Global Polygenic Risk Score Testing Market Size and Forecast – 2026-2033
The global polygenic risk score testing market is expected to grow from USD 1,608.2 Mn in 2026 to USD 3,980.9 Mn by 2033, registering a compound annual growth rate (CAGR) of 16.1%. The global polygenic risk score testing market is poised for significant expansion, driven by the rising prevalence of chronic diseases such as cardiovascular disorders, cancer, and diabetes.
The World Health Organization reports that non-communicable diseases (NCDs) account for 75% of global mortality, which translates to approximately 43 million deaths each year. This statistic highlights the critical need for advanced predictive tools, such as polygenic risk score (PRS), to help with early disease detection and the development of personalized treatments.
(Source: World Health Organization)
Key Takeaways of the Global Polygenic Risk Score Testing Market
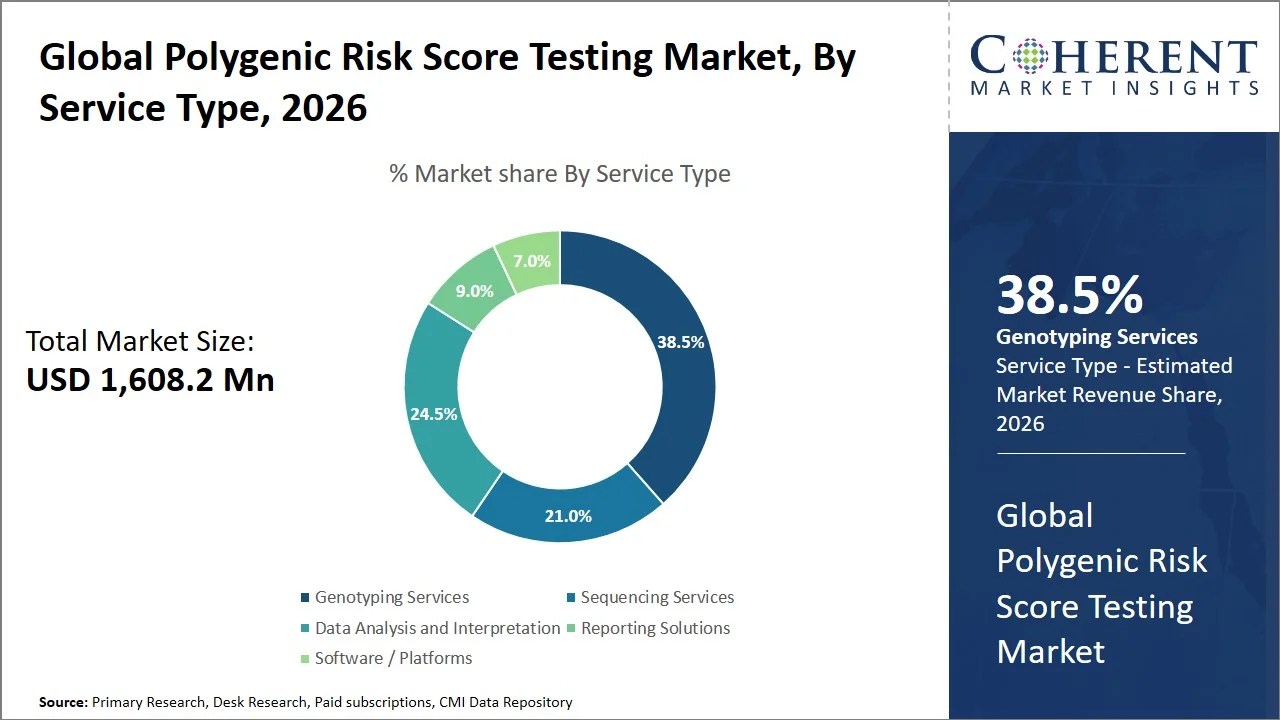
- Genotyping Services are projected to hold 38.5% of the global polygenic risk score testing market share in 2026, making it the dominant service type segment, especially in North America owing to the availability of large genomics projects like the All of Us Research Program, from which genomic information has been derived from over 750,000 individuals for affordable and efficient Single Nucleotide Polymorphism (SNP) analysis for polygenic risk score creation.
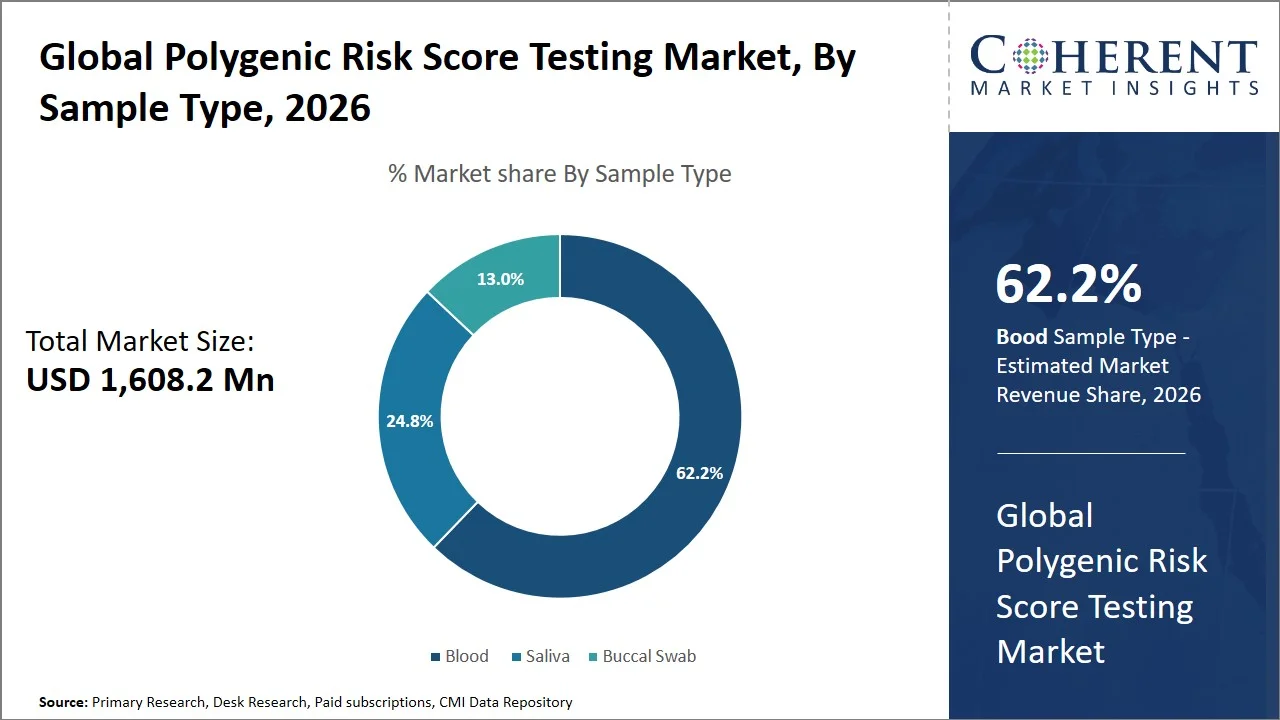
- Blood is projected to hold 62.2% of the global polygenic risk score testing market share in 2026, making it a dominant sample type segment, with a dominant presence in Europe, backed by robust biobanking facilities and genomic research programs (such as the UK Biobank) that rely on blood samples given that blood-based samples offer a high-quality yield of DNA with >99% reliability in genomics testing and are commonly employed in both clinical and biobanks, facilitating large-scale genomic data creation.
- Cardiovascular diseases segment is projected to hold 34.5% of the global polygenic risk score testing market share in 2026, making it the dominant disease category segment, especially common in North America and Europe, owing to the heavy burden of disease with more than 19.8 million deaths recorded annually throughout the world, and the growing role of polygenic risk score in disease prevention. (Source: World Health Organization)
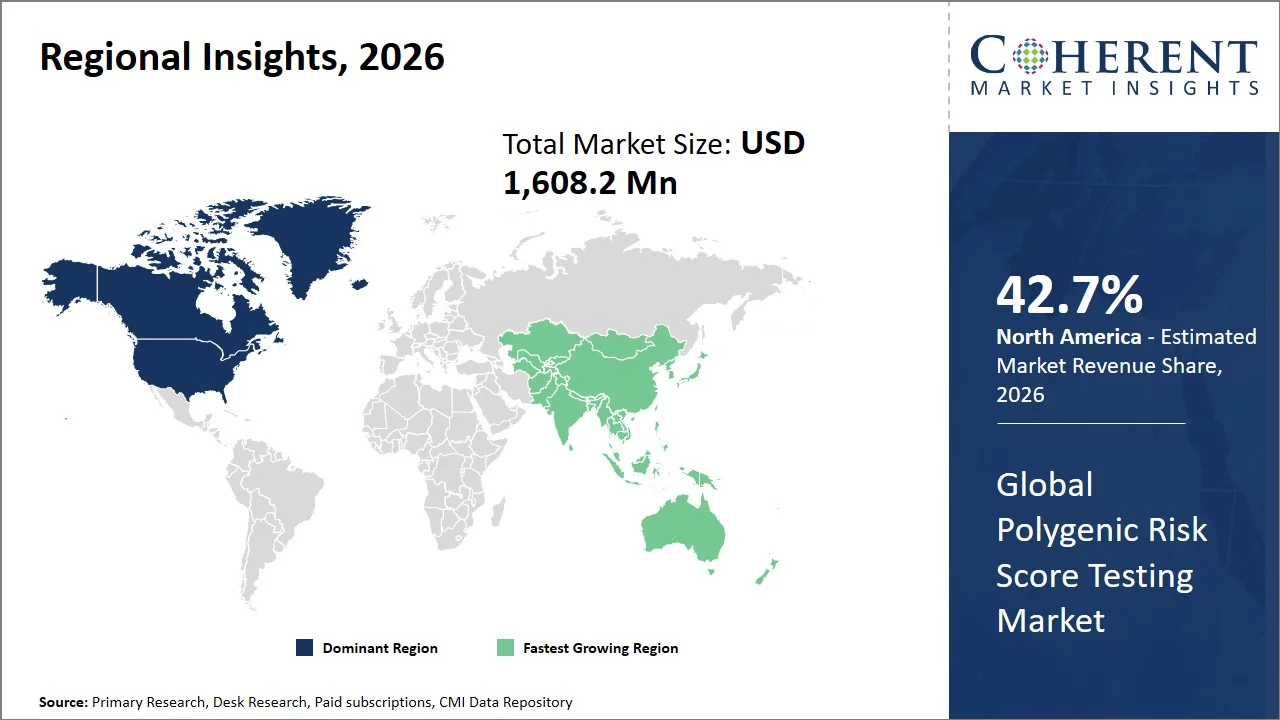
- North America maintains its dominance with an expected share of 42.7%in 2026, due to its sophisticated genomics research infrastructure. Government programs, which seek to obtain information from over a million individuals, contribute significantly to making scalable PRS testing possible. Moreover, high rates of chronic diseases among adults (6 in 10 adults in the U.S.) also stimulate interest in predictive genetic testing. (Source: CDC)
- Asia Pacific is expected to exhibit the fastest growth with an estimated contribution of 20.9% share in 2026, owing to the rapid expansion of genomics programs (such as the Genome India Project in India and the Precision Medicine Initiative in China), and increasing healthcare investments, indicating a shift in future growth toward emerging markets.
- Integration of Polygenic Risk Score (PRS) into Direct-to-Consumer (DTC) Genetic Testing Platforms: The rise of direct-to-consumer genetic testing represents an untapped niche that would be conducive to using PRS technology, allowing people to assess their personal risks of diseases without having to visit a clinic. The use of PRS in consumer genomics products by firms is making PRS technology more popular as it helps engage more customers.
- Increasing Use of Polygenic Risk Score (PRS) in Clinical Trial Patient Stratification: Polygenic risk score tools are being used by pharmaceutical and biotechnology firms in order to determine patient groups who pose higher risks during trials. This is an area-specific application that aids in boosting the trend of precision medicine.
Why Genotyping Services are Dominant in the Global Polygenic Risk Score Testing Market?
Genotyping services are projected to hold a market share of 38.5% in 2026, owing to their efficiency and economy in terms of the volume of tests carried out. Data provided by the U.S. government through the National Human Genome Research Institute reveal that even though whole genome sequencing has been expensive (costing millions of dollars) and complicated in terms of analysis, genotyping services make it possible to conduct high-throughput analyses in a short time and less expensively. The economic benefit of genotyping services, together with their efficiency, makes them the most favored method for conducting Polygenic Risk Score (PRS) testing. (Source: National Human Genome Research Institute)
Why is Blood the Most Preferred Sample Type?
To learn more about this report, Request Free Sample
Blood is projected to hold a market share of 62.2% in 2026, as it is the most sought-after specimen in diagnostics since it gives a holistic and up-to-date picture of physiological and biochemical processes occurring inside a patient’s body, thereby making blood ideal for disease diagnosis and prevention purposes. According to the U.S. National Library of Medicine, blood tests are one of the most popular laboratory tests performed, with blood being analyzed for cells, proteins, hormones, and biomarkers to detect diseases, evaluate organ functioning, and monitor progress in treatment. This is supported by the estimates associated with NIH, which suggests that around 500 million blood tests like CBC tests are performed each year in the U.S. (Source: National Library of Medicine)
Moreover, research published in National Library of Medicine revealed that clinical specialists shows that blood samples represent the most collected biologic specimens in laboratory diagnostics because they provide information on multiple diseases and conditions affecting a range of body systems. As blood travels throughout the body delivering oxygen and nutrients and removing metabolites, any abnormal changes may signal some problems, allowing detecting diseases at their early stages. (Source: National Library of Medicine)
Cardiovascular Diseases Segment Dominates the Global Polygenic Risk Score Testing Market
The cardiovascular diseases segment is projected to hold a market share of 34.5% in 2026, owing to the massive global disease burden for heart diseases and the clinical usefulness of Polygenic Risk Score (PRS) in predicting and preventing risks earlier. As per statistics from the World Health Organization, cardiovascular diseases are the main cause of death across the world, claiming an estimated 19.8 million lives every year, which translates into a third of all global deaths (Source: World Health Organization). These statistics have been crucial in encouraging governments and healthcare systems to prioritize early screening and preventative measures.
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
Macro Shift: Government-led Population Genomics Programs Driving Polygenic Risk Score (PRS) Adoption |
|
|
Expansion of NHS Genomic Medicine Service (U.K.) Driving Polygenic Risk Score (PRS) Integration |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
(Source: Department of Health & Social Care, NHS England)
Global Polygenic Risk Score Testing Market Dynamics
To learn more about this report, Request Free Sample
Market Drivers
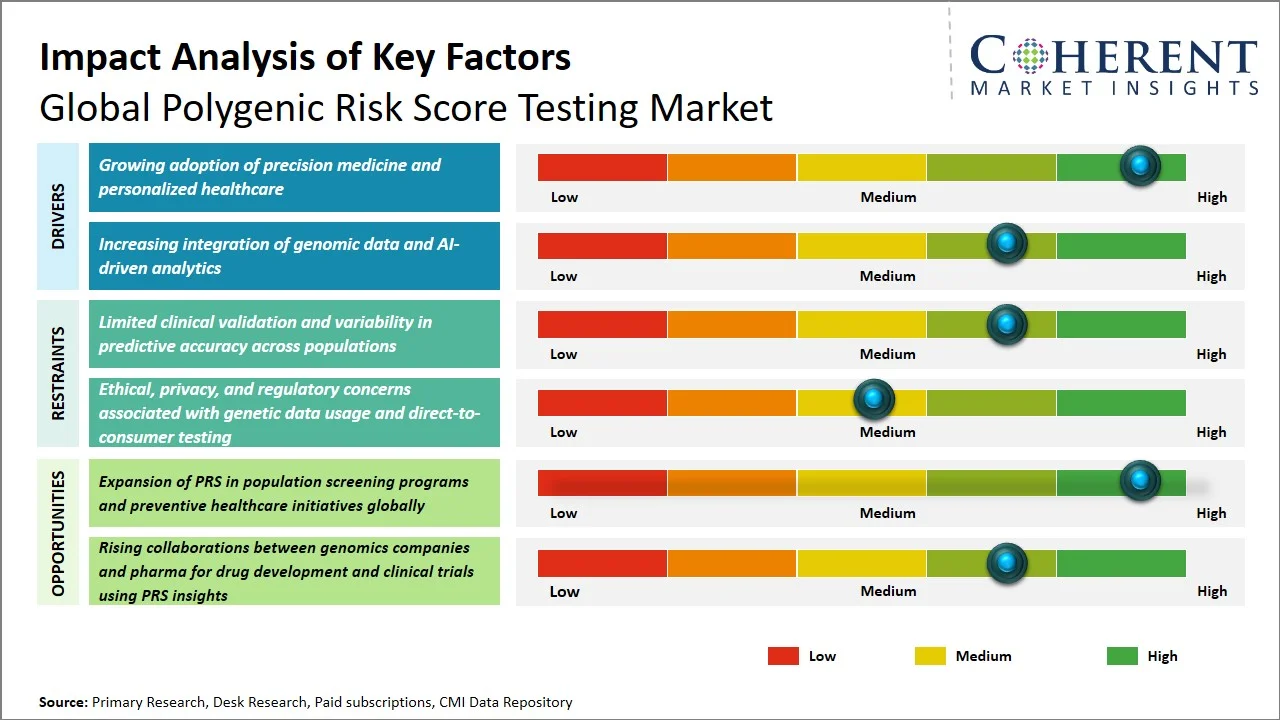
- Growing adoption of precision medicine and personalized healthcare: The polygenic risk score testing market is gaining momentum, largely due to the increasing adoption of precision medicine. Healthcare is evolving, moving beyond a purely reactive model to one that emphasizes prediction and personalization. For instance, the U.S. National Institutes of Health's All of Us Research Program seeks to gather health and genomic data from more than a million people living in the U.S. The goal is to enable tailored approaches to forecasting and managing illnesses (Source: National Institutes of Health). Correspondingly, the U.K.'s strategy in genomics reflects this trend, aiming to weave genomic medicine into the fabric of routine healthcare. Projects like the 100,000 Genomes Project are setting the stage for the broader adoption of precision diagnostics (Source: Department of Life Sciences, UK Government). This institutional support is, in turn, driving up the need for polygenic risk score testing, positioning it as a fundamental element in personalized risk assessment.
- Increasing integration of genomic data and AI-driven analytics: The combination of large genomic datasets and artificial intelligence is significantly improving the accuracy and practical use of polygenic risk score (PRS) testing, which, in turn, is creating considerable opportunities for progress. For instance, UK's Genome, U.K. initiative was allocated USD 34.9 million (£26 million) to combine genomic sequencing with AI and imaging technologies, all with the goal of improving how we diagnose and predict diseases (Source: Department of Health & Social Care). Moreover, national initiatives such as the NHS Genomic Medicine Service have already performed more than 810,000 genomic tests in 2024, generating vast datasets that are perfect for advanced analysis and risk modeling (Source: NHS England). The rapid expansion of data infrastructure, coupled with AI's integration, is enabling more precise polygenic risk score calculations, consequently accelerating its use in both clinical and research settings.
Emerging Trends
- Multi-ancestry Polygenic Risk Score (PRS) Models Gaining Traction: Traditional polygenic risk score (PRS) models have largely been built using datasets from European populations. This has limited their accuracy when applied to diverse populations. To improve predictive accuracy and encourage global use, especially in Asia Pacific and Africa, companies and researchers are now developing multi-ancestry PRS models that use larger genomic datasets.
- Integration of Polygenic Risk Score (PRS) into Clinical Decision Making: Polygenic risk score (PRS) is rapidly gaining ground as an essential component in clinical settings, particularly in the identification of patients who are at risk of developing heart disease, cancer, and diabetes. Polygenic risk score (PRS) technology, when used along with electronic health records (EHRs), has proven to be effective in preventive medicine and personalized therapy outside of experimental studies.
Regional Insights
To learn more about this report, Request Free Sample
Why is North America a Strong Market for Polygenic Risk Score Testing?
North America leads the global polygenic risk score testing market, accounting for an estimated 42.7% share in 2026, owing to the advanced infrastructure related to genomics, funding provided by the government, and initiatives involving precision medicine on a massive scale. Indeed, the U.S. government has been involved in financing research in this area via initiatives like the All of Us Research Program, which currently has more than 750,000 participants with genomic data that can be used for scientific studies, allowing for the development of polygenic risk score (PRS) models at a massive scale. (Source: National Institutes of Health)
Furthermore, the federal government's investment in precision medicine (e.g., the U.S. government allocated over USD 1.45 billion to the Precision Medicine Initiative, including USD 1.3 billion to the NIH’s All of Us Research Program), coupled with the incorporation of genomic data, is accelerating the adoption of predictive testing within the healthcare system. This robust framework, which includes financial resources, accessible data, and established research infrastructure, is positioning North America as a leader in the implementation and commercial application of polygenic risk score testing.
Why Does Asia Pacific Polygenic Risk Score Testing Market Exhibit High Growth?
Why are the U.S. Leading Innovation and Adoption in the Polygenic Risk Score Testing Market?
The U.S. is at the forefront of polygenic risk score (PRS) testing development and adoption owing to the country’s federal support for genomics, advanced research environment, and swift implementation of precision medicine in clinical practice. Federal programs such as the All of Us Research Program have been instrumental in developing huge databases of genomic information, where the program has so far been able to generate more than 750,000 full genome sequences, providing a solid foundation for polygenic risk score (PRS) models. Moreover, the U.S. government has earmarked roughly USD 1.45 billion to 1.5 billion for precision medicine projects, specifically those that integrate genomics into everyday clinical applications. (Source: National Library of Medicine)
Is China a Favorable Market for Polygenic Risk Score Testing Market?
China presents a rapidly expanding and potentially lucrative market for the polygenic risk score testing, driven by strong government involvement in genomics as well as the expansion of the precision medicine market. There has been increased focus on genomics in China due to initiatives like the China Precision Medicine Initiative which seeks to incorporate genetics into disease prevention and treatment programs. (Source: UC Institute on Global Conflict and Cooperation)
Furthermore, the National Bureau of Statistics of China reported that in 2023, the nation was home to more than 216 million individuals aged 65 and older. This demographic accounted for 21.1% of the total population. This increase in population along with the expansion of genomic information bases and digital health care is driving the use of tools like PRS. (Source: The State Council, The People’s Republic of China)
Is U.K. Emerging as a Key Growth Hub for the Polygenic Risk Score Testing Market?
U.K. has become an emerging market for polygenic risk score testing, owing to several government-led genomic projects and population-wide health initiatives in place. In line with the U.K. government’s Genome UK initiative, projects such as Our Future Health target to enroll five million individuals, with the program actively adopting PRS for delivering personalized disease risk information to the population (Source: Department of Health & Social Care). Furthermore, the U.K. has made substantial advancements in building genomic capacity via the implementation of projects like the 100,000 Genomes Program and NHS adoption, allowing for practical applications of predictive genomics.
Why Does Germany Top the European Polygenic Risk Score Testing Market?
Is Polygenic Risk Score Testing Market Developing in India?
The polygenic risk score testing market is experiencing a surge in India, fueled by genomics initiatives, supported by the government, and increasing investments in precision medicine (with the Department of Biotechnology allocating over USD 107 million (₹1,000 crore) under the Genome India Project to advance genomic research and personalized healthcare) Spearheaded by the Government of India, the Department of Biotechnology has launched the "Genome India Project." This project has sequenced over 10,000 genomes and gathered more than 20,000 biospecimens from 83 distinct populations, thereby establishing a nationwide database (Source: Department of Biotechnology). Additionally, the Indian Genomic Data Set (2025) is set to provide streamlined access to vast genomic data. This data is essential for disease prediction and the implementation of personalized treatment strategies. (Source: Ministry of Health and Family Welfare)
Regulatory Landscape Governing the Global Polygenic Risk Score Testing Market
|
Region |
Key Regulations |
Regulatory Overview |
Impact on Market |
|
North America |
U.S. Food and Drug Administration (FDA) – In Vitro Diagnostics (IVDs), Laboratory Developed Tests (LDTs); CLIA certification |
PRS tests that make clinical claims must meet analytical and clinical validity requirements similar to other diagnostic tests, though many remain under LDT pathways with evolving oversight |
Strong regulatory scrutiny ensures reliability and trust, supporting clinical adoption; however, evolving classification of PRS creates uncertainty and delays commercialization |
|
Europe |
In Vitro Diagnostic Regulation (IVDR); General Data Protection Regulation (GDPR) |
Highly stringent framework requiring clinical evidence, performance evaluation, and strict genomic data protection compliance; increasing push toward standardized PRS validation |
Enhances credibility and cross-border adoption but increases cost, certification timelines, and compliance burden for new entrants |
|
Asia Pacific |
National Medical Products Administration (China), PMDA (Japan), regional genomics policies |
Regulatory landscape is fragmented and evolving, with growing oversight on genetic testing and increasing government-led genomics initiatives |
Creates entry complexity due to variability; however, rising regulatory maturity and national precision medicine programs drive strong long-term growth |
|
Latin America |
ANVISA (Brazil), COFEPRIS (Mexico) |
Developing regulatory frameworks with limited PRS-specific guidelines and general genetic testing oversight |
Lower regulatory barriers enable early adoption and pilot testing, but lack of standardization limits large-scale clinical integration |
|
Middle East & Africa |
Saudi FDA, South African Health Products Regulatory Authority (SAHPRA) |
Nascent but improving frameworks focused on genomic medicine expansion and healthcare digitization |
Early-stage regulation supports innovation and partnerships, but lack of clear PRS-specific pathways slows widespread adoption |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
How is the expansion of polygenic risk score (PRS) in population screening programs and preventive healthcare initiatives globally creating new growth opportunities in the polygenic risk score testing market?
The increasing popularity of polygenic risk score (PRS) testing for population screening and prevention medicine is opening up substantial growth opportunities in terms of identifying individuals at high risk in large populations Governments are actively integrating polygenic risk score (PRS) into national genomic initiatives—for instance, the UK government’s Genome UK strategy supports the “Our Future Health” program, which aims to recruit up to 5 million participants and calculate PRS to provide personalized disease risk insights and support preventive care pathways (Source: Department of Health & Social Care).
Moreover, the National Health Service is advancing plans for genomic screening across the board. They're also testing how well polygenic risk scores (PRS) work, when they are added to regular health check-ups, and assessments of cardiovascular risk with research suggesting that using polygenic risk scores (PRS) can actually change risk levels in a significant way for roughly a quarter of people, which can then affect how they are treated (Source: Department of Health & Social Care). These government-supported projects are speeding up the process of proving polygenic risk scores (PRS) works and getting it used more widely. As a result, its use is expanding in early diagnosis, identifying people at risk, and preventive healthcare, which will eventually help the market growth.
Market Players, Key Development, and Competitive Intelligence

To learn more about this report, Request Free Sample
Key Developments
- On April 7, 2026, Allelica entered into a partnership with Mass General Brigham. The goal is to push forward research into polygenic risk scores (PRS) for cardiovascular disease. The aim is to refine early risk prediction and preventive care approaches. This collaboration will center on validating PRS within clinical environments, ultimately paving the way for wider use of genomic risk assessment tools.
- In November 2025, MyOme introduced its integrated polygenic risk score (PRS) test, specifically designed for prostate cancer. The goal is to improve early risk assessment and help customize screening strategies. This test combines genetic data with clinical information, hoping to enhance predictive accuracy and highlight the growing significance of PRS in precision medicine.
- In February 2025, Seonix Bio unveiled SightScore, a groundbreaking clinical polygenic risk score (PRS) test for glaucoma, marking its debut in the U.S. to facilitate early risk assessment and encourage proactive management of the disease. This test also supports personalized screening approaches, expanding the use of PRS in clinical diagnostics.
Competitive Landscape
The polygenic risk score testing market is highly competitive, fueled by swift progress in genomics, the rising embrace of precision medicine, and the expanding use of AI-powered analytics within healthcare. Established companies, with their robust expertise in genomic data processing, bioinformatics, and extensive population datasets, are broadening their reach. They are doing so while also grappling with the hurdles of regulatory approval and clinical validation. Key focus areas include:
- Advancement in genotyping and sequencing technologies
- Integration of AI and machine learning algorithms
- Expansion of biobank databases and population-scale genomic datasets
- Strategic collaborations with healthcare providers, pharma companies, and research institutions
Market Report Scope
Polygenic Risk Score Testing Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1,608.2 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 16.1% | 2033 Value Projection: | USD 3,980. 9 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
23andMe, Myriad Genetics, Color Genomics, Quest Diagnostics, Illumina, Invitae Corporation, F. Hoffmann-La Roche, Thermo Fisher Scientific, Eurofins Scientific, and BGI Genomics |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Analyst Opinion (Expert Opinion)
- In terms of the future of the market will witness a transition from the current state of being a highly scientific tool to the widespread use of the tool as a diagnostic application, enabled by innovations in artificial intelligence-based genomic tools and the increasing proof of the predictive power of polygenic risk score in detecting chronic illnesses, is expected to occur. In connection with the trend toward the preventive nature of health care and the personalization thereof, the use of polygenic risk score testing will become widespread in developed countries, such as the U.S., the U.K., and some EU member states.
- Potential for growth will be most significant in the fields of cardiology and oncology in North America, owing to factors such as high prevalence rates, access to big genomic databases (such as UK Biobank studies), and well-developed systems for reimbursement. In addition, there is potential for growth in the area of direct-to-consumer genetic tests in the U.S. as well as population-wide screenings offered by countries such as the U.K. Other emerging areas include those in Asia Pacific, notably China and Japan.
- For a competitive edge against other firms, there is a need for a focus on developing ethnic and evidence-based polygenic risk score models, which continue to be among the most defining features in terms of precision for varied populations. Collaborations with medical organizations, biobanks, and pharmaceutical firms would also be crucial when it comes to accessing genomics data of high quality and finding new uses.
Market Segmentation
- Type Insights (Revenue, USD Mn, 2021 - 2033)
- Genotyping Services
- Sequencing Services
- Data Analysis and Interpretation
- Reporting Solutions
- Software / Platforms
- Sample Type Insights (Revenue, USD Mn, 2021 - 2033)
- Blood
- Saliva
- Buccal Swab
- Disease Category Insights (Revenue, USD Mn, 2021 - 2033)
- Cardiovascular diseases
- Cancer
- Genetic diseases
- Neurodegenerative disorders
- Others
- Application Insights (Revenue, USD Mn, 2021 - 2033)
- Disease Risk Assessment
- Personalized Medicine
- Research
- End User Insights (Revenue, USD Mn, 2021 - 2033)
- Hospitals and Clinics
- Pharmaceutical and Biotech Companies
- Research Institutes
- Others
- Regional Insights (Revenue, USD Mn, 2021 - 2033)
- North America
- U.S.
- Canada
- Latin America
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Europe
- Germany
- U.K.
- Spain
- France
- Italy
- Russia
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Australia
- South Korea
- ASEAN
- Rest of Asia Pacific
- Middle East
- GCC Countries
- Israel
- Rest of Middle East
- Africa
- South Africa
- North Africa
- Central Africa
- North America
- Key Players Insights
- 23andMe
- Myriad Genetics
- Color Genomics
- Quest Diagnostics
- Illumina
- Invitae Corporation
- Hoffmann-La Roche
- Thermo Fisher Scientific
- Eurofins Scientific
- BGI Genomics
Sources
Primary Research Interviews
- Polygenic Risk Score testing companies
- Clinical genomics labs and service providers
Stakeholders
- Illumina (PRS software & arrays), Thermo Fisher Scientific (genotyping platforms), F. Hoffmann-La Roche (diagnostics & genomics), BGI Genomics
- End-use Sectors
- Hospitals & diagnostic labs (e.g., PRS-based disease risk testing)
- Biopharma companies integrating PRS in trials (e.g., Genomics plc collaboration with GSK)
- Research institutes using PRS catalogs and genomic datasets
Databases
- Data World Bank Healthcare Expenditure Database
- PGS Catalog – global repository of published PRS models and metadata
- Genome-wide association study (GWAS) datasets used for PRS development
- Clinical genomics datasets used by companies like Allelica for multi-ancestry risk modeling
Magazines
- GenomeWeb – coverage on PRS commercialization and genomics companies
- BioPharma Dive – adoption of PRS in drug development and trials
- Genetic Engineering & Biotechnology News – PRS innovations and precision medicine trends
Journals
- Nature Medicine – PRS clinical validation studies
- BMJ Medicine – evaluation of PRS performance in screening and prediction
- American Journal of Human Genetics – PRS methodologies and applications
- Genetics in Medicine – clinical utility of PRS testing
Newspapers
- The Wall Street Journal – genomics investments and precision medicine
- Financial Times – global genomics and biotech market trends
Associations
- International Common Disease Alliance – PRS standardization initiatives
- Global Alliance for Genomics and Health
- American Society of Human Genetics
Public Domain Sourcesa
- National Institutes of Health – genomics & precision medicine programs
- U.S. Food and Drug Administration – genetic testing regulatory framework
- European Commission – genomics and health data policies
Proprietary Elements
- CMI Data Analytics Tool, Proprietary CMI Existing Repository of information for last 10 years.
Share
Share
About Author
Nikhilesh Ravindra Patel is a Senior Consultant with over 8 years of consulting experience. He excels in market estimations, market insights, and identifying trends and opportunities. His deep understanding of the market dynamics and ability to pinpoint growth areas make him an invaluable asset in guiding clients toward informed business decisions. He plays a instrumental role in providing market intelligence, business intelligence, and competitive intelligence services through the reports.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients