Global Neurology Drugs Market Size and Forecast: 2026-2033
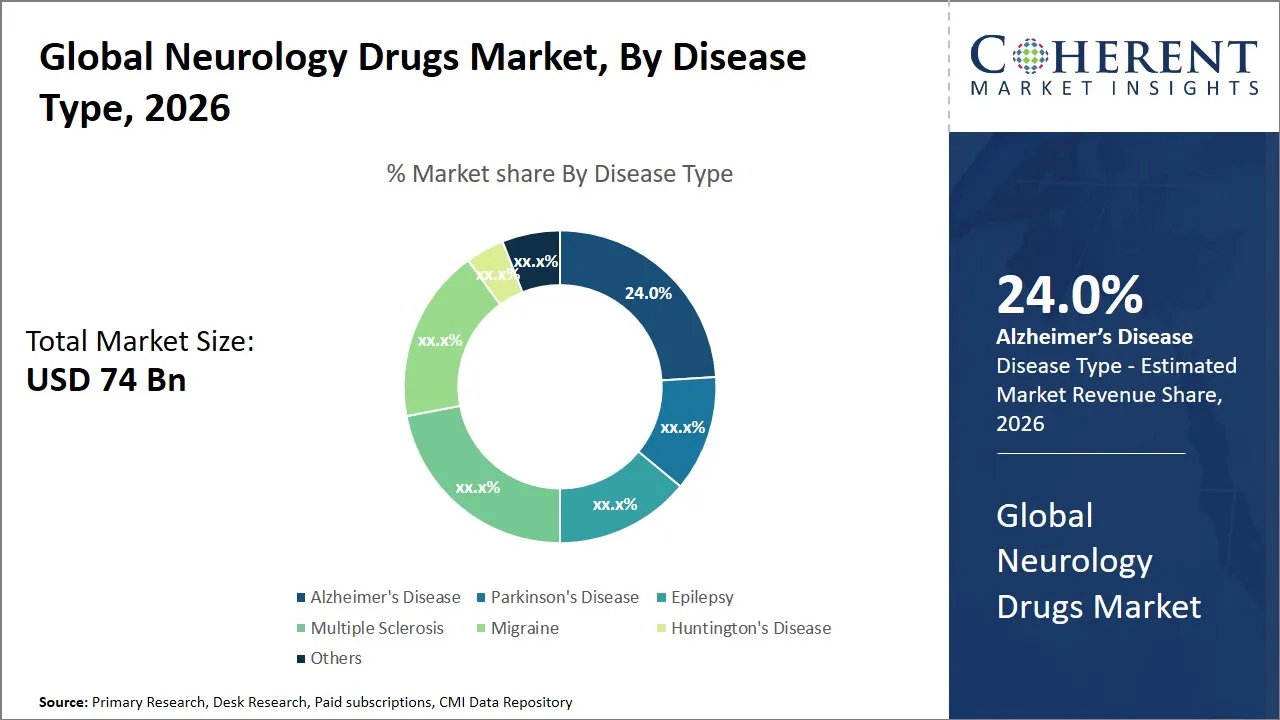
The global neurology drugs market is estimated to be valued at USD 74 Bn in 2026 and is expected to reach USD 104 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 4.98% from 2026 to 2033. The consistent expansion observed in the market can be attributed to the increasing incidence of neurological disorders, progress in pharmaceutical research, and heightened cognizance among both patients and healthcare providers. Furthermore, market dynamics are influenced by the expanding elderly demographic and enhancements in healthcare systems within developing economies.
Key Takeaways of the Global Neurology Drugs Market
- The Alzheimer’s disease segment is expected to dominate the global neurology drugs market by disease type, accounting for approximately 24.0% of the total market share in 2026.
- The antiepileptics segment is expected to dominate the global neurology drugs market by drug class, accounting for approximately 30.0% of the total market share in 2026.
- The oral segment is expected to dominate the global neurology drugs market by route of administration, accounting for approximately 70.0% of the total market share in 2026.
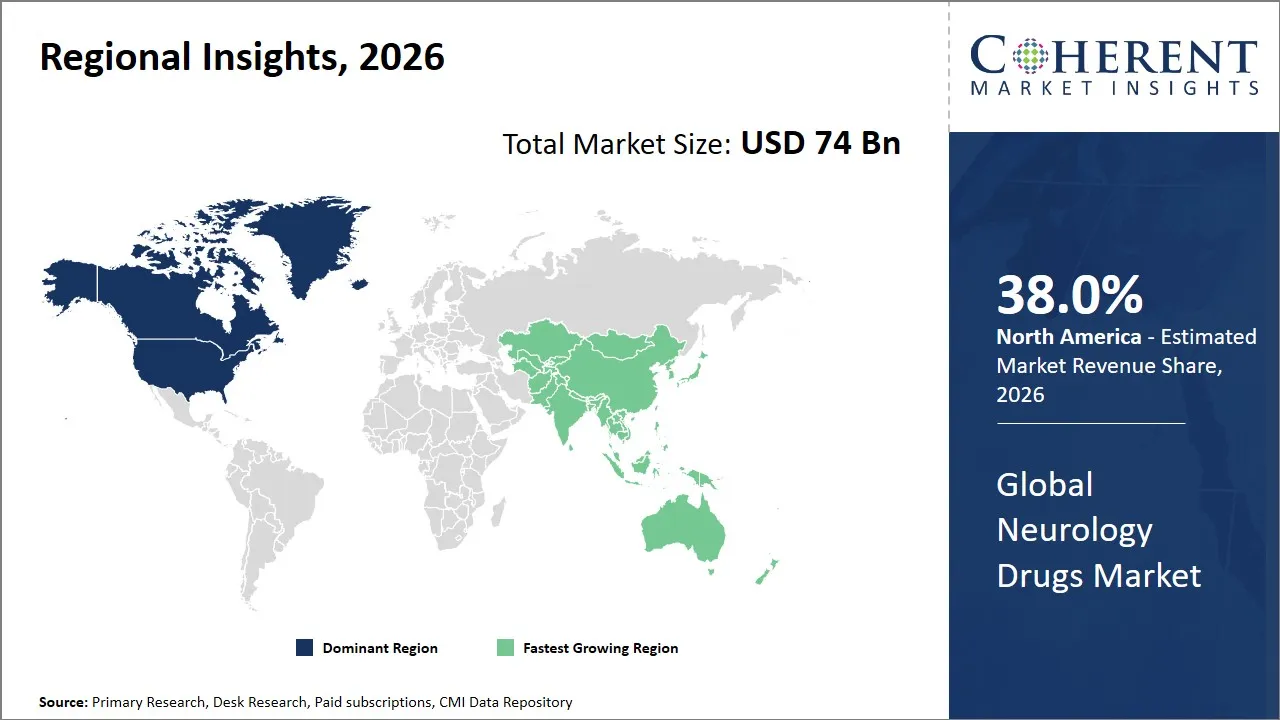
- North America is expected to dominate the global neurology drugs market in 2026, accounting for approximately 38.0% of the total market share.
- Asia Pacific is anticipated to witness the fastest growth, holding a market share of 22.0% in 2026.
Market Overview
- Neurology drugs are defined as pharmaceutical products used for the treatment and management of diseases related to the central nervous system and peripheral nervous system, such as Alzheimer's, Parkinson's, epilepsy, multiple sclerosis, migraines, and many other neurodegenerative disorders.
- The market for neurology drugs is growing rapidly, as the prevalence of neurological disorders is increasing worldwide. In addition, the elderly population is also at high risk of developing such diseases. The market is also growing due to the increased ability to diagnose such diseases, as well as the awareness regarding the diseases.
- In the past few years, the market for neurology drugs has been witnessing tremendous growth. The growth is due to the continuous developments in the pharmaceutical industry, including biologics, monoclonal antibodies, and gene therapies. In addition, the focus on disease modification is also driving the growth of the market. Investments in neuroscience research is also propelling the growth of the market.
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
Expansion of Biosimilars in Multiple Sclerosis (MS) |
|
|
Strategic M&A Strengthening Central Nervous System (CNS) Pipelines |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Why Does the Alzheimer’s Disease Segment Dominate the Global Neurology Drugs Market in 2026?
The Alzheimer’s disease segment is anticipated to hold a market share of 24.0% in 2026, attributed to the demographic changes and rising cases of neurological disorders. The rising population of aged people globally, especially in developed countries and rapidly aging emerging countries, has caused a surge in Alzheimer’s, which is the most common type of dementia.
For instance, according to Alzheimer’s Disease International, every 3 seconds, someone in the world develops dementia, with over 55 million people living with dementia worldwide as of 2020. The figures are also set to rise to 78 million by 2030 and 139 million by 2050, almost doubling every 20 years. Furthermore, 60% of the people living with dementia worldwide are from low- and middle-income countries, with the figures set to rise to 71% by 2050, with the largest increases occurring in countries such as China, India, and the rest of the Asia Pacific region.
(Source: Alzheimer’s Disease International)
In addition, Alzheimer's disease is a complex drug target, which has led to a significant investment in both symptomatic and disease-modifying therapies. Recent advancements in the understanding of some of the key pathological mechanisms, such as amyloid beta plaques and tau protein tangles, have further accelerated drug development programs in these areas.
At the same time, increased policy support, funding initiatives, and awareness campaigns have resulted in a general improvement in diagnosis and treatment access. The increased trend in using early diagnostic tools is further contributing to a larger patient pool, while increased regulatory support is helping in faster approvals of new treatments. All these aspects further reinforce the highest market share of the Alzheimer's disease segment in the neurology drugs market.
Why Does the Antiepileptics Segment Dominate the Global Neurology Drugs Market in 2026?
The antiepileptics segment is projected to hold a market share of 30.0% in 2026, attributed to the extensive use of these drugs and consistent demand for them among a wide population of patients. Epilepsy is considered to be one of the most prevalent neurologic disorders globally and is seen in all age groups of the population in varying degrees of severity. Hence, a wide spectrum of drugs is required for the treatment of this condition. In addition to this, these drugs are also being increasingly used for the treatment of other conditions such as bipolar disorder, neuropathic pain, and migraine prevention which reinforces its consistent market demand.
Furthermore, constant innovation in anti-epileptic drug therapy is contributing to the growth of the segment. The fact that, drug-resistant epilepsy has led to the creation of new-generation anti-epileptic drugs that have better mechanisms of actions, safety, and tolerability. In addition, improvements in drug delivery systems have enhanced the pharmacokinetics of these drugs, thus reducing side effects. Consequently, there is patient compliance. Moreover, the availability of generics has made these drugs more accessible, especially in developing countries. All these factors have ensured that anti-epileptics remain dominant in the neurology drugs segment.
The Oral Segment Dominates the Global Neurology Drugs Market
The oral segment is projected to hold a market share of 70.0% in 2026, attributed to their ease of use, cost-effectiveness, and high patient compliance advantages. These are preferred forms of administration in neurology drugs due to their ease of self-administration by patients without requiring medical supervision, as in the case of chronic neurological diseases like epilepsy, Parkinson’s disease, and multiple sclerosis. These diseases require long-term pharmacotherapy, and therefore, oral forms are more suitable due to their ease of administration, which is crucial in neurological diseases as noncompliance can lead to a worsening of the condition.
Moreover, oral forms have shown significant flexibility in dosage forms and design, such as tablets, capsules, and even liquid suspensions for different patient groups such as pediatric and geriatric patients. The advantage of combination formulations in a single dosage form will also improve patient compliance by limiting the number of pills to be taken. Though injectable and topical formulations are important for some diseases and conditions, these are limited by their costs and complexities of use. Therefore, oral forms of medications have the largest market share due to ease of access and lower costs of production and acceptance among patients.
Global Regulatory Frameworks and Guidelines Influencing the Neurology Drugs Market
|
Region |
Key Regulatory Frameworks & Policies |
Market-Specific Impact on Neurology Drugs |
|
North America |
Accelerated approval pathways (Fast Track, Breakthrough Therapy), orphan drug incentives, strong reimbursement integration (Medicare/insurance-driven access) |
Enables early commercialization of innovative therapies (e.g., Alzheimer’s biologics, rare disease drugs) but also introduces pricing pressure through payer scrutiny and reimbursement controls |
|
Europe |
Centralized drug approval system, orphan drug regulations, strong Health Technology Assessment (HTA) frameworks |
Ensures pan-European access to neurology drugs, but strict cost-effectiveness evaluations delay or restrict uptake, especially for high-cost biologics |
|
Asia Pacific |
Expedited approval reforms (China, Japan), national reimbursement inclusion (NRDL in China), increasing regulatory harmonization |
Drives faster approval timelines and expanding patient access, with strong growth potential due to improving healthcare infrastructure and policy support |
|
Middle East & Africa |
Import-dependent regulatory systems, limited local approval pathways, growing public healthcare investments |
Results in slower adoption of advanced therapies, but government-led healthcare expansion is improving access to essential neurology drugs |
|
Latin America |
Evolving regulatory frameworks, reliance on reference pricing, gradual adoption of biosimilars |
Leads to delayed product launches but increasing affordability, particularly for generic and biosimilar neurology drugs |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Regional Insights
To learn more about this report, Request Free Sample
North America Neurology Drugs Market Analysis and Trends
The North America region is projected to lead the market with a 38.0% share in 2026, attributed to the strong healthcare system, high R&D investments, and support from the government for the treatment of neurology disorders. The region has a well-established pharmaceutical environment in terms of drug development. There are a large number of multinational corporations in the region, along with drug development companies focused on neurology drugs. The government has a conducive environment for drug development through expedited approval procedures and intellectual property rights.
In addition, the high incidence of neurology disorders in the aging population of these countries has fueled the demand for advanced neurology drugs. Companies such as Pfizer, Biogen, and Abbvie have a strong foothold in the neurology drugs market in North America. These companies are continuously improving their drug portfolios by launching new neurology drugs, and investing in the development of innovative neurology drugs for neurology disorders such as multiple sclerosis, Alzheimer’s, and Parkinson’s.
For instance, in January 2025, Merz Therapeutics commercialized FAMPYRA (fampridine) in Canada as part of a broader multi-regional strategy, further increasing access to symptomatic management of multiple sclerosis walking impairments. This demonstrates continued investment in improving neurology therapy availability in North America.
(Source: Merz Therapeutics)
Asia Pacific Neurology Drugs Market Analysis and Trends
Asia Pacific is anticipated to witness the fastest growth, holding a market share of 22.0% in 2026, driven by increasing health awareness among the population and growing government healthcare expenditure in the region. Countries such as China, India, and Japan are experiencing an increasing number of cases of neurological disorders due to an aging population and changing lifestyles.
Government initiatives are also contributing to the growing demand for neurology drugs. The presence of local players such as Sun Pharma and Aurobindo Pharma, along with global players such as Novartis and Takeda Pharmaceutical, is increasing the penetration of neurology drugs in the market. In addition, an increasing number of research activities and strategic alliances between regional and global players are also contributing to the growing demand for neurology drugs.
Neurology Drugs Market Outlook for Key Countries
How is the U.S. Helping in the Growth of the Neurology Drugs Market?
U.S. holds a vital position in the neurology drugs market, especially considering the advanced healthcare delivery systems and substantial investment in R&D. The regulatory authorities in the country, like the U.S. FDA, accelerate the approval of drugs for the treatment of neurological disorders. The country has witnessed the launch of breakthrough drugs in Alzheimer’s and multiple sclerosis by major pharmaceutical companies like Biogen and Eli Lilly. The country has an established insurance system, which, combined with patient awareness, boosts the market for advanced neurology drugs.
For instance, in November 2025, Sandoz debuted TYRUKO (natalizumab-sztn) in the U.S., a significant milestone as the inaugural biosimilar approved for multiple sclerosis. This launch provides patients with more affordable options compared to current biologic treatments, thereby improving accessibility. The introduction of TYRUKO is anticipated to stimulate price competition and foster the acceptance of biosimilars in the neurology drugs market.
(Source: Sandoz)
How is China Helping in the Growth of the Neurology Drugs Market?
The neurology drugs market in China is growing significantly due to the increasing elderly population and the government's focus on enhancing neurological care services. The government's efforts to improve the process of approving drugs and enhancing innovation have attracted multinational companies like Novartis and Johnson & Johnson to strengthen their presence. At the same time, companies like Sino Biopharmaceutical are investing heavily in the research and development of neurology drugs. The increasing per capita health expenditure and number of hospitals are also contributing to the growth of the market.
Key Drivers for the Growth of the Germany Neurology Drugs Market
The neurology drugs market in Germany can be described as a robust pharmaceutical manufacturing industry and universal healthcare through the national health insurance scheme. The government has been supportive of the pharmaceutical industry, which has led to improvements in drug development in neurology through joint research with academic institutions. Companies like Bayer and Merck KGaA are actively involved in improving neurology drugs for a large patient pool in Europe. Germany is a key player in the European landscape of clinical trials for neurology drugs.
Japan Neurology Drugs Market Trends
Japan remains the leader in the innovation of neurology drugs in the Asia Pacific region, driven by the country’s aging population and emphasis on technological advancements. The government has created an environment conducive to the development of regenerative and precision medicine in the treatment of neurological diseases.
Takeda Pharmaceutical and other major players have been at the forefront in the development of drugs for neurodegenerative diseases, making Japan the leading country in the development of advanced treatment solutions in the field of neurology.
Reimbursement and Pricing Landscape in the Global Neurology Drugs Market
|
Region |
Key Authorities |
Reimbursement Framework |
Impact on Neurology Drugs Market |
|
North America |
Centers for Medicare & Medicaid Services (CMS), Private Insurers, PBMs |
Mixed system with public (Medicare/Medicaid) and private insurance; formulary-based coverage for biologics, specialty CNS drugs, and chronic therapies |
High reimbursement coverage supports strong adoption of advanced neurology drugs (e.g., Alzheimer’s biologics, MS therapies), but pricing pressure, prior authorization, and co-pay variability impact patient access |
|
Europe |
National Health Systems, Statutory Health Insurance Bodies, HTA Agencies (e.g., NICE, IQWiG) |
Universal healthcare with government-funded reimbursement; strict HTA-based pricing and reference pricing models |
Broad access ensures stable demand; however, stringent cost-effectiveness criteria delay uptake of high-cost therapies and promote biosimilar adoption in segments like MS |
|
Asia Pacific |
National Healthcare Security Administration (China), PMDA/MHLW (Japan), Public Insurance Schemes |
Expanding public insurance coverage; inclusion in reimbursement drug lists (e.g., NRDL in China); price negotiations and periodic revisions |
Increasing reimbursement coverage drives market expansion, though aggressive price negotiations reduce margins; strong volume growth in emerging markets |
|
Middle East & Africa |
Government Health Authorities, Private Insurers |
Government-funded healthcare in GCC; limited reimbursement frameworks in Africa; high reliance on imports |
Access to advanced neurology therapies is uneven; high-income regions show better adoption, while other areas rely on essential and generic drugs |
|
Latin America |
Public Health Systems, Social Security Funds, Private Insurers |
Hybrid systems with public funding and out-of-pocket components; reliance on reference pricing and generics |
Limited reimbursement leads to delayed adoption of premium neurology drugs; generics and biosimilars dominate improving affordability but limiting revenue growth |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Market Players, Key Developments, and Competitive Intelligence
To learn more about this report, Request Free Sample
Key Developments
- On March 25, 2026, Insilico Medicine and Tenacia Biotechnology deepened their collaboration with a fresh agreement. This deal is projected to elevate their partnership's worth to US$ 94.75 million, primarily aimed at propelling early-stage drug development using artificial intelligence. The move underscores the growing significance of AI in neurology, a field where it's already making a noticeable difference in drug discovery.
- On March 23, 2026, Ionis Pharmaceuticals revealed that the U.S. FDA accepted its application for a new drug named zilganersen for the treatment of Alexander disease (AxD). The development is an important milestone for RNA-targeting drugs for rare CNS disorders. The development does not have a significant influence on overall revenue but is an important development in the neuroscience pipeline.
Top Strategies Followed by Global Neurology Drugs Market Players
|
Player Type |
Strategic Focus |
Examples |
|
Established Market Leaders |
Established players in the neurology drugs market prioritize high R&D investments to develop disease-modifying therapies, particularly for complex conditions such as Alzheimer’s disease and multiple sclerosis. They focus on advanced biologics, monoclonal antibodies, and precision medicine approaches to improve clinical outcomes and address unmet needs. These companies leverage large-scale clinical trials, strong regulatory expertise, and global commercialization capabilities to maintain market leadership. Additionally, strategic acquisitions, pipeline diversification, and lifecycle management strategies are adopted to sustain long-term growth and competitive advantage across major markets. |
In July 2023, F. Hoffmann-La Roche received U.S. FDA approval for Ocrevus Zunovo, a subcutaneous formulation for multiple sclerosis, enhancing patient convenience and expanding its neurology portfolio. |
|
Mid-Level Players |
Mid-level players focus on strengthening their CNS portfolios through development of disease-modifying and symptomatic therapies targeting high-burden neurological disorders such as Alzheimer’s and Parkinson’s disease. They leverage strategic collaborations, co-development agreements, and regulatory-driven growth strategies to accelerate clinical advancement and commercialization. These companies emphasize innovation in biologics and novel mechanisms of action to compete with larger players while expanding their presence in key markets. |
In January 2023, Eisai and Biogen received U.S. FDA approval for Leqembi (lecanemab) for Alzheimer’s disease, strengthening Eisai’s position in disease-modifying therapies. |
|
Small-Scale Players |
Small-scale players concentrate on niche and underserved neurological indications, particularly in neuropsychiatric and rare CNS disorders, where unmet need is high. Their strategy centers on developing first-in-class or fast-acting therapies with differentiated mechanisms, supported by pipeline-driven growth and accelerated approval pathways. They often rely on licensing, partnerships, and innovation-led positioning to compete with established firms. |
In October 2023, Sage Therapeutics received U.S. FDA approval for Zurzuvae (zuranolone), expanding its CNS portfolio and demonstrating innovation in neuropsychiatric treatment approaches. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Market Report Scope
Neurology Drugs Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 74 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 4.98% | 2033 Value Projection: | USD 104 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
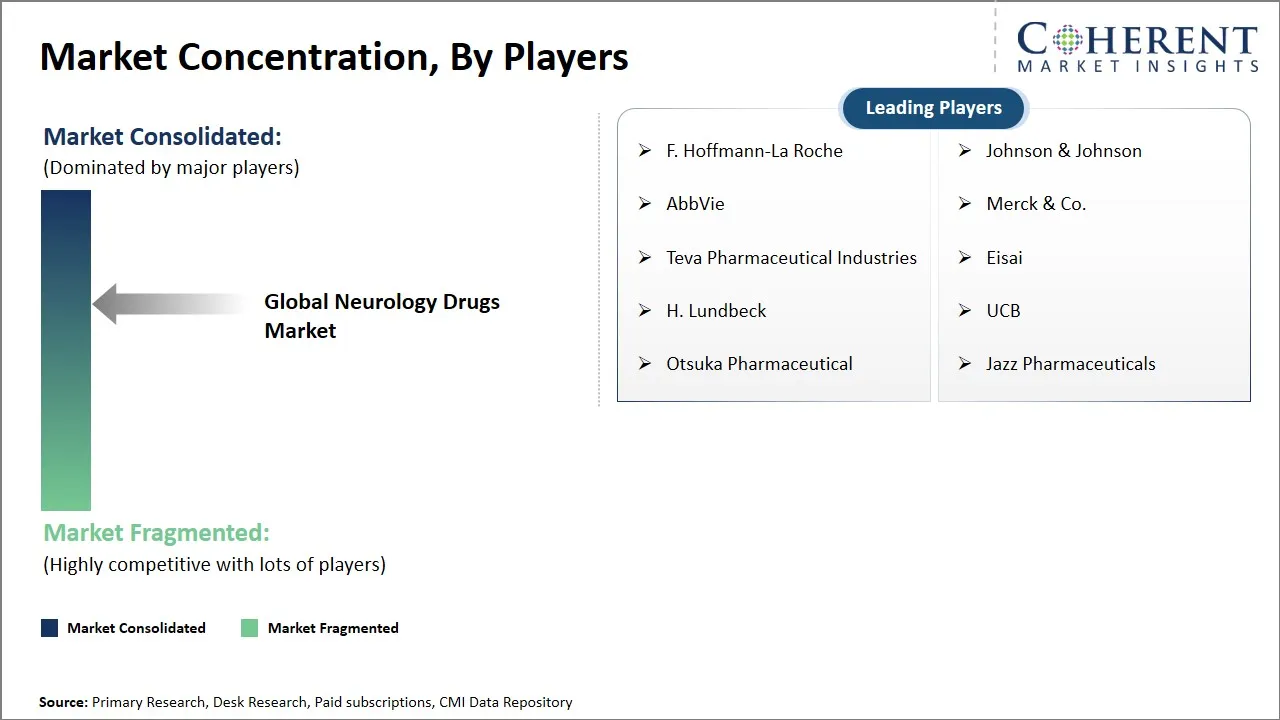
F. Hoffmann-La Roche, Johnson & Johnson, AbbVie, Merck & Co., Teva Pharmaceutical Industries, Eisai, H. Lundbeck, UCB, Otsuka Pharmaceutical, Jazz Pharmaceuticals |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Neurology Drugs Market Dynamics
To learn more about this report, Request Free Sample
Global Neurology Drugs Market Driver - Rising Prevalence of Neurological Disorders
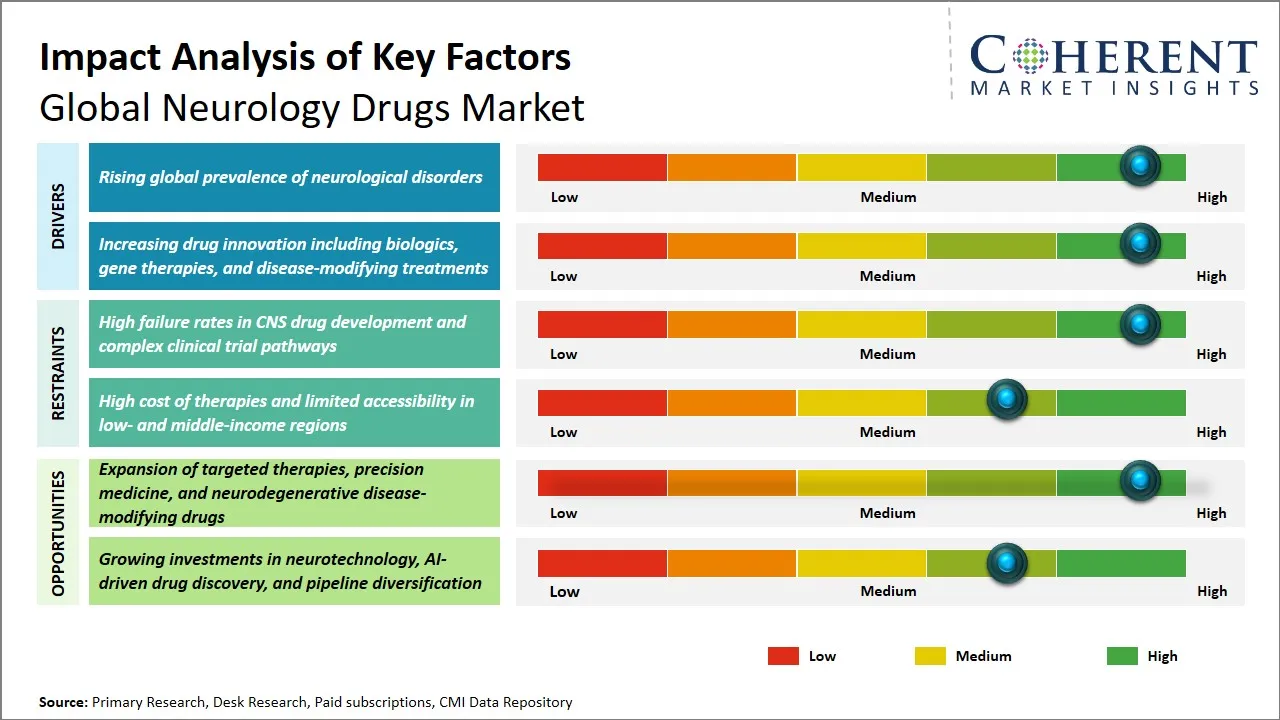
The rise in the number of people suffering from neurological disorders across the world is a key factor that is boosting the growth of the market for neurology drugs. This includes factors such as an aging population, a rise in the number of people suffering from chronic diseases such as Alzheimer's, Parkinson's, multiple sclerosis, and epilepsy. In addition, a number of factors such as lifestyle, environmental factors, and advances in diagnosis have led to a rise in number of people suffering from such disorders, thus boosting growth in the market for neurology drugs. Furthermore, increasing awareness among people, as well as health care providers, concerning early diagnosis and treatment of various types of neurologic disorders, is further propelling growth in the market.
For instance, according to the World Health Organization, neurological disorders affect more than 3 billion people worldwide, which is greater than 40% of the world's population, and are the biggest contributor to illness and disability worldwide. In addition, neurological disorders contribute to 11 million deaths worldwide every year. In addition, the overall burden of neurological disorders worldwide has increased by 18% since 1990 as a result of population growth and aging. In conclusion, more than 80% of the health loss and deaths due to neurological disorders occur in low- and middle-income countries.
(Source: World Health Organization)
Global Neurology Drugs Market Opportunity - Expansion of Targeted Therapies, Precision Medicine, and Neurodegenerative Disease-Modifying Drugs
The global neurology drugs market has huge growth prospects due to the introduction of targeted therapies, precision medicine, and the development of disease-modifying therapies in neurology, which cover neurodegenerative diseases. Recent advancements in the fields of genomics and the identification of biomarkers have made it possible to target neurological diseases more precisely, such as Alzheimer’s, Parkinson’s, multiple sclerosis, amyotrophic lateral sclerosis, or ALS.
Precision medicine has changed the course of drug regimens for these debilitating diseases by personalizing medicine according to the individual’s genetic composition, thus improving the benefits of drugs and reducing side effects. Pharmaceutical companies in the pharmaceutical and biotechnology industry are shifting their focus from merely treating these diseases to developing drugs that can halt the progress of these diseases. This is a major shift in the pharmaceutical industry. Regulatory bodies worldwide are promoting these revolutionary therapies by expediting the approval of these drugs, which is a major company for investing in R&D activities in this field.
For instance, in May 2025, Renovaro Biosciences introduced Augusta, a platform that uses AI to improve how neurological disorders are diagnosed and treated. Augusta employs sophisticated data analysis and machine learning to assist doctors in making decisions and fine-tuning treatment plans. This launch underscores the increasing use of AI in the field of neurology, fostering innovation and pushing forward personalized treatment options.
(Source: Renovaro Biosciences)
Furthermore, the rising incidence of neurodegenerative diseases worldwide, partly due to the aging population, emphasizes the need for effective disease-modifying therapies. Joint initiatives by academic institutions, research bodies, and industry players are helping to expedite the translation of new scientific discoveries into commercialized products. Therefore, the combination of new technologies in neurological drugs and the rising need for these drugs creates a significant opportunity for growth in the field of targeted and precision neurology drugs.
Analyst Opinion (Expert Opinion)
- The neurology drugs market is in a transition phase from symptomatic drugs to disease-modifying drugs. However, the success of these new drugs will depend on clinical differentiation and efficacy in Alzheimer’s and Parkinson’s diseases.
- The neurology drugs market has witnessed rapid innovation in biologics, gene therapy, monoclonal antibody therapies, which is likely to continue in the future. However, the major challenges for these new drugs in the neurology market will be the price pressure and the challenges of reimbursements.
- The major trend in the neurology drugs market is the emphasis on precision medicine, which is likely to change the current drug development paradigm. However, this trend may also increase the complexity in diagnosing the diseases, which may slow down the adoption of these drugs in the short term.
- The major players in the neurology drugs market are likely to consolidate their market leadership through acquisitions. Small players will remain essential for innovation in the neurology drugs market. In the long term, the neurology drugs market growth will not depend on the prevalence of the diseases. Rather, it will depend on the success of new drugs in terms of commercialization.
Market Segmentation
- Disease Type Insights (Revenue, USD Bn, 2021 - 2033)
- Alzheimer’s Disease
- Parkinson’s Disease
- Epilepsy
- Multiple Sclerosis
- Migraine
- Huntington’s Disease
- Others
- Drug Class Insights (Revenue, USD Bn, 2021 - 2033)
- Antiepileptics
- Antipsychotics
- Dopamine Agonists
- Cholinesterase Inhibitors
- NMDA Receptor Antagonists
- Analgesics (Migraine-specific)
- Others
- Route of Administration Insights (Revenue, USD Bn, 2021 - 2033)
- Oral
- Injectable
- Transdermal
- Distribution Channel Insights (Revenue, USD Bn, 2021 - 2033)
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
- Regional Insights (Revenue, USD Bn, 2021 - 2033)
- North America
- U.S.
- Canada
- Latin America
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Europe
- Germany
- U.K.
- Spain
- France
- Italy
- Russia
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Australia
- South Korea
- ASEAN
- Rest of Asia Pacific
- Middle East
- GCC Countries
- Israel
- Rest of Middle East
- Africa
- South Africa
- North Africa
- Central Africa
- North America
- Key Players Insights
- F. Hoffmann-La Roche
- Johnson & Johnson
- AbbVie
- Merck & Co.
- Teva Pharmaceutical Industries
- Eisai
- Lundbeck
- UCB
- Otsuka Pharmaceutical
- Jazz Pharmaceuticals
Sources
Primary Research Interviews
Industry Stakeholders
- Pharmaceutical Manufacturers (Neurology Drugs & CNS Biologics)
- Biotechnology Companies (Neurodegenerative & Rare Neurological Therapies)
- Contract Research Organizations (CROs – CNS Clinical Trials)
- Contract Manufacturing Organizations (CMOs – Complex Biologics & Injectables)
- Specialty Pharmacy Providers (Neurology Drug Distribution)
End Users
- Hospitals and Neurology Departments
- Specialty Neurology Clinics and Movement Disorder Centers
- Physicians (Neurologists & Neurospecialists)
- Retail and Hospital Pharmacists
- Patients undergoing chronic neurological therapies
Government and International Databases
- World Health Organization (WHO) – Global burden of neurological disorders
- U.S. Food and Drug Administration (FDA) – Drug approvals, clinical guidelines, and safety updates for CNS drugs
- European Medicines Agency (EMA) – Neurology drug approvals and pharmacovigilance data
- Centers for Disease Control and Prevention (CDC) – Prevalence of neurological disorders
- National Institutes of Health (NIH) – Clinical trials and neurological research data
Trade Publications
- Neurology Today
- Fierce Pharma
- Pharmaceutical Technology
- BioPharma Dive
Academic Journals
- The Lancet Neurology
- JAMA Neurology
- Neurology (American Academy of Neurology Journal)
- Brain: A Journal of Neurology
- Nature Reviews Neurology
Reputable Newspapers
- The Wall Street Journal – Pharmaceutical industry trends, M&A activity, and drug commercialization insights
- Financial Times – Global healthcare investments, regulatory landscape, and pharma strategy
- The New York Times – Healthcare policy, drug pricing, and patient access trends
- The Guardian – Public health developments and regulatory discussions
- The Economic Times – Pharmaceutical market trends and regional industry developments
Industry Associations
- American Academy of Neurology (AAN)
- European Academy of Neurology (EAN)
- Alzheimer’s Association
- Parkinson’s Foundation
- Multiple Sclerosis International Federation (MSIF)
Public Domain Resources
- ClinicalTrials.gov – Ongoing neurology clinical trials
- WHO Neurological Disorders Atlas
- FDA Orange Book & Drug Databases
- EMA Public Assessment Reports (EPAR)
Proprietary Elements
- CMI Data Analytics Tool: Proprietary analytics tool to analyze real-time market trends, consumer behavior, and technology adoption in market
- Proprietary CMI Existing Repository of Information for Last 10 Years
Share
Share
About Author
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients