U.S. and Europe Autoinjectors Market Size and Forecast – 2026 To 2033
The U.S. and Europe autoinjectors market is expected to grow from USD 4,421.7 Mn in 2026 to USD 12,706.6 Mn by 2033, registering a compound annual growth rate (CAGR) of 16.3%. The U.S. and Europe autoinjectors market is poised for significant expansion, fueled by increasing occurrence of chronic illnesses that necessitate self-medication.
According to the Centers for Disease Control and Prevention (CDC), about 38 million U.S. adults have diabetes, and 1 in 5 of them don't know it. This statistic underscores the significant need for easy-to-use drug delivery systems, such as autoinjectors.
(Source: Centers for Disease Control and Prevention (CDC))
Key Takeaways of the U.S. and Europe Autoinjectors Market
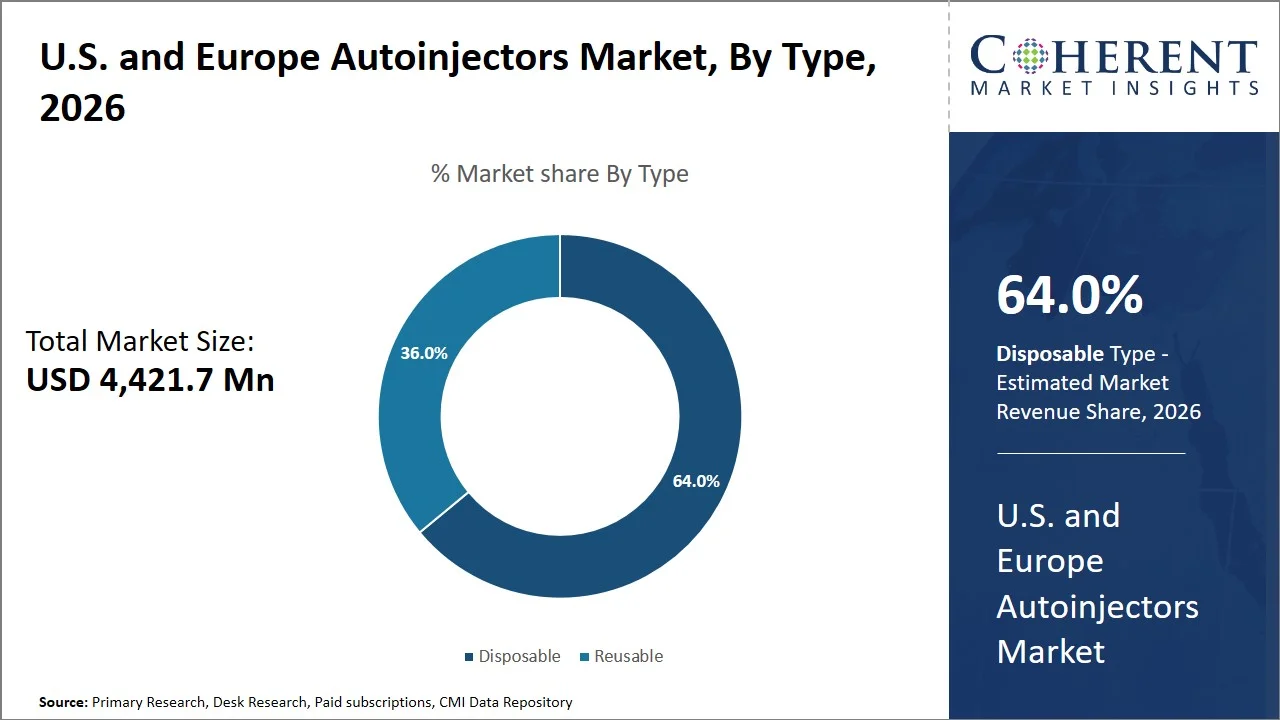
- Disposable is projected to hold 64.0% of the U.S. and Europe autoinjectors market share in 2026, making it the dominant type segment. This dominance is driven by the increasing preference for single-use, pre-filled devices across the U.S. and key European markets such as the U.K. and Germany, particularly for both chronic and emergency treatments. For instance, disposable epinephrine autoinjectors like EpiPen by Viatris are widely used for emergency allergy management, highlighting their ease of use, reliability, and reduced contamination risk, which collectively support segment growth.
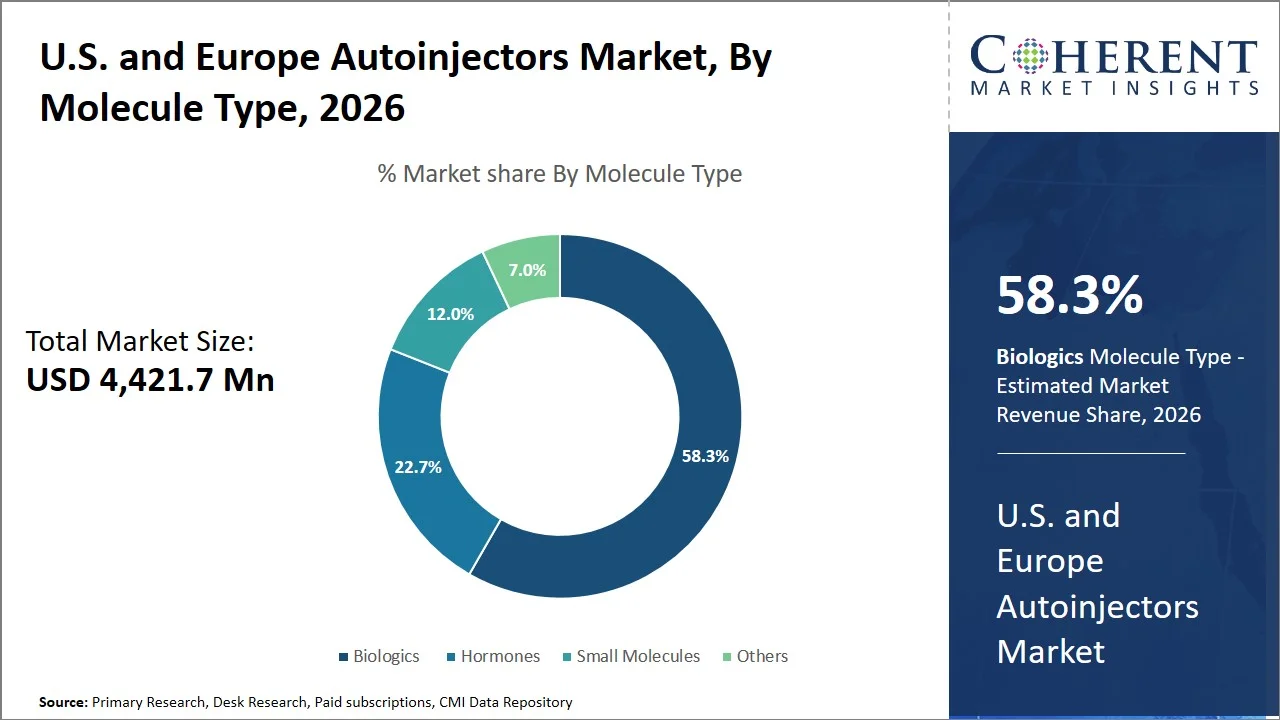
- Biologics are projected to hold 58.3% of the U.S. and Europe autoinjectors market share in 2026, making it the dominant molecule type segment. The dominance of the segment is witnessed across the U.S. and Europe, with high penetration in terms of the occurrence of autoimmune diseases and monoclonal antibody treatments. Factors that would fuel growth for the demand for autoinjectors include the precision needed for subcutaneous delivery, along with the biologics pipeline. For example, widely used biologics like Enbrel by Amgen are delivered via autoinjectors for conditions such as rheumatoid arthritis, supporting demand due to the need for precise subcutaneous administration and a strong biologics pipeline.
- Anaphylaxis segment is projected to hold 22.0% of the U.S. and Europe autoinjectors market share in 2026, making it the dominant indication segment owing to the high number of people with severe allergies (affecting up to ~10% of the population in the U.S.) and the presence of strict clinical practices in terms of emergency readiness (including guidelines from the National Institute for Health and Care Excellence and the American Academy of Allergy, Asthma & Immunology recommending immediate access to epinephrine autoinjectors).
- Rising Demand for Combination Drug-Device Products: There is an increasing approval of biologics together with specialized delivery devices that have been creating a demand for personalized autoinjectors. In doing so, drug manufacturers can benefit from better product differentiation and increased compliance among patients.
- Focus on Patient-Centric & Ergonomic Device Design: With a growing focus on user experience, particularly for older adults and children, manufacturers are incorporating improvements such as reduced injection force, one-button activation, ergonomic grip design, audible/visual feedback indicators, hidden needles, and shorter injection times. These enhancements improve ease of use, minimize administration errors, and support better patient adherence in self-administration settings.
Why Does the Disposable Segment Dominate the U.S. and Europe Autoinjectors Market?
The disposable segment is projected to hold a market share of 64.0% in 2026, owing to its convenience, low risk of contamination, and usage in both emergency and chronic conditions that require accurate dosage. Disposables are prefilled and intended for one-time use only; hence, they eliminate cartridge handling or maintenance. For example, the U.S. FDA-approved EpiPen (epinephrine autoinjector) is a disposable, single-dose device used for treating emergency conditions such as anaphylaxis. (Source: Food and Drug Administration) Moreover, the trend of home care has increased in recent years in the U.S. and Europe with over 3.3 million patients receiving home healthcare services in the U.S. and nearly 28.3% of individuals requiring long-term care in Europe opting for professional homecare, reflecting a strong shift toward decentralized treatment models. (Source: Centers for Disease Control and Prevention, Eurostat) Hence, the demand for disposable autoinjectors will continue to increase since they are easy to use and provide consistent delivery of medicines.
Why are Biologics the Most Preferred Molecule Type in the U.S. and Europe Autoinjectors Market?
To learn more about this report, Request Free Sample
Biologics are projected to hold 58.3% of the market share in 2026, owing to their high effectiveness in dealing with chronic and autoimmune diseases as well as the ability to be administered via the subcutaneous route. These treatments, that include monoclonal antibodies are commonly prescribed for diseases like rheumatoid arthritis and multiple sclerosis and, thus, autoinjectors are the best form for delivering those medications. This market leadership is further boosted by a large number of activities on the regulatory side, with the European Medicines Agency giving recommendation for 77 medicines to be authorized in 2023, among which 39 contain new active substances. (Source: European Medicines Agency)
Anaphylaxis Segment Dominates the U.S. and Europe Autoinjectors Market
Anaphylaxis segment is expected to hold 22.0% of the U.S. and Europe autoinjectors market share in 2026, owing to the requirement for instant life-saving intervention in the case of severe allergic responses. Auto-injectors are the preferred treatment for delivering epinephrine during emergencies. The occurrence of severe allergies is growing, and therefore, there is a high demand for convenient equipment for administering the drug. An example includes the epinephrine auto-injectors that have received approval from the U.S. Food and Drug Administration for the emergency treatment of anaphylaxis. (Source: U.S. Food and Drug Administration) Moreover, strong clinical guidelines recommending that individuals at risk of severe reactions consistently carry such medications further enhance the segment’s market prominence.
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
EU Proposal to Simplify Medical Device Regulations (December 2025) |
|
|
EU Pharmaceutical Legislation Reform to Improve Access & Innovation (2025–2026) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
(Source: European Commission, European Commission)
U.S. and Europe Autoinjectors Market Dynamics
To learn more about this report, Request Free Sample
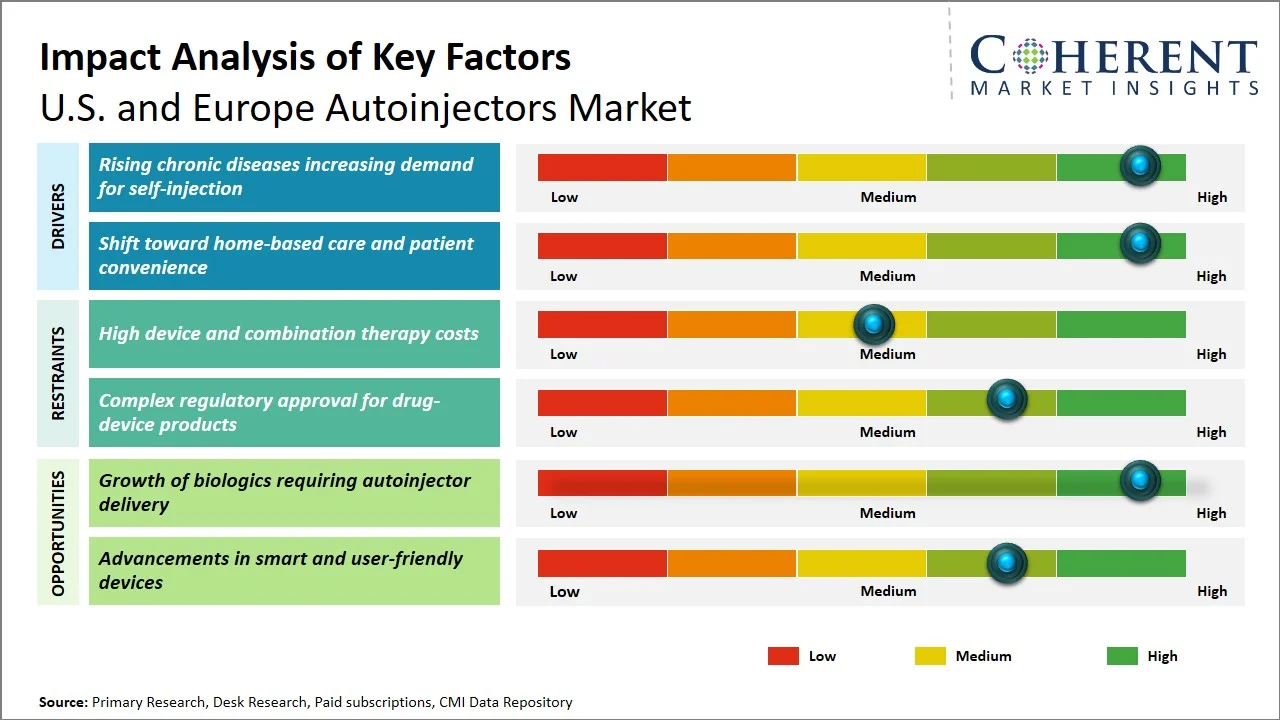
Market Drivers
- Rising chronic diseases increasing demand for self-injection: The rising burden of chronic illness form a major factor boosting the usage of autoinjector as they necessitate regular medication intake by patients. According to Centers for Disease Control and Prevention, an estimated 129 million people in the U.S. have at least 1 major chronic disease (e.g., heart disease, cancer, diabetes, obesity, hypertension). (Source: Centers for Disease Control and Prevention) This trend has created an increased demand for convenient methods for administering drugs from the comfort of one’s own home, thus promoting the use of autoinjectors in the U.S. and Europe.
- Shift toward home-based care and patient convenience: The move towards home care and increased convenience for patients are greatly contributing to the use of autoinjectors, especially since the health care system in the U.S. and Europe has been focusing on minimizing hospital visits and promoting self-care among chronic disease patients. The use of self-care medications that can be used within the comfort of one's own house saves time and does not require constant interaction with health care providers. In addition, there is a significant cost pressure on the health care system, and there are calls for increased efficiency in health care. For example, self-administered biologics such as Humira by AbbVie allow patients to treat chronic conditions at home, reducing hospital visits.
Emerging Trends
- Integration of Smart & Connected Autoinjectors: Increasing numbers of autoinjectors are integrating Bluetooth and applications on cell phones to monitor dosage adherence. This approach aids the process of real-time monitoring of the patient and corresponds to the overall trend towards digitalization of healthcare.
- Growth of Biosimilars Driving Device Innovation: The development of biosimilar products markets drives the increasing need for affordable and unique autoinjectors. The key point is the development of personalized solutions aimed at improving patient experience and facilitating biologics differentiation.
Reimbursement as a Core Growth Lever in the U.S. and Europe Autoinjectors Market
Reimbursement is a critical component that influences the U.S. and Europe autoinjectors market, since autoinjectors are used mainly for chronic diseases and biological medicines, their affordability is affected greatly by payers like the Centers for Medicare & Medicaid Services and European health organizations. Positive reimbursement increases affordability, leading to increased adoption rates of autoinjectors since the patients will prefer easy-to-use autoinjectors for self-management.
Reimbursement Impact on U.S. and Europe Autoinjectors Market
|
Factor |
Impact on Market |
|
Coverage by Payers |
Expands patient access to autoinjectors for chronic and biologic therapies |
|
Out-of-Pocket Costs |
Lower costs increase patient willingness to adopt self-injection devices |
|
Chronic Disease Coverage |
Ensures continuous demand due to long-term treatment needs |
| Homecare Reimbursement Policies |
Encourages shift toward self-administration and reduces hospital visits |
|
Formulary Inclusion |
Drives physician prescribing behavior and product preference |
|
Specialty Drug Support |
Boosts uptake of biologics delivered via autoinjectors |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
How is the growth of biologics requiring autoinjector delivery creating new growth opportunities in the U.S. and Europe autoinjectors market?
The swift rise in the number of biologics presents lucrative prospects within the U.S. and European markets for autoinjectors because they necessitate a precise and convenient subcutaneous injection, and hence, autoinjectors provide an optimal way to deliver biologics. This development is further supported by favorable regulatory developments, as evidenced by the fact that the U.S. FDA has already approved more than 40 biosimilars, which shows the robust and promising pipeline of injectable biologics. (Source: U.S. Food and Drug Administration) With an increase in pharmaceuticals developing biologics for self-administration, the need for tailor-made, user-friendly autoinjectors will rise, which will encourage innovation and create opportunities for growth in the markets.
Market Players, Key Development, and Competitive Intelligence

To learn more about this report, Request Free Sample
Key Developments
- In December 2025, a positive CHMP opinion was obtained by Celltrion for its ustekinumab biosimilar, SteQeyma, which had been formulated as an autoinjector. This move highlights the growing trend of biosimilars that have been combined with devices to be administered by the patients themselves.
- In September 2025, Ypsomed AG secured U.S. FDA 510(k) clearance for SmartPilot. This digital connectivity add-on for the YpsoMate autoinjector allows for the immediate capture and transmission of injection data. This move underscores the increasing embrace of connected drug delivery systems. The goal is to boost patient adherence, facilitate monitoring, and ultimately, improve treatment results within the autoinjectors market.
- In June 2025, SHL Medical played a key role in the European debut of the first ustekinumab biosimilar autoinjector, utilizing its Molly platform. This launch represents a notable step forward in patient-focused drug delivery. It underscores the growing convergence of biosimilars and sophisticated autoinjector technologies, furthering the trend toward self-administration and broadening the use of platform-based delivery systems throughout Europe.
Competitive Landscape
The U.S. and Europe autoinjectors market is highly competitive with major pharmaceutical and medical device firms vying for dominance in hospitals, pharmacies, and patients' homes. To stand out, these companies are investing in product innovation, particularly in sophisticated drug-device pairings and smart autoinjector technologies. Patient-centered design, user-friendliness, and compatibility with digital health tools are becoming increasingly important. Key focus areas include:
- Advanced Biologic-Compatible & Combination Autoinjectors
- Strong Hospital, Retail & Homecare Distribution Networks
- Regulatory Compliance & Patient-Centric Device Innovation
Market Report Scope
U.S. and Europe Autoinjectors Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 4,421.7 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 16.3% | 2033 Value Projection: | USD 12,706.6 Mn |
| Segments covered: |
|
||
| Companies covered: |
SHL Medical AG, Ypsomed AG, BD, West Pharmaceutical Services, Inc., Gerresheimer AG, Owen Mumford Ltd., Recipharm AB, Phillips-Medisize, Amgen Inc., and Eli Lilly and Company |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Analyst Opinion (Expert Opinion)
- The future outlook will be influenced by the fast-growing use of biologics and self-injectables, especially in chronic diseases like diabetes, rheumatoid arthritis, multiple sclerosis, and anaphylaxis. There has been a shift towards a patient-centered approach to drug delivery, as well as growing need for easy-to-use and safer injection systems that offer connectivity. Moreover, there is likely to be incorporation of smart injectors and at-home treatment, which is expected to influence market growth in these geographies.
- Most opportunities can be found in the autoinjectors market in the U.S. and key EU countries such as Germany, France, and the U.K., considering that biologics utilization is high in these regions. In addition, there are many prospects in the development of biosimilars and volume-based biologics treatments due to the need for affordable and scalable delivery devices.
- In order to outpace competitors, it would be reasonable for companies to create distinctive products by developing unique designs, additional features such as safety systems, and digital technologies. Another important step would be to establish partnerships with major pharmaceutical companies to develop combined products. Furthermore, it would be essential to achieve greater scalability of production and regulatory compatibility in the U.S. and Europe to accelerate the product approval process.
Market Segmentation
- Type Insights (Revenue, USD Mn, 2021 - 2033)
- Disposable
- Reusable
- Molecule Type Insights (Revenue, USD Mn, 2021 - 2033)
- Biologics
- Hormones
- Small Molecules
- Others
- Indication Insights (Revenue, USD Mn, 2021 - 2033)
- Anaphylaxis
- Diabetes
- Autoimmune Diseases
- Multiple Sclerosis
- Migraine
- Hormonal Disorders
- Others
- End User Insights (Revenue, USD Mn, 2021 - 2033)
- Hospitals
- Home Care Settings
- Ambulatory Surgical Centers
- Others
- Key Players Insights
- SHL Medical AG
- Ypsomed AG
- BD
- West Pharmaceutical Services, Inc.
- Gerresheimer AG
- Owen Mumford Ltd.
- Recipharm AB
- Phillips-Medisize
- Amgen Inc.
- Eli Lilly and Company
Sources
Primary Research Interviews
- Biologic drug manufacturers using autoinjectors (e.g., insulin, epinephrine, biologics)
- Medical device engineers and autoinjector design specialists
- Hospital pharmacists and specialty drug procurement heads
- Regulatory affairs experts – combination products (U.S. & Europe)
Databases
- U.S. FDA – Drugs@FDA and Combination Products database (approvals of autoinjectors)
- European Medicines Agency (EMA) – EPAR database (biologics with autoinjector delivery)
- ClinicalTrials.gov – Trials involving self-injection and autoinjector devices
Magazines
- Pharmaceutical Technology – Injectable drug delivery and device innovations
- Medical Device Network – Drug-device combination product trends
- Contract Pharma – Biologics delivery and device manufacturing insights
- Drug Delivery Business News – Injectable and autoinjector device developments
Journals
- Journal of Controlled Release – Drug delivery systems and injectables
- International Journal of Pharmaceutics – Device-based drug delivery research
- Advanced Drug Delivery Reviews – Injectable and biologics delivery technologies
- PDA Journal of Pharmaceutical Science and Technology – Parenteral delivery systems
Newspapers
- The Wall Street Journal – Pharmaceutical and biologics market trends
- Financial Times – Global pharma and injectable drug market developments
Associations
- Parenteral Drug Association (PDA) – Injectable drug delivery standards
- Combination Products Coalition – Regulatory and industry alignment
- MedTech Europe – European medical device regulatory developments
- Advanced Medical Technology Association (AdvaMed) – U.S. device industry
Public Domain Sources
- U.S. FDA – Combination Products & Medical Devices regulations
- European Medicines Agency (EMA) – Drug-device combination guidance
- European Commission – Medical Device Regulation (MDR 2017/745)
- Health Canada – Combination products and medical device framework
Proprietary Elements
- CMI Data Analytics Tool, Proprietary CMI Existing Repository of information for last 10 years.
Share
Share
About Author
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients