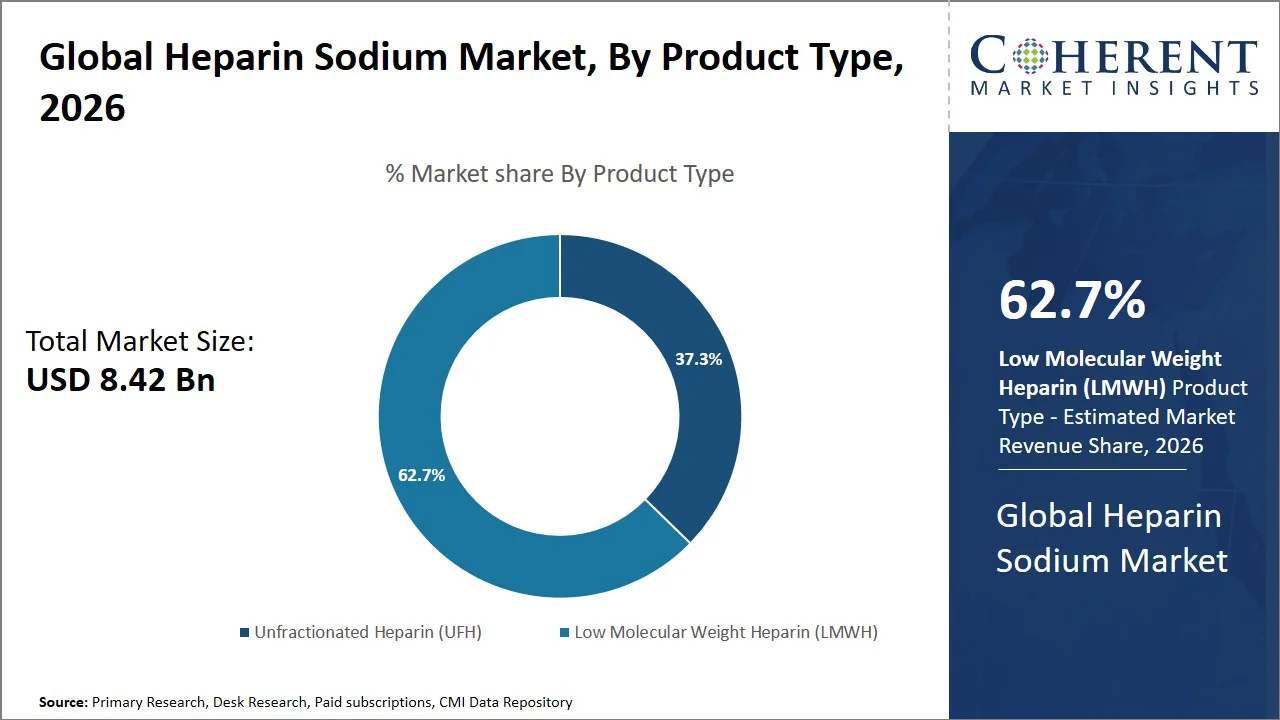
- The Low Molecular Weight Heparin (LMWH) segment is projected to hold 62.7% of the global heparin sodium market share in 2026, making it the dominant product type segment, with dominance in Europe owing to frequent administration for postoperative thromboprophylaxis and orthopedic management procedures. There is an increasing trend for the use of Low Molecular Weight Heparin (LMWH) in ERAS programs in Europe owing to the easy administration and suitability for outpatient anticoagulant therapy. For instance, the European Society of Cardiology (ESC) Clinical Practice Guidelines continue to recommend anticoagulant therapy using Low Molecular Weight Heparin (LMWH) in cases of thromboembolic disease in both cardiovascular and perioperative settings. (Source: European Society of Cardiology)
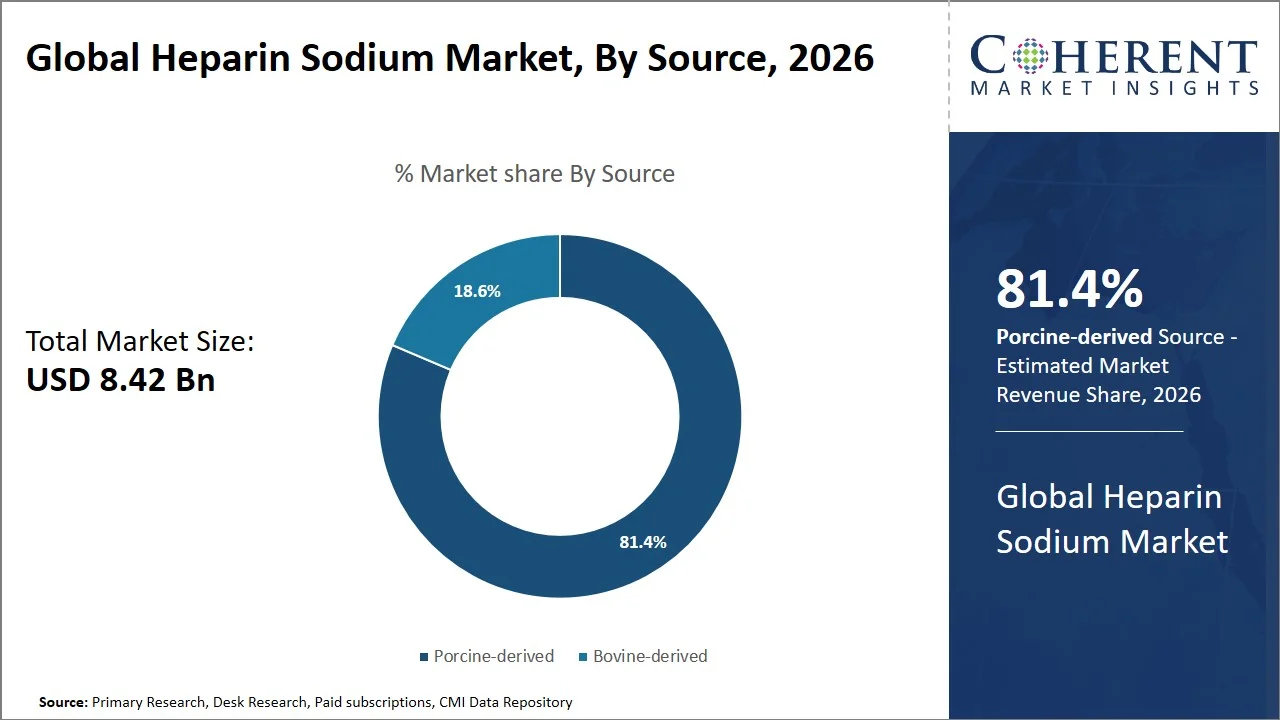
- Porcine-derived is projected to hold 81.4% of the global heparin sodium market share in 2026, making it the dominant source segment, with high penetration in Asia Pacific attributed to the presence of a well-established industrialized sector that deals with pork processing and crude heparin extraction to manufacture drugs. This region holds a significant stake in supplying porcine intestine mucosa needed for anticoagulants. For instance, China Animal Agriculture Association emphasizes the importance of China being one of the largest producers of pork in the world, ensuring the availability of porcine raw materials required for commercial heparin extraction and large-volume anticoagulant manufacturing. (Source: National Library of Medicine)
- Intravenous is projected to hold 58.6% of the global heparin sodium market share in 2026, making it the dominant route of administration segment, especially in North America where intravenous anticoagulation continues to be highly prevalent in extracorporeal treatments and intervention procedures involving the heart. Clinics and other medical facilities in the area continue to emphasize the use of intravenous heparin therapy during catheterization, extracorporeal membrane oxygenation, and emergency procedures of the coronary arteries. For instance, the Clinical Guidelines by the Society of Thoracic Surgeons (STS) encompass intravenous anticoagulation therapy for cardiothoracic procedures as well as extracorporeal treatment, supporting sustained clinical utilization of IV heparin therapies in advanced surgical care settings. (Source: Society of Thoracic Surgeons)
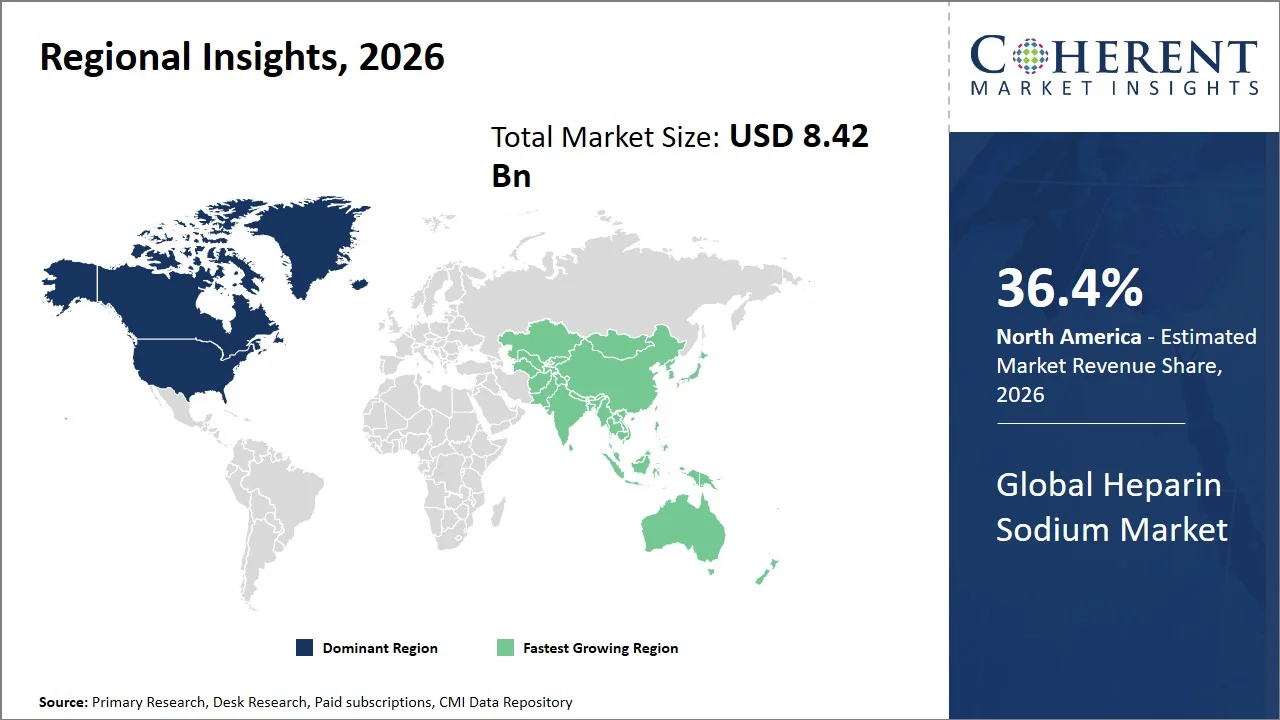
- North America maintains its dominance with an estimated share of 36.4%in 2026, as a result of the highly organized system for managing anticoagulant medication in the region and the widespread use of heparin in cardiac surgery, dialysis, and extracorporeal procedures. For instance, Centers for Medicare & Medicaid Services (CMS) ESRD Program continues to provide support for the development of facilities providing dialysis therapy in the U.S., wherein heparin sodium is administered in all hemodialysis sessions in order to prevent the formation of clots within the circuit. (Source: Centers for Medicare & Medicaid Services)
- Asia Pacific is expected to exhibit the fastest growth with an estimated contribution of 24.8% share in 2026, owing to fast development in critical care infrastructure, higher volumes of cardiovascular interventions, and improved heparin API manufacturing environments in China, India, and Southeast Asia. For instance, the government of China launched the “Opinions on Enhancing the Development Capacity of Emergency Medicine Services,” a policy focusing on enhancing ICU bed capacity and emergency critical care facilities across healthcare institutions by 2025. (Source: ResearchGate)
- Growing utilization of heparin sodium in extracorporeal life-support systems: Rising adoption of extracorporeal membrane oxygenation (ECMO), cardiopulmonary bypass systems and continuous renal replacement therapy (CRRT) is presenting niche growth opportunities for manufacturers of heparin sodium. These procedures necessitate continuous anticoagulation management to prevent clot formation in external circulation circuits, propelling specialized demand for hospital-grade injectable heparin products in critical care units.
- Expansion of biosafety and contamination testing services in anticoagulant manufacturing: Rising regulatory scrutiny surrounding animal-sourced components in pharmaceutical products is leading to higher demands for sophisticated testing and purification systems in the production of heparin. This presents an emerging business opportunity for companies that have invested in high-purity processes and pathogen testing systems.
Why Does the Low Molecular Weight Heparin (LMWH) Segment Dominate the Global Heparin Sodium Market?
The Low Molecular Weight Heparin (LMWH) segment is projected to hold a market share of 62.7% in 2026 owing to better pharmacodynamics properties, predictable anticoagulant response, reduced risks of heparin-induced thrombocytopenia (HIT), and decreased need for constant coagulation monitoring compared with the unfractionated heparin. The use of Low Molecular Weight Heparin (LMWH) is widely accepted among cardiovascular therapy, venous thromboembolism (VTE) treatment, orthopedic surgery, and dialysis treatments as the product allows achieving greater ease in subcutaneous drug administration and providing reliable anticoagulation results under both inpatient and outpatient treatment scenarios.
Moreover, clinical practice guidelines and institutional protocols continue to recommend Low Molecular Weight Heparin (LMWH) more often in prophylaxis and postoperative anticoagulation management because of fewer complications and greater convenience to patients. Furthermore, a study published by the National Library of Medicine, Low Molecular Weight Heparin (LMWH) was found to be safer and more effective than traditional unfractionated heparin in some areas of medical practice, owing to its reduced potential for causing unwanted side effects associated with blood clotting, supporting its widespread clinical adoption globally. (Source: National Library of Medicine)
Why Does Porcine-derived Represent the Largest Source Segment in the Heparin Sodium Market?
To learn more about this report, Download Free Sample
Porcine-derived is projected to hold a market share of 81.4% in 2026, owing to its better biological compatibility, proven safety profile, and greater yield in terms of anticoagulants when compared to alternatives from different animal species. Porcine intestinal mucosa continues to be the raw material primarily used in the commercial manufacturing of heparin owing to its consistent purity level and large-scale manufacturing capacity.
Moreover, regulations governing heparin and pharmacopoeia have been largely designed for porcine formulations, which helps to increase its acceptance rate in cardiovascular surgeries, dialysis, and intensive care unit treatments. The segment enjoys easy availability of raw materials and an efficient supply chain network, especially in key production markets such as China and the U.S. For instance, a study published by the National Institutes of Health (NIH) stated that “at present, all the unfractionated heparin used around the world comes from porcine mucosa” in most of the international markets, emphasizing its dominant commercial and regulatory position in the anticoagulants segment. (Source: National Library of Medicine)
Intravenous Segment Dominates the Global Heparin Sodium Market
The intravenous segment is projected to hold a market share of 58.6% in 2026, attributed to the quick onset of anti-coagulation effects and use in emergencies, heart surgeries, kidney dialysis, and other critical cases where instant anti-coagulation becomes necessary. An intravenous delivery route enables medical practitioners to regulate dosage and monitor anti-coagulation continuously, which makes the method of choice in complex operations, such as extracorporeal membrane oxygenation (ECMO) treatment, catheterization, and heart surgery. Moreover, intravenous anti-coagulation drugs are increasingly adopted by hospitals in various acute conditions.
For instance, the National Blood Clot Alliance notes that unfractionated heparin delivered through intravenous injection is often prescribed for hospitalized patients in order to rapidly initiate anti-coagulation due to its instant effectiveness and ability to regulate dosage. (Source: National Blood Clot Alliance) It can thus be anticipated that the growing trend towards rapid and controllable anticoagulant drugs in hospitals and intensive care units will continue to drive global demand for intravenous heparin sodium.
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
Regulatory Shift: Updated UK Kidney Association Anticoagulation Guidelines for Advanced CKD and VTE Management (September 2025) |
|
|
Regulatory & Quality Compliance Shift: Increased Global Regulatory Focus on Heparin Product Quality and Traceability (August 2025) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
(Source: UK Kidney Association, Therapeutic Goods Administration)
Heparin Sodium Market Dynamics
To learn more about this report, Download Free Sample
Market Drivers
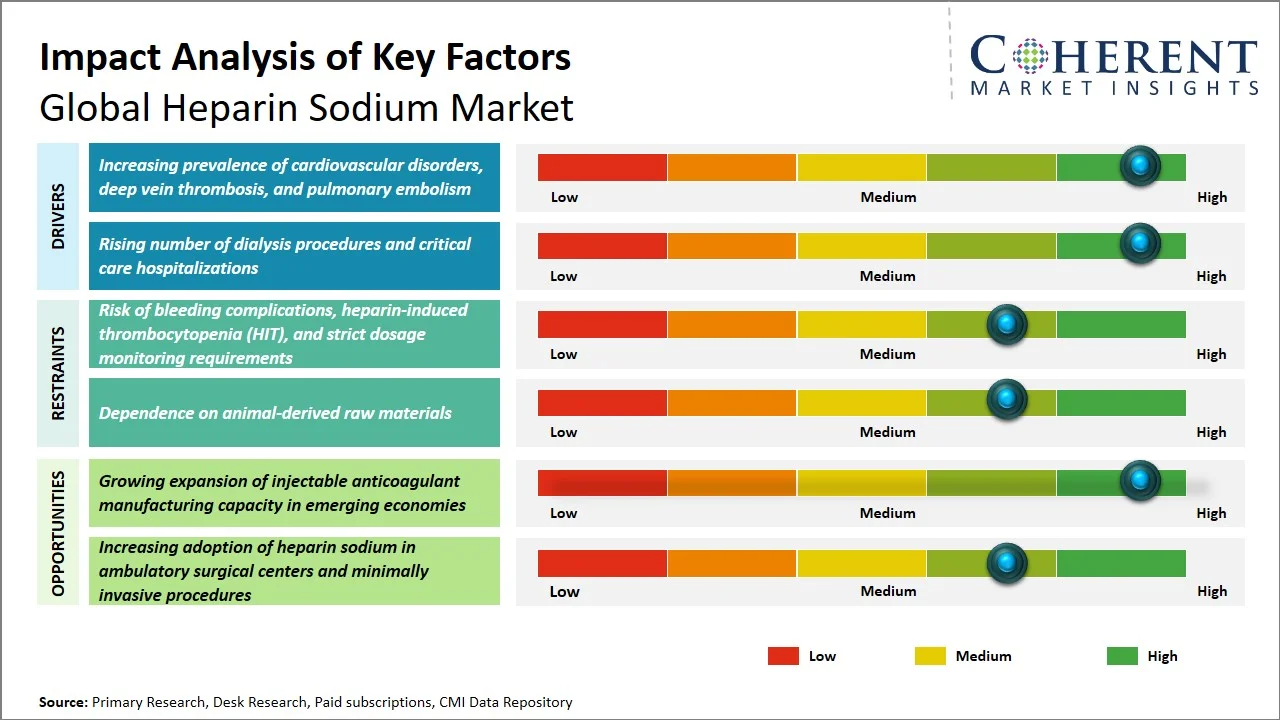
- Increasing prevalence of cardiovascular disorders, deep vein thrombosis, and pulmonary embolism: The rising occurrence of heart conditions, deep vein thrombosis (DVT), and pulmonary embolism is creating an increased demand for heparin sodium because of its importance in preventing and curing the formation of blood clots in cases involving cardiovascular surgery, dialysis, and critical care treatments. According to the World Health Organization, cardiovascular diseases (CVDs) are the leading cause of death globally, with an estimated 19.8 million deaths from CVDs, representing approximately 32% of all global deaths. (Source: World Health Organization) Additionally, according to Centers for Disease Control and Prevention, a heart attack occurs in the U.S. approximately every 40 seconds. Every year, about 805,000 people in the U.S. have a heart attack. Of these, 605,000 are a first heart attack, and 200,000 happen to be people who have already had a heart attack. (Source: Centers for Disease Control and Prevention) It is thus anticipated that heparin sodium will continue to be in demand due to the rising burden of thromboembolic and cardiovascular emergencies around the world.
- Rising number of dialysis procedures and critical care hospitalizations: The rise in the incidence of chronic kidney disease, acute kidney injury, and critical care patients will greatly boost the demand for heparin sodium owing to its regular application in avoiding clot formation during the hemodialysis process and extracorporeal circulation procedures. In addition, the rising number of ICU admissions and the expansion of dialysis facilities in hospitals will fuel the use of anticoagulants in acute care hospitals. For instance, according to Centers for Disease Control and Prevention, more than 1 in 10 (14%) adults aged 18 or older (37 million people) were estimated to have chronic kidney disease. (Source: Centers for Disease Control and Prevention) Moreover, as per the National Kidney Foundation, hemodialysis needs to be performed with the use of anticoagulant drugs due to their ability to prevent blood clotting inside the dialysis machine during the process of dialysis. As a result, heparin sodium will continue to have strong demand throughout the world.
Emerging Trends
- Increasing adoption of premixed and ready-to-administer anticoagulant formulations: Hospitals and critical care centers are increasingly inclined towards using premixed heparin sodium injections, as they reduce the chances of any medication preparation mistakes while ensuring precise dosages are administered and improving operational efficiency. This trend is supporting higher demand for standardized sterile injectable products.
- Expansion of traceability and quality monitoring systems for animal-derived APIs: Advanced systems are being used increasingly by manufacturers for traceability, contamination prevention, and raw material verification when manufacturing heparin derived from porcine. This development is being spurred by the increasing focus on ensuring that pharmaceuticals manufactured and distributed globally are safe, secure, and sourced correctly.
Regional Insights
To learn more about this report, Download Free Sample
Why is North America a Strong Market for Heparin Sodium?
North America leads the global heparin sodium market, accounting for an estimated 36.4% share in 2026, attributed to its wide application in anticoagulants used during cardiovascular surgery, dialysis therapy, catheterization, and intensive care operations. North America is supported by well-equipped hospitals, a high prevalence of sterile drug administration, and an extensive use of anticoagulation management techniques in intensive care units.
In addition, the demand for heparin injection will be encouraged due to the availability of facilities specialized in cardiac care along with the extensive application of heparin during extracorporeal circulation therapies like extracorporeal membrane oxygenation and hemodialysis. For instance, ESRD (End-Stage Renal Disease) Program from the U.S. Centers for Medicare & Medicaid Services (CMS) provides substantial funding for dialysis facilities, where heparin sodium is commonly used to avoid clotting during hemodialysis procedures. (Source: Centers for Medicare & Medicaid Services)
Why Does the Asia Pacific Heparin Sodium Market Exhibit High Growth?
The Asia Pacific heparin sodium market is expected to exhibit the fastest growth with an estimated contribution of 24.8% share to the market in 2026, owing to the increasing establishment of cardiovascular treatment centers, the use of extracorporeal treatments, and enhanced export capacities for pharmaceuticals in the region. Nations such as China, Japan, and South Korea have seen increasing demand for anticoagulants for minimally invasive cardiology, intensive care, and dialysis treatment. Moreover, the Asia Pacific region enjoys economies of scale with biologic and injectable drug manufacturing facilities capable of producing affordable anticoagulant drugs. Furthermore, the increasing access to healthcare facilities and modernization of tertiary hospitals in the region are driving growth in the uptake of injectable medicines. For instance, the Japan Agency for Medical Research and Development (AMED) is providing support in advanced clinical research and development in critical care and cardiology treatment methods in Japan. (Source: Japan Agency for Medical Research and Development)
Global Heparin Sodium Market Outlook for Key Countries
Why is the U.S. Leading Innovation and Adoption in the Heparin Sodium Market?
The U.S. has the potential for leadership in innovation and adoption in the field of heparin sodium attributed to the strong implementation of anticoagulation procedures during cardiovascular surgeries, extracorporeal membrane oxygenation (ECMO), and dialysis treatment processes. There is a rising trend among U.S. hospitals and health care facilities toward using heparin sodium products in a ready-to-inject form in order to minimize the risk of medical mistakes when administering the product.
There are highly sophisticated technologies of making anticoagulants available within their sterility standards, as well as advanced quality control procedures. Moreover, the presence of dedicated thrombosis management programs (such as Anticoagulation Stewardship Programs, Venous Thromboembolism Prevention Programs, and Hospital-based Anticoagulation Clinics) and high rates of catheterization contribute to constant demand for heparin sodium in the U.S.
Is Japan a Favorable Market for Heparin Sodium?
Japan is considered a favorable market for heparin sodium as it is well-known for its clinical importance in terms of cardiovascular medicines, dialysis, and peri-operative anticoagulation treatment. There is already an established system of hemodialysis in Japan, where the use of heparin sodium plays an important role in the prevention of formation of clots during extracorporeal circulation procedures. In addition, due to stringent quality requirements of injectables, combined with controlled procedures of anticoagulation administration, there is consistent demand for heparin medicines. Additionally, increased application of minimally invasive cardiovascular surgeries, along with advancements in intensive treatment, has made the importance of heparin sodium even more pronounced.
Is China Emerging as a Key Growth Hub for the Heparin Sodium Market?
Heparin sodium market in China is currently experiencing significant development attributed to the high prominence the nation holds in the area of heparin raw material processing and the large scale of active pharmaceutical ingredient (API) manufacturing within the region. China has a vital role in the global heparin supply chain, which includes the production of large quantities of heparin components that have anti-coagulation properties and are used in the injectable form across the globe. For instance, according to research published by National Library of Medicine, most of the unfractionated and low molecular weight heparins available in the world are manufactured by Chinese manufacturers from porcine mucosa.
Furthermore, the article emphasized that China is the largest pork producer in the world, hence its significance as a source of raw materials for the manufacture of injectable heparin. Moreover, there has been continuous growth in the manufacturing of sterile injectables along with the development of hospitals in China owing to rising needs of anti-coagulants in surgeries, dialysis, and intensive care treatments. Moreover, pharmaceutical manufacturing modernization and export-driven production methods further solidify China’s presence within the global heparin sodium market.
Why Does Germany Top the European Heparin Sodium Market?
Germany dominates the heparin sodium market in Europe owing to its highly developed hospital facilities, extensive use of anticoagulants in surgeries, and use of heparin in cardiovascular and dialysis procedures. Germany has highly developed industries that manufacture sterile injectable medicines along with strict guidelines for use in cases of thrombosis. Furthermore, Germany gains much from having well-integrated systems for anticoagulation therapy within intensive care units, catheterization laboratories, and perioperative treatment methods. The presence of some of the best pharmaceutical manufacturing plants and cardiovascular treatment centers (such as B. Braun Melsungen AG, Fresenius Kabi, German Heart Center Berlin, and University Heart & Vascular Center Hamburg) remains instrumental in sustaining a constant need for heparin sodium in Germany.
Is the Heparin Sodium Market Developing in India?
The demand for heparin sodium in India is poised to witness an increase owing to the robust expansion witnessed by hospitals in terms of their infrastructure, high demand for dialysis treatments, and increasing usage of anticoagulant injections for cardiovascular surgeries and critical care. Furthermore, increased investment in the establishment of sterile injectable facilities and API facilities by pharmaceuticals in India will lead to the development of manufacturing capacity in the country (over USD 575 million invested and manufacturing capacity of 56,800 metric tons per annum created under India’s PLI scheme for pharmaceuticals and bulk drugs) (Source: Ministry of Chemicals and Fertilizers) In addition, enhanced tertiary care coverage and improved access to surgical treatment will drive the increased consumption of heparin sodium.
Regulatory Landscape Governing the Global Heparin Sodium Market
|
Region |
Key Regulatory Bodies |
Approval & Compliance Framework |
Impact on Market |
|
North America |
U.S. Food and Drug Administration (FDA), Health Canada |
Current Good Manufacturing Practices (cGMP), sterile injectable quality standards, anticoagulant safety monitoring, pharmacovigilance requirements |
Strong regulatory oversight enhances product quality, traceability, and supply chain reliability while increasing compliance costs for manufacturers |
|
Europe |
European Medicines Agency (EMA), Medicines and Healthcare products Regulatory Agency (MHRA) |
E GMP guidelines, biologic raw material traceability standards, centralized marketing authorization procedures |
Supports harmonized anticoagulant approvals across European countries and strengthens patient safety and contamination control measures |
|
Asia Pacific |
National Medical Products Administration (NMPA), Central Drugs Standard Control Organization (CDSCO), Pharmaceuticals and Medical Devices Agency (PMDA) |
Local manufacturing compliance, injectable drug registration requirements, evolving pharmacovigilance standards |
Expanding regulatory modernization improves market access opportunities while increasing quality compliance requirements for domestic manufacturers |
|
Latin America |
National Health Surveillance Agency (ANVISA), Federal Commission for Protection against Health Risks (COFEPRIS) |
Import approval regulations, GMP certification, injectable pharmaceutical registration frameworks |
Increasing regulatory alignment with international standards supports growth of imported and locally manufactured anticoagulants |
|
Middle East & Africa |
Saudi Food and Drug Authority (SFDA), South African Health Products Regulatory Authority (SAHPRA) |
Drug registration procedures, quality assurance standards, import and distribution regulations |
Growing regulatory standardization is improving pharmaceutical accessibility and encouraging multinational participation in regional anticoagulant markets |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
How is growing expansion of injectable anticoagulant manufacturing capacity in emerging economies creating new growth opportunities in the heparin sodium market?
The expanding manufacturing capacity for anticoagulant injections in developing countries is creating significant growth opportunities in the heparin sodium market. The expansion will not only improve access to medication locally but also enhance the strength of their supply chain in addition to decreasing reliance on the importation of anticoagulants. With the increasing demands for cardiovascular surgery, dialysis, and intensive care purposes in India and China, there are many investments being made to improve their sterile injection capabilities. Besides, the expansion of healthcare and increase in hospitals constructions in these countries have also increased the use of anticoagulants in the acute care environment.
For instance, India’s pharmaceutical industry receives considerable governmental assistance via projects like the Production Linked Incentive (PLI) Scheme for Pharmaceuticals. The Production Linked Incentive (PLI) scheme is meant to boost manufacturing capability in India regarding essential drugs and pharmaceutical ingredients, indirectly assisting increased manufacturing capabilities of injectable anticoagulants such as heparin sodium. (Source: Ministry of Chemicals and Fertilizers)
Market Players, Key Development, and Competitive Intelligence
To learn more about this report, Download Free Sample
Key Developments
- In August 2025, Cormedix Inc. made an announcement regarding the purchase of Melinta Therapeutics as part of its effort to bolster its position in the acute care specialty pharmaceuticals industry and further its reach through hospital-related treatment centers. The acquisition played a significant role in helping the firm increase its competencies in critical care and injectables, which have a strong correlation with anticoagulants used in catheterization, dialysis, and surgical treatments.
- In July 2025, Braun Melsungen AG introduced a total of seven heparin sodium injection premixes into its product line of heparin sodium injections. These were introduced in an effort to improve the process of preparing medications, reduce dosage errors, and enhance the safety associated with the use of anticoagulants. This initiative by B. Braun Melsungen AG highlights a trend in the pharmaceutical sector toward the introduction of ready-for-use injectable products for increased efficiency.
Competitive Landscape
The global heparin sodium market is moderately consolidated based on the activities of multi-national pharmaceutical companies, injectable drug manufacturers, and anticoagulant API producers, all of which seek to extend their range of hospital products. The market players' emphasis is being put on integration into the supply chain, adherence to regulations, and purity of heparin used to provide stable access to quality products. Moreover, increasing investments in premixed injectables, ready-to-use anticoagulants, and expansion of sterile manufacturing plants have become typical competitive tactics. Key focus areas include:
- Expansion of sterile injectable manufacturing capacity
- Development of premixed and ready-to-administer heparin formulations
- Strengthening porcine raw material sourcing and supply chain security
- Enhancing regulatory compliance and contamination control standards
- Expanding hospital and critical care distribution networks
Market Report Scope
Heparin Sodium Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 8.42 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 6.4% | 2033 Value Projection: | USD 12.96 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Pfizer Inc., Sanofi, Baxter International Inc., B. Braun Melsungen AG, Aspen Pharmacare, LEO Pharma A/S, Dr. Reddy’s Laboratories Ltd., Fresenius Kabi, Teva Pharmaceutical Industries Ltd., and Shenzhen Hepalink Pharmaceutical Group Co., Ltd. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Analyst Opinion (Expert Opinion)
- In the coming years, the prospects of the heparin sodium market will significantly influenced by the growing incidence of heart-related conditions, surgery cases, and dialysis patients, especially in aging populations. The need for anticoagulant medicines will be projected to grow as hospitals and other critical facilities continue to place more emphasis on quick and efficient treatment of blood clots during surgeries and extracorporeal processes. Moreover, the market is expected to gradually move towards the use of safe and pre-filled heparin injections as well as better quality traceable heparin manufacturing processes.
- The highest opportunities are anticipated for low molecular weight heparin (LMWH) segment, which can be attributed to the increase in cardiovascular and dialysis applications in China and India due to the improvement in the hospital infrastructure, high prevalence of chronic kidney disease, and cardiovascular admissions. The growing focus on developing sterile injectables and improved accessibility to healthcare in tier-2 and tier-3 cities will also drive demand in Asia Pacific.
- For gaining any competitive advantage in the market, company’s strategies must revolve around improving vertical integration in supply chains, increasing the capacity of sterile injectable production, and exploring alternative sources of animal-based materials to ensure uninterrupted supplies. In addition to this, companies must consider premixed formulations of heparin, state-of-the-art quality monitoring tools, and localized regulatory compliance practices.
Market Segmentation
- Product Type Insights (Revenue, USD Bn, 2021 - 2033)
- Unfractionated Heparin (UFH)
- Low Molecular Weight Heparin (LMWH)
- Source Insights (Revenue, USD Bn, 2021 - 2033)
- Porcine-derived
- Bovine-derived
- Route of Administration Insights (Revenue, USD Bn, 2021 - 2033)
- Intravenous
- Subcutaneous
- Application Insights (Revenue, USD Bn, 2021 - 2033)
- Venous Thromboembolism (VTE)
- Atrial Fibrillation & Coronary Artery Disease
- Kidney Dialysis
- Surgery & Critical Care
- Others
- End User Insights (Revenue, USD Bn, 2021 - 2033)
- Hospitals
- Ambulatory Surgical Centers (ASCs)
- Dialysis Centers
- Specialty Clinics
- Others
- Distribution Channel Insights (Revenue, USD Bn, 2021 - 2033)
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
- Regional Insights (Revenue, USD Bn, 2021 - 2033)
- North America
- U.S.
- Canada
- Latin America
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Europe
- Germany
- U.K.
- Spain
- France
- Italy
- Russia
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Australia
- South Korea
- ASEAN
- Rest of Asia Pacific
- Middle East
- GCC Countries
- Israel
- Rest of Middle East
- Africa
- South Africa
- North Africa
- Central Africa
- North America
- Key Players Insights
- Pfizer Inc.
- Sanofi
- Baxter International Inc.
- Braun Melsungen AG
- Aspen Pharmacare
- LEO Pharma A/S
- Reddy’s Laboratories Ltd.
- Fresenius Kabi
- Teva Pharmaceutical Industries Ltd.
- Shenzhen Hepalink Pharmaceutical Group Co., Ltd.
Sources
Primary Research Interviews
- Cardiologists, nephrologists, and critical care specialists from hospitals and cardiac care centers
- Hospital pharmacists and anticoagulant procurement managers involved in injectable drug supply
- Dialysis center administrators and healthcare professionals managing hemodialysis procedures
- Pharmaceutical manufacturers specializing in anticoagulants and sterile injectable formulations
- Clinical researchers and investigators involved in thrombosis and coagulation therapy studies
Stakeholders
- Heparin sodium manufacturers and injectable anticoagulant suppliers (e.g., Pfizer Inc., Baxter International Inc., B. Braun Melsungen AG)
- End-use Sectors
- Hospitals and Specialty Cardiac Care Centers
- Dialysis Centers and Nephrology Clinics
- Ambulatory Surgical Centers
- Retail and Hospital Pharmacies
- Regulatory & Health Bodies: U.S. Food and Drug Administration (FDA) – anticoagulant approvals and injectable drug regulations, European Medicines Agency (EMA) – guidelines for anticoagulant safety and manufacturing compliance, World Health Organization (WHO) – cardiovascular disease and thrombosis burden statistics, Centers for Disease Control and Prevention (CDC) – hospitalization and cardiovascular disease data, Central Drugs Standard Control Organization (CDSCO) – pharmaceutical manufacturing and injectable drug approvals
Databases
- ClinicalTrials.gov – studies on anticoagulants, thrombosis treatment, and heparin therapies
- PubMed – research on coagulation disorders, heparin safety, and anticoagulant efficacy
- WHO Global Health Observatory – cardiovascular and chronic disease epidemiology data
Magazines
- Nature Reviews Cardiology – advancements in cardiovascular and anticoagulant therapies
- Pharmaceutical Technology – sterile injectable manufacturing and supply chain trends
- Drug Discovery Today – anticoagulant innovation and pharmaceutical R&D developments
Journals
- Journal of Thrombosis and Haemostasis
- Blood Journal
- The Lancet Haematology
- American Journal of Cardiovascular Drugs
- Clinical and Applied Thrombosis/Hemostasis
Newspapers
- Financial Times – pharmaceutical supply chain and injectable drug market developments
- The Wall Street Journal (Health Section) – hospital drug shortages and healthcare investments
- The Guardian (Health & Science) – cardiovascular disease burden and healthcare access trends
Associations
- International Society on Thrombosis and Haemostasis (ISTH)
- American Society of Hematology (ASH)
- American College of Cardiology (ACC)
- Renal Physicians Association (RPA)
Public Domain Sources
- National Institutes of Health (NIH) – thrombosis, coagulation, and anticoagulant therapy research
- Centers for Disease Control and Prevention (CDC) – cardiovascular disease and hospitalization statistics
- World Health Organization (WHO) – chronic disease prevalence and healthcare burden data
- National health ministries (e.g., U.S., Germany, China, India) – hospital infrastructure and disease burden statistics
Proprietary Elements
- CMI Data Analytics Tool, Proprietary CMI Existing Repository of information for last 10 years.
Share
Share
About Author
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients