The Iv infusion bottles seals and caps Market is anticipated to grow at a CAGR of 2.6% with a market value of about USD 124.3 Mn in 2026 and is expected to reach around USD 148.2 Mn in 2033. Steady growth is driven by increasing demand for safe and contamination-free drug delivery systems, rising hospital admissions and IV therapies, expansion of pharmaceutical manufacturing, and growing emphasis on regulatory compliance and packaging integrity in healthcare systems.
Innovations in bio-pharmaceuticals, oncological drugs, and specialized drug delivery technologies are changing the packaging demands in the pharmaceutical industry. These drugs require packaging which is very stable and contamination-proof, thus creating the need for rubber stoppers, aluminum closures, and polypropylene caps.
Advanced elastomer compounds and rubber closure coating technologies have increased compatibility with biological drugs while minimizing interactions and extending shelf life. Furthermore, advances in the process of sterile production technology are making possible the mass production of infusion packaging systems with higher safety standards.
The European Medicines Agency (EMA) emphasizes strict requirements for primary packaging materials used in biologics to ensure compatibility and patient safety, further driving innovation in closure systems.
Increase in the number of hospitalizations, surgeries, and emergency treatment cases on a worldwide scale will increase the need for intravenous therapy. The requirement for infusion bottles and sealing devices will be enhanced because they play a vital role in administering medications to patients.
The geriatric population is growing, and cases of chronic illnesses are rising, which further increases the utilization of intravenous medications. Hospitals are implementing standard infusion systems to increase efficiency, reduce contamination possibilities, and adhere to global health regulations.
As per the World Bank Health Statistics, healthcare utilization and hospital-based treatments have increased steadily over the last decade, particularly in developing regions, supporting higher demand for IV drug delivery systems.
|
Current Event |
Description and its Impact |
|
FDA Strengthening of Container Closure System Requirements (U.S.) |
|
|
EU Pharmaceutical Packaging Compliance Tightening under GMP and MDR Frameworks |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The rubber seals (elastomeric closures) are expected to capture the highest market share in the Iv infusion bottle seals and caps market by 2026, accounting for around 52% of the overall product type segment share. Their dominant position in the market is justified by the fact that rubber seals are necessary for the maintenance of sterility and container-closure integrity of injectables.
The extensive use of these packaging materials has been highly influenced by their biocompatibility, efficient barriers, and their ability to resist sterilization methods including autoclaving and gamma radiation. In addition, regulatory requirements from agencies such as the U.S. FDA and quality standards under USP <381> for elastomeric closures reinforce their necessity in sterile pharmaceutical packaging systems.
To learn more about this report, Request Free Sample
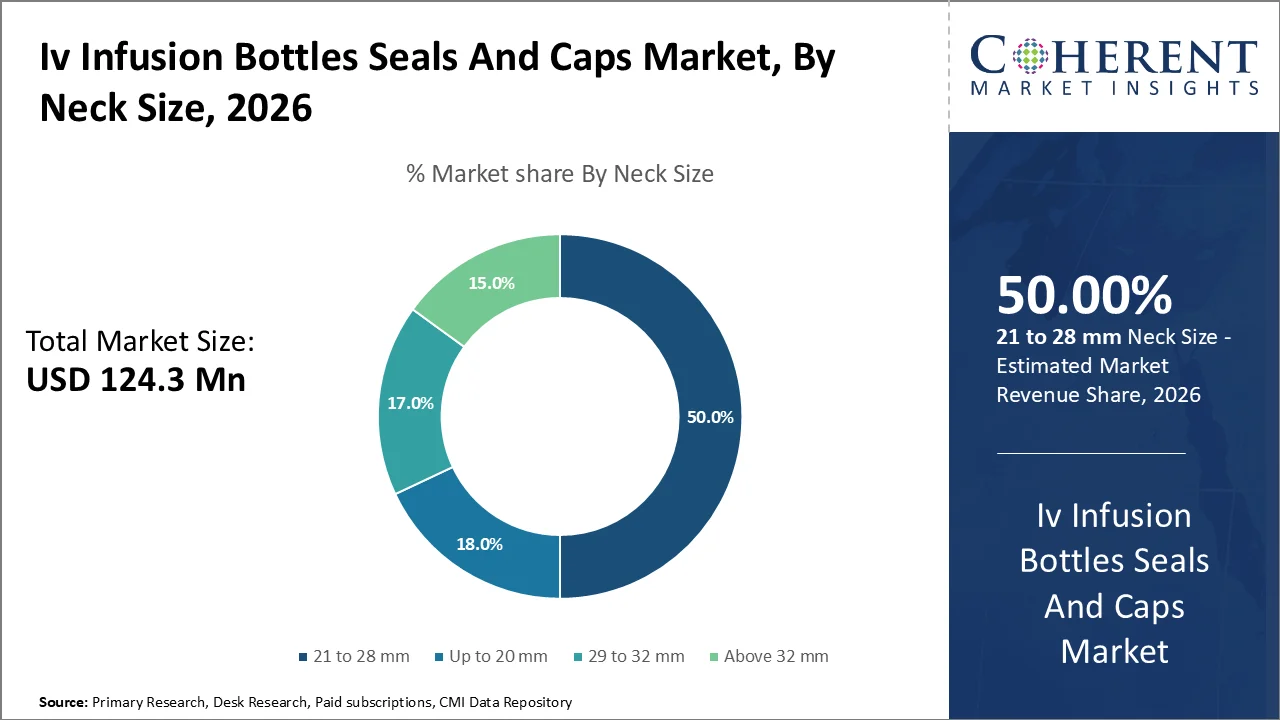
The neck sizes of 21-28 mm are expected to hold the highest market share of around 50% by 2026, due to its being the most standardized neck size for IV infusion bottles that are utilized in pharmaceutical industries across the world. The neck size of 21 to 28 mm is utilized a lot in antibiotics, cancer drugs, anesthetic, and nutritional fluids.
Another reason for the popularity of this neck size is the increasing trend toward intravenous therapy in hospital environments, and the need for universality in automated fill lines and aseptic processing plants. The pharmaceutical industry favors the use of this neck size owing to its excellent balance between stiffness, sealability, and manufacturability.
In March 2025, the International Organization for Standardization (ISO) released ISO 8536-6:2025, which defines updated technical requirements for closures used in infusion bottles, including dimensional, material, and performance specifications for sterile drug container closure systems.
To learn more about this report, Request Free Sample
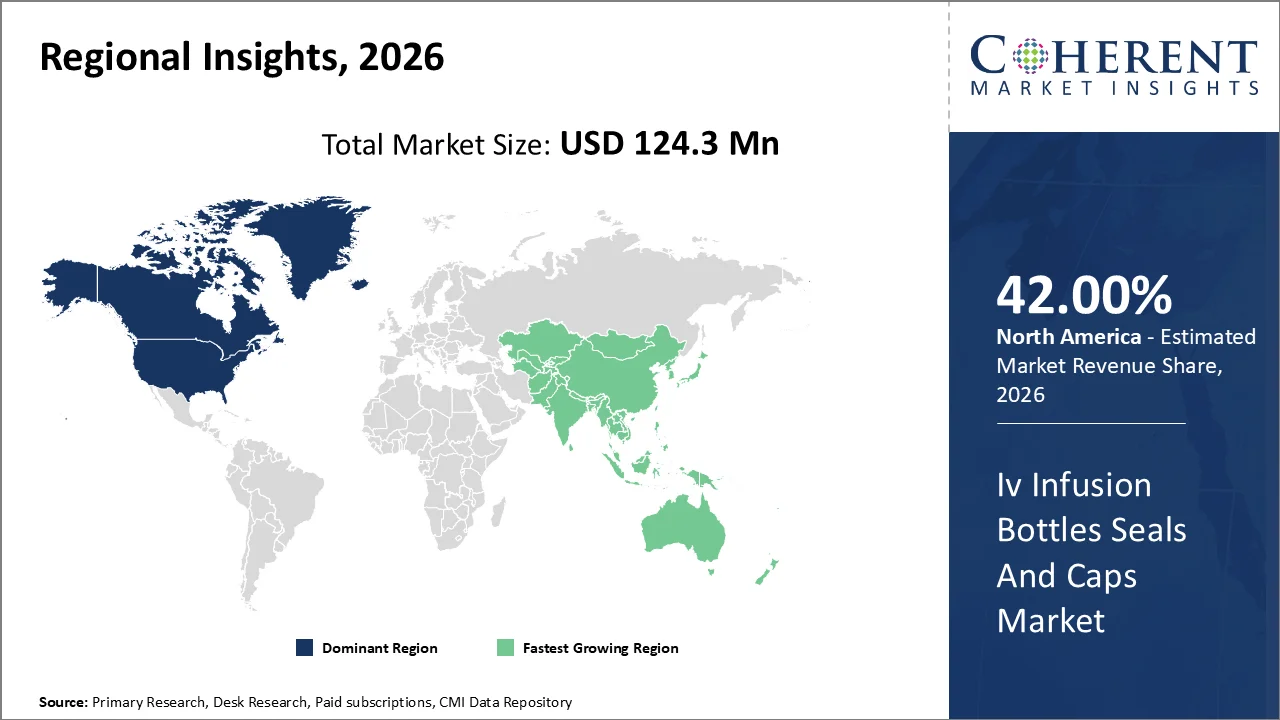
North America is expected to capture 42% market share in the global Iv infusion bottle seals and caps industry owing to the large consumption of sterile injectable drugs and biologics as well as advanced intravenous infusions at medical facilities and outpatient centers. Also, the region will gain a strong position in the market due to stringent regulations on container closure integrity, extractables & leachable, and parenteral aseptic filling requirements.
In March 2025, West Pharmaceutical Services (U.S.), a leading North American pharmaceutical packaging company, announced continued expansion of its advanced elastomeric closure technologies, including high-purity rubber stoppers designed for biologics and sensitive injectable therapies.
The Asia-Pacific region is anticipated to exhibit the highest growth rate during 2026-2033 due to the swift expansion of the pharmaceutical industry, the development of hospitals, and an increased need for injectable drugs in developing countries like China and India.
China and India are major drivers of regional economic growth because of their mass production of injectable drugs and hospitals. Investments made in pharmaceutical packaging automation and aseptic fill have improved production efficiencies while increasing demand for infusion bottle closures.
In February 2026, pharmaceutical packaging standardization initiatives across Asia Pacific reinforced the use of ISO-aligned infusion bottle systems, particularly 21–28 mm neck size containers, improving compatibility for IV drug manufacturing and aseptic filling operations.
The U.S. Iv infusion bottles seals and caps market is expanding rapidly owing to the high demand for sterile injections, biological products, and intravenous therapies. The USA ranks amongst the countries that provide a high number of injections to patients across the globe due to the advanced healthcare facilities in the country and strict CCI laws set by the FDA.
In January 2025, the U.S. FDA reinforced container closure system requirements for sterile injectable drugs, emphasizing improved testing for sterility assurance, extractables, and leachable in infusion packaging systems. This regulatory tightening is pushing manufacturers to adopt high-performance rubber seals and advanced closure technologies.
The China Iv infusion bottles seals and caps market is witnessing rapid expansion because of its significant pharmaceutical manufacturing capability and rising usage of injectables. China is one of the largest manufacturers of generic injectable drugs and IV solutions worldwide, and therefore it plays an important role in the packaging of IV solutions.
In addition, China’s increasing alignment with WHO GMP guidelines for sterile pharmaceutical manufacturing is accelerating the adoption of standardized infusion bottle closure systems, particularly in high-volume injectable drug production facilities.
Some of the major key players in Iv Infusion Bottle Seals and Caps are Cardinal Health Inc., Jiangsu changjiang Lids, Taiwan Hon Chuan Enterprise, PrasadMeditech, Winfield Laboratories Inc., Datwyler HoldingAG, Wuxi Qitian Medical Technology Co., Ltd, Medline Industries Inc., RENOLITSE, and B. Braun Medical Inc.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 124.3 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 2.6% | 2033 Value Projection: | USD 148.2 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Cardinal Health Inc., Jiangsu changjiang Lids, Taiwan Hon Chuan Enterprise, PrasadMeditech, Winfield Laboratories Inc., Datwyler HoldingAG, Wuxi Qitian Medical Technology Co., Ltd, Medline Industries Inc., RENOLITSE, and B. Braun Medical Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Market expansion is essentially driven by increasing demand, backed by growing global incidences of chronic diseases and an increasing reliance on IV procedures using hospital equipment.
Share
Share
Manisha Vibhute is a consultant with over 5 years of experience in market research and consulting. With a strong understanding of market dynamics, Manisha assists clients in developing effective market access strategies. She helps medical device companies navigate pricing, reimbursement, and regulatory pathways to ensure successful product launches.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients