The Community-acquired Bacterial Pneumonia (CABP) Treatment Drugs Market is anticipated to grow at a CAGR of 7.7% with USD 1,926.8 Mn in 2026 and is expected to reach USD 3,207.6 Mn in 2033. The Community‑Acquired Bacterial Pneumonia (CABP) treatment drugs market reflects a substantial global health burden, driven by high incidence and hospitalization rates for bacterial CAP and ongoing antibiotic use. CABP remains a leading cause of infectious morbidity and mortality, with annual hospitalization rates up to ~340 per 100,000 adults reported in recent U.S. data. Treatment patterns rely heavily on empiric antibiotic therapy recommended in 2026 CDC guidance on pneumonia, highlighting continuing clinical demand.
Pleuromutilin Antibiotic is projected to account for the largest share of community-acquired bacterial pneumonia (CABP) treatment drugs market in 2026, representing approximately 35.0% of the total volume. The pleuromutilin antibiotic segment leads the community‑acquired bacterial pneumonia (CABP) treatment drugs market because it delivers novel, broad‑spectrum activity against key CABP pathogens with low resistance, addressing rising resistance to older antibiotics. Lefamulin (Xenleta) developed by Nabriva Therapeutics and approved by the U.S.FDA in August 2019 and the European Medicines Agency (EMA) in 2020 continues to show strong in‑vitro efficacy against Streptococcus pneumoniae, Haemophilus influenzae and Staphylococcus aureus with resistance rates of 0–2.6 % in 2026 data, supporting clinician confidence and uptake. Regulatory endorsements and real‑world surveillance reinforce pleuromutilins as a preferred option in both hospital and outpatient CABP settings, sustaining their market dominance through 2025–26.
(Source: NCBI)
Based on route of administration, cloud dominate the market, accounting for a significant 65.0% share in 2026, because most CABP cases are effectively managed with oral antibiotics, which broaden access and reduce healthcare burden. According to WHO guidance (updated 2024), “most cases of pneumonia require oral antibiotics, often prescribed at health centres and treatable at the community level,” highlighting first‑line choices like amoxicillin dispersible tablets. Additionally, Medscape’s March 27, 2025 clinical overview notes that mild to moderate CAP patients can be treated entirely with oral antibiotics either as outpatients or inpatients without compromising outcomes. This preference supports outpatient care, reduces hospital stays, and improves patient adherence, reinforcing oral therapy’s leading role.
Hospital Pharmacies account for the largest share of 50.0% in 2026 due to because CABP frequently leads to hospital admissions where complex antibiotic management and stewardship are critical. For instance, a 2025 U.S. study reported ~340 annual hospitalizations per 100 000 adults for all‑cause community‑acquired pneumonia (CAP) a proxy for CABP burden in clinical settings. Additionally, Medscape data from March 27, 2025 estimated nearly 967,470 U.S. adults aged 65+ were hospitalized annually from CAP, driving inpatient drug use and pharmacist involvement. Within hospitals, pharmacists ensure medication accuracy, manage procurement and inventory, and support antimicrobial stewardship programs to optimize CABP antibiotic effectiveness and safety, reducing errors and resistance risk. Hospital pharmacies also handle specialized antibiotic dispensing and dosing adjustments, enhancing clinical care quality. These roles make them central to CABP drug distribution and drive the hospital pharmacist segment’s dominant market share.
(Source: American Medical Association; Medscape)
Rising prevalence of community-acquired bacterial pneumonia has driven the community-acquired bacterial pneumonia (CABP) treatment drugs market growth over the forecast period. The rising prevalence of community‑acquired bacterial pneumonia (CABP) significantly fuels demand for effective treatment drugs, as higher disease burden increases prescriptions and hospital use. For instance, according to recent analyses published by the ResearchGate show overall CABP incidence post‑COVID‑19 surged to ~42.1 cases per 1,000 person‑years in 2026, with high incidence particularly among older adults and comorbid patients, reflecting broader CABP trends. In the U.S. in 2025, estimated annual CAP hospitalizations reached ~340 per 100,000 adults, with pneumococcal infections contributing substantially, underscoring persistent bacterial disease burden. CDC mortality data (2024) reported ~41,627 pneumonia deaths at 12.2 per 100,000 population, highlighting serious outcomes driving therapeutic use. As CABP cases rise globally, utilization of targeted antibiotics increases, expanding the CABP treatment drugs market.
Increasing launch of the new product by the key players operating in the market has driven the community-acquired bacterial pneumonia (CABP) treatment drugs market growth over the forecast period. For instance, in February 2026, the Wockhardt group, pharmaceutical company developed nafithromycin. This new macrolide was created especially to treat community-acquired bacterial pneumonia (CABP). The Biotechnology Industry Research Assistance Council (BIRAC), a non-profit Section 8 Public Sector Enterprise of the Department of Biotechnology (DBT), provided partial funding for the phase III clinical investigation of nafithromycin in India. The Central Drugs Standard Control Organization (CDSCO) has granted M/s Wockhardt Limited marketing authority to sell and distribute 400 mg Nafithromycin Tablets for the treatment of people (≥18 years) with CABP.
A robust product pipeline fuels growth in the Community‑Acquired Bacterial Pneumonia (CABP) treatment drugs market by advancing newer antibiotics through late‑stage clinical evaluation, expanding therapeutic options and addressing resistance concerns. For instance, in April 2025, Paratek Pharmaceuticals, Inc., pharmaceutical company, revealed the Data from multiple new studies of NUZYRA (omadacycline) will be presented at ESCMID Global 2025, the Congress of the European Society of Clinical Microbiology and Infectious Diseases, which will take place in Vienna, Austria, from April 11–15, 2025. Furthermore, ClinicalTrials.gov lists NCT07099976, a Phase 3 trial initiated in March 2025 by the University of California, San Diego evaluating optimized antibiotic care pathways in non‑severe CAP, showing ongoing commitment to refining treatment strategies with active status in 2026. Such late‑stage studies attract investment and support adoption of improved therapeutic regimens, strengthening market dynamics.
(Source: Paratek Pharmaceuticals, Inc.)
|
Current Event |
Description and its Impact |
|
Government Initiatives to Combat CABP Through Innovation and Funding |
|
|
Regulatory Shifts Fueling Growth in CABP Treatment Market |
|
|
Increased Public Funding for CABP Treatment Research |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Clinical practice guidelines are evolving to optimize antibiotic use and treatment strategies for community‑acquired pneumonia, directly impacting CABP drug prescribing patterns. For example, the American Thoracic Society released updated CAP guidelines in 2025, recommending more selective empiric antibiotic therapy, appropriate treatment durations, and refined use of adjunct therapies like corticosteroids. These updates are driving clinicians toward more targeted antibiotic choices and influencing developers to align drug profiles with current evidence on efficacy and safety.
(Source: MJH Life Sciences)
Antibiotic resistance remains a central trend shaping CABP treatment strategies. In October 2025, WHO warned that one in six laboratory‑confirmed bacterial infections show antibiotic resistance, with resistance rising in ~40% of tested samples across over 100 countries. This trend propels greater stewardship efforts and demand for new antibiotics with novel mechanisms, influencing market focus toward safer, resistance‑mitigating therapies.
Updated evidence‑based clinical guidelines are influencing how CABP is diagnosed and treated globally, with a shift toward more targeted and optimized antibiotic use. For instance, the American Thoracic Society (ATS) released new CAP guidelines in 2025, refining recommendations on antibiotic use, treatment duration, and imaging strategies to improve outcomes. These guidelines emphasize shorter courses for stable patients and more selective empiric antibiotic choices, reflecting evolving evidence and encouraging adoption of more appropriate CABP drug regimens. This trend drives demand for antibiotics aligned with updated therapeutic protocols, enhancing clinical efficacy and influencing drug preference in treatment guidelines, thereby expanding appropriate CABP drug utilization.
In February 2026, the IDSA/PIDS 2026 pediatric CAP guidelines were updated, offering more specific management guidance for children older than 3 months with community‑acquired pneumonia. These include recommendations for antibiotic use tailored to age groups and pathogen considerations. Clear pediatric guidance supports rational antibiotic selection and use in younger populations, expanding the treatable patient base for effective CABP drugs and improving market demand across age segments.
To learn more about this report, Request Free Sample
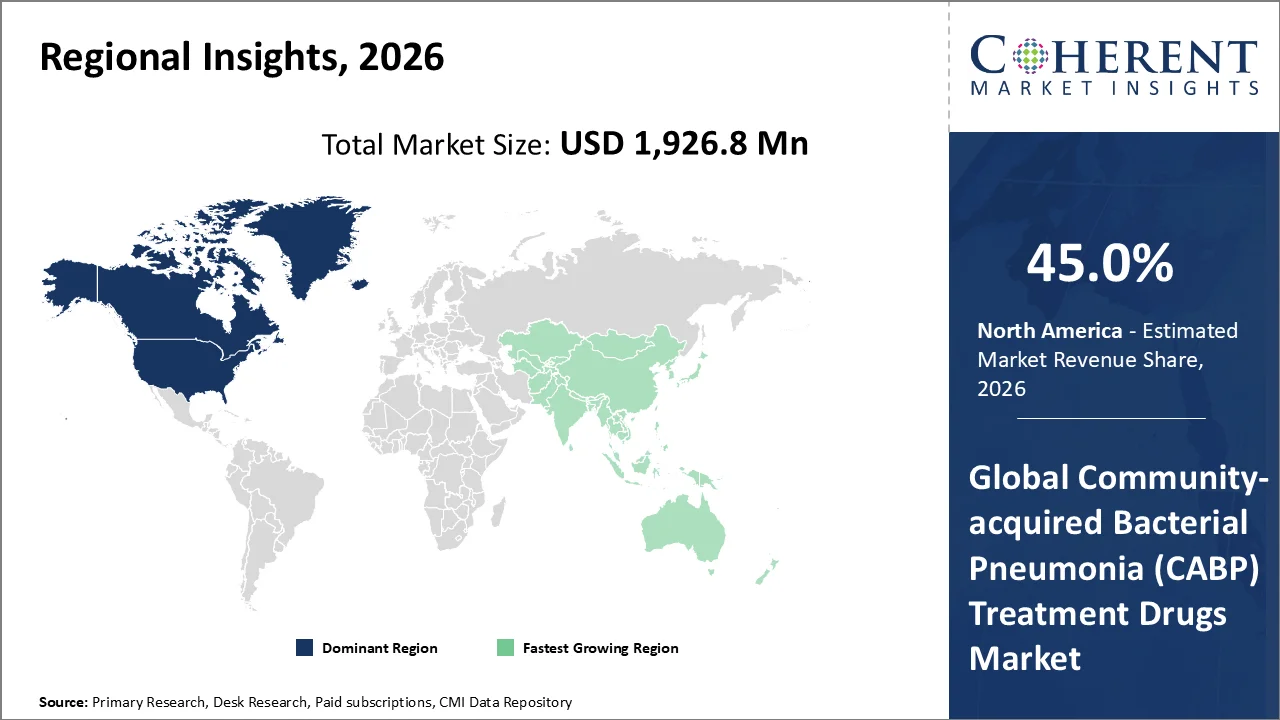
North America account 45.0% market share in 2026, supported by high disease burden and advanced healthcare infrastructure. According to the U.S. CDC’s 2026 pneumonia guidelines, pneumonia remains a significant public health concern, with more than 41,000 U.S. deaths reported from pneumonia in 2022 (adults being most affected), reinforcing ongoing treatment demand. Epidemiologic studies by NIH in the U.S. show high annual hospitalization rates for community‑acquired pneumonia (about 340 hospitalizations per 100,000 adults, with higher rates among older adults), indicating persistent clinical workload that drives utilization of effective CABP therapies. The region’s dominance is further supported by advanced healthcare infrastructure, widespread antibiotic stewardship programs, strong regulatory frameworks (including FDA approvals for novel CABP drugs), and extensive public health initiatives that enhance disease detection, management, and treatment uptake throughout North America.
The Asia-Pacific region is poised to be as the fastest-growing region through 2026-2033, owing to increasing cases of community-acquired bacterial pneumonia, government initiatives and developing healthcare infrastructure. In the Asia‑Pacific region, countries like India have documented high pneumonia burden and ongoing efforts to combat this via expanded pneumococcal vaccination within the Universal Immunization Programme (UIP) as reviewed in March 2025 research on pneumococcal disease and vaccination strategies. Additionally, WHO pneumonia vaccine policy resources emphasize pneumococcal immunization to reduce severe respiratory infections. Strategic public health initiatives in India, China, and Japan improving vaccine access and healthcare infrastructure are accelerating diagnosis and treatment uptake, contributing to Asia‑Pacific’s rapid growth in the CABP treatment drugs market.
Moreover, high admission of children in hospital due to community-acquired bacterial pneumonia (CABP) treatment drugs has driven the market growth in Asia Pacific region. For instance, in 2024, in Southeast Asia, WHO research shows a severe CAP incidence of ~11.7 to 17.8 per 1,000 child‑years among hospitalized children under 5 illustrating the ongoing pediatric burden that feeds into drug demand and healthcare utilization strategies. Furthermore, a 2025 review of CABP in the Asia‑Pacific region reports that aging populations, dense urbanization, and limited healthcare access contribute to a heavy CAP burden, with bacterial pathogens such as Streptococcus pneumoniae, Mycoplasma pneumoniae, and Haemophilus influenzae frequently implicated.
(Source: WHO; Asian Journal of Pediatric Research; ResearchGate)
The U.S. community-acquired bacterial pneumonia (CABP) treatment drugs market dominates North America due to its high disease burden, with over 41,000 deaths from pneumonia annually (2022). Hospitalization rates of 340 per 100,000 adults (2024) highlight the ongoing clinical need.
Furthermore, the U.S. remains the dominant country in the North American Community‑Acquired Bacterial Pneumonia (CABP) treatment drugs market due to high clinical burden, strong government initiatives, and progressive regulatory shifts. According to the CDC’s 2026 pneumonia management guidelines, pneumonia prevention and appropriate treatment remain key public health priorities, sustaining demand for CABP drugs. Additionally, the CDC updated its outpatient antibiotic stewardship guidance in February 2026 to standardize appropriate antibiotic use and reduce resistance in both hospital and outpatient settings, strengthening treatment quality and drug utilization.
From a regulatory standpoint, the U.S. FDA launched the Commissioner's National Priority Voucher (CNPV) pilot program in 2025, enabling rapid review and approval of priority antibiotics for example, Augmentin XR was approved under this program in December 2025, significantly shortening approval timelines and supporting domestic antibiotic availability for CABP care.
These combined public health strategies, stewardship initiatives, and regulatory innovations reinforce the U.S.’s leadership in shaping CABP treatment adoption and market demand.
India has become the biggest player in the Asia Pacific community-acquired bacterial pneumonia (CABP) treatment drugs market in 2026. India is a dominant country in the Asia‑Pacific Community‑acquired Bacterial Pneumonia (CABP) treatment drugs market because it carries a disproportionately high disease burden and active public health responses that drive demand for treatments. According to WHO estimates published via NCBI in 2025, India accounts for ~23% of the global pneumonia burden and ~36% of the WHO South‑East Asia regional burden, with approximately 4 million CAP cases annually and about 20% requiring hospitalization, underscoring major clinical need. Pneumonia is a leading cause of mortality in children under five in India, with studies indicating substantial child deaths from the disease. Government initiatives such as pneumococcal vaccination scale‑ups under India’s public immunization programs and enhanced treatment protocols push broader antibiotic access and utilization. These factors significant disease prevalence, hospitalization rates, mortality impact, and strong healthcare responses position India as the fastest‑growing and dominant CABP treatment market in the Asia‑Pacific region.
(Source: NCBI; ResearchGate)
Some of the major key players in global community-acquired bacterial pneumonia (CABP) treatment drugs market are Nabriva Therapeutics, Paratek Pharmaceuticals, Inc., Melinta Therapeutics, Inc., Allergan plc, Bayer AG, Lupin Pharmaceuticals, Inc., Mylan N.V., Sanofi S.A., Pfizer Inc., Dainippon Sumitomo Pharma, Takeda Pharmaceutical Company Limited, and Basilea Pharmaceutica International AG.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1,926.8 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7.7% | 2033 Value Projection: | USD 3,207.6 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Nabriva Therapeutics, Paratek Pharmaceuticals, Inc., Melinta Therapeutics, Inc., Allergan plc, Bayer AG, Lupin Pharmaceuticals, Inc., Mylan N.V., Sanofi S.A., Pfizer Inc., Dainippon Sumitomo Pharma, Takeda Pharmaceutical Company Limited, and Basilea Pharmaceutica International AG. |
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients