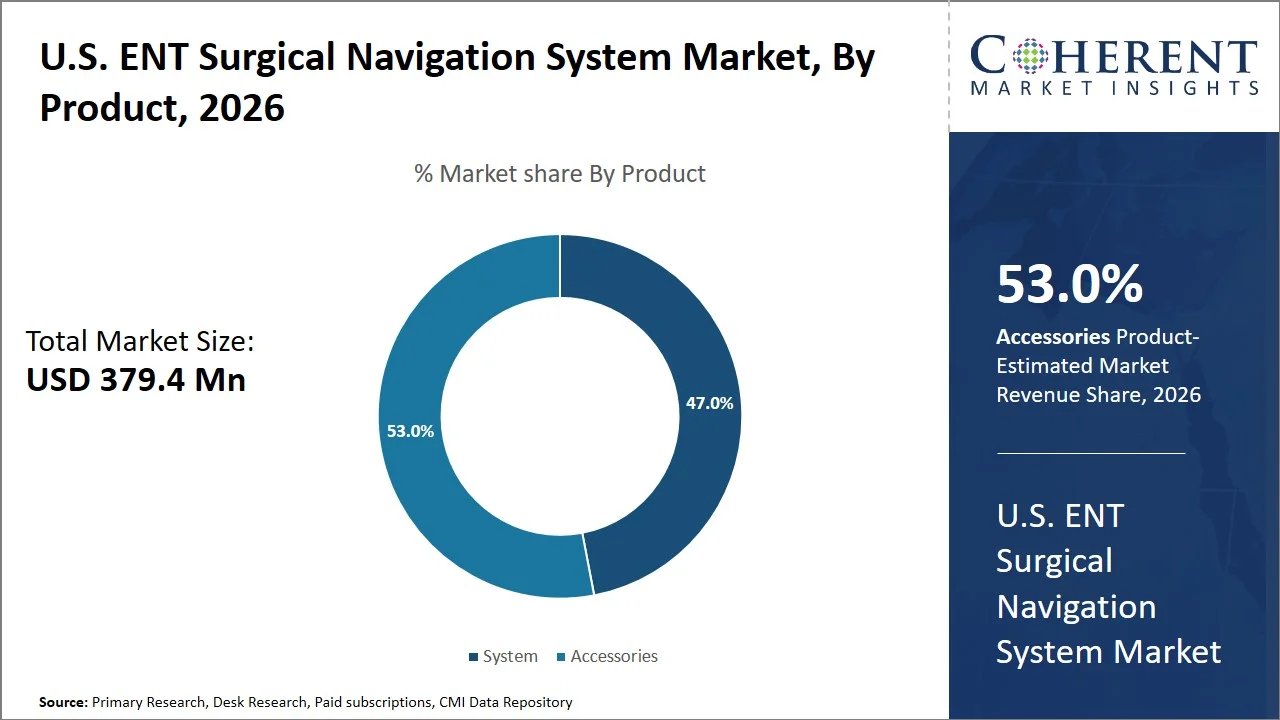
U.S. ENT Surgical Navigation System Market Size and Forecast – 2026-2033
The U.S. ENT surgical navigation system market is expected to grow from USD 379.4 Mn in 2026 to USD 617.2 Mn by 2033, registering a compound annual growth rate (CAGR) of 7.2%. The U.S. ENT surgical navigation system market is poised for significant expansion, fueled by the rising prevalence of ENT disorders, particularly chronic sinusitis.
According to the U.S. Centers for Disease Control and Prevention (CDC), approximately 28.9 Million adults in the U.S. are diagnosed with sinusitis, representing around 11.6% of the adult population.
(Source: Centers for Disease Control and Prevention)
Key Takeaways of the U.S. ENT Surgical Navigation System Market
- Accessories are projected to hold 53.0% of the U.S. ENT surgical navigation system market share in 2026, making it the dominant product segment, driven by the need for consumable items associated with procedures such as navigation trackers, reference frames, and instrument adaptors which have to be changed after each Functional Endoscopic Sinus Surgery (FESS) or skull base surgical procedure. For instance, Endoscopic Sinus Surgery (ESS) is one of the most common surgical procedures in the U.S., with over 250,000 cases performed annually. (Source: National Library of Medicine)
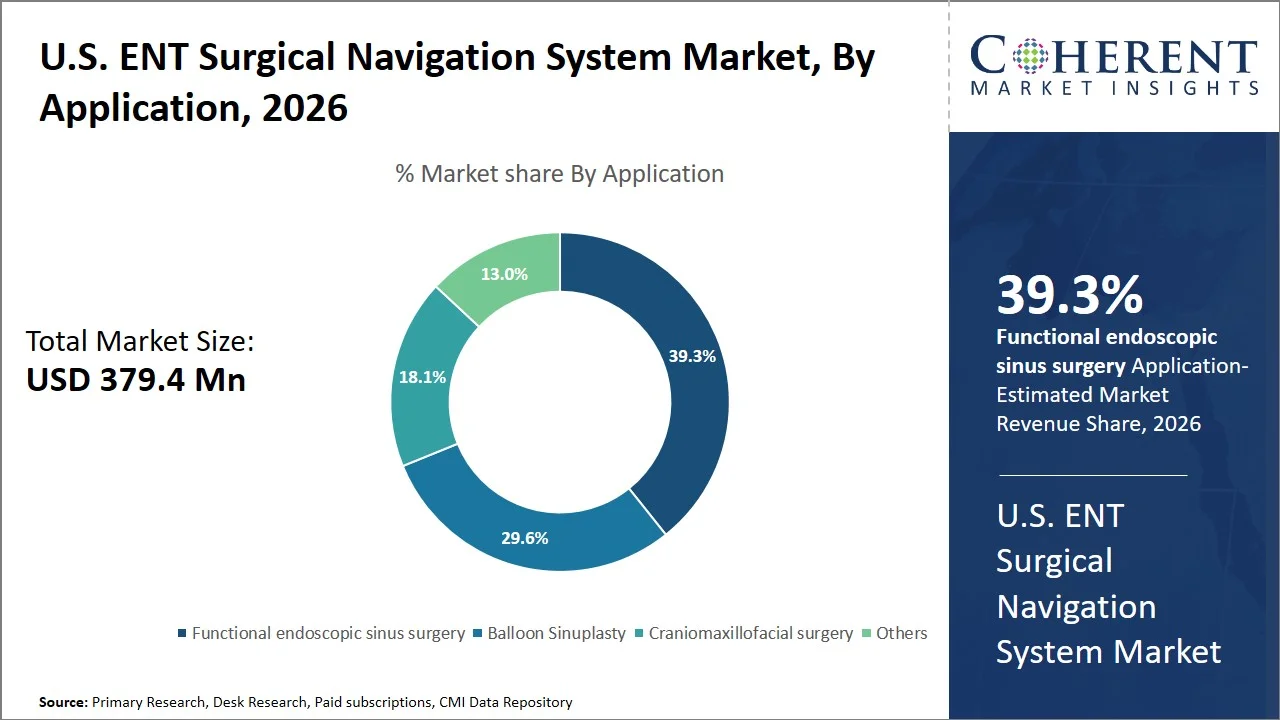
- Functional endoscopic sinus surgery is projected to hold 39.3% of the U.S. ENT surgical navigation system market share in 2026, making it the dominant application segment, supported by the clinical dependence on navigation systems in complex sinus and revision surgeries, in which proximity to key anatomical structures (orbit, brain) requires real-time imaging. For instance, American Academy of Otolaryngology–Head and Neck Surgery clinical practice guidelines advise the application of image guidance in complicated and revision nasal sinus surgery due to the potential benefits in terms of surgical safety and results. (Source: American Academy of Otolaryngology–Head and Neck Surgery)
- Hospitals are projected to hold 42.5% of the U.S. ENT surgical navigation system market share in 2026, making it the dominant end user segment, as tertiary hospitals and academic medical centers conduct most of the complicated ENT surgery and skull base operations and serve as major consumers of expensive navigation systems that come along with CT/MRI imaging machines. For instance, according to the statistics published by the National Center for Health Statistics (NCHS), about 18% of emergency room visits in the U.S. involve advanced imaging such as CT scan or MRI imaging, thus indicating the high degree of reliance on imaging equipment that enables image-guided surgery in hospitals. (Source: Centers for Disease Control and Prevention)
- Increasing Integration with Image-Guided, Minimally Invasive ENT Procedures: The increasing trend towards minimally invasive ENT surgical operations in the U.S., especially functional endoscopic sinus surgery (FESS), is resulting in the increased use of surgical navigation systems. The clinical guidelines and hospital policies for performing surgeries mandate that the surgeon rely on image-guided navigation while working in anatomically complex areas such as the skull base. Thus, there will be an ongoing need for advanced navigation technology systems.
- Expansion of Ambulatory Surgical Centers (ASCs) for ENT Procedures: With the movement of ENT surgeries from hospital environments to Ambulatory Surgical Centers (ASCs) due to U.S. healthcare cost reduction initiatives, there are emerging opportunities for compact and cost-effective surgical navigation technology solutions. Increasingly, Ambulatory Surgical Centers (ASCs) are requiring portable surgical navigation technologies to help carry out outpatient sinus and nasal surgery procedures.
Why Does the Accessories Segment Dominate the U.S. ENT Surgical Navigation System Market?
Accessories are projected to hold a market share of 53.0% in 2026, because these are used or replaced after every procedure rather than capital items, which are purchased only once. Accessories such as trackers and adaptors for imaging instruments are required during image-guided ENT surgeries like functional endoscopic sinus surgery to navigate with accuracy. According to the U.S. FDA, the computer-assisted surgical system uses accessories such as tracking systems to help in guiding surgical instruments (Source: U.S. Food and Drug Administration). This means more usage of accessories in these surgeries, and since accessories are either single-use or sterilized before every operation, there is continuous demand for accessories.
Why is Functional Endoscopic Sinus Surgery Most Preferred Application in U.S. ENT Surgical Navigation System Market?
To learn more about this report, Request Free Sample
Functional endoscopic sinus surgery is projected to hold 39.3% of the market share in 2026, due to the high procedure volume and need for precision imaging to achieve the treatment goal. The functional endoscopic sinus surgery (FESS) is typically performed on chronic rhinosinusitis patients, a condition that affects many individuals in the nation and therefore represents a common ENT surgery procedure. In line with the National Institutes of Health data, the application of image-guided technology in the fields of rhinology and skull base surgeries enhances the success of the surgery by providing real-time imaging of critical anatomical structures (Source: National Library of Medicine). This makes the use of functional endoscopic sinus surgery inevitable due to its high prevalence of disease, minimal invasiveness, and requirement of navigation.
Hospitals Segment Dominates the U.S. ENT Surgical Navigation System Market
The hospitals segment is expected to hold 42.5% of the U.S. ENT surgical navigation system market share in 2026, attributed to their capability to perform large numbers of complex ENT and skull base surgeries that need the support of imaging and navigation. This kind of facility is provided by hospitals as they have the capability of integrating operating rooms imaging facilities, and surgical teams to make full use of the navigation technology. In the U.S., according to the Agency for Healthcare Research and Quality, a significant share of surgical procedures is performed in hospital settings (Source: Agency for Healthcare Research and Quality). Thus, hospitals can be regarded as the major end users of the market owing to the presence of infrastructure for performing surgeries with these advanced facilities.
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
Industry Shift: Growth of Minimally Invasive and Outpatient Surgical Care |
|
|
Regulatory & Technology Shift: Increasing FDA Focus on Digital Health & Interoperability in Surgical Systems (Ongoing) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
(Source: National Institutes of Health, U.S. Food and Drug Administration)
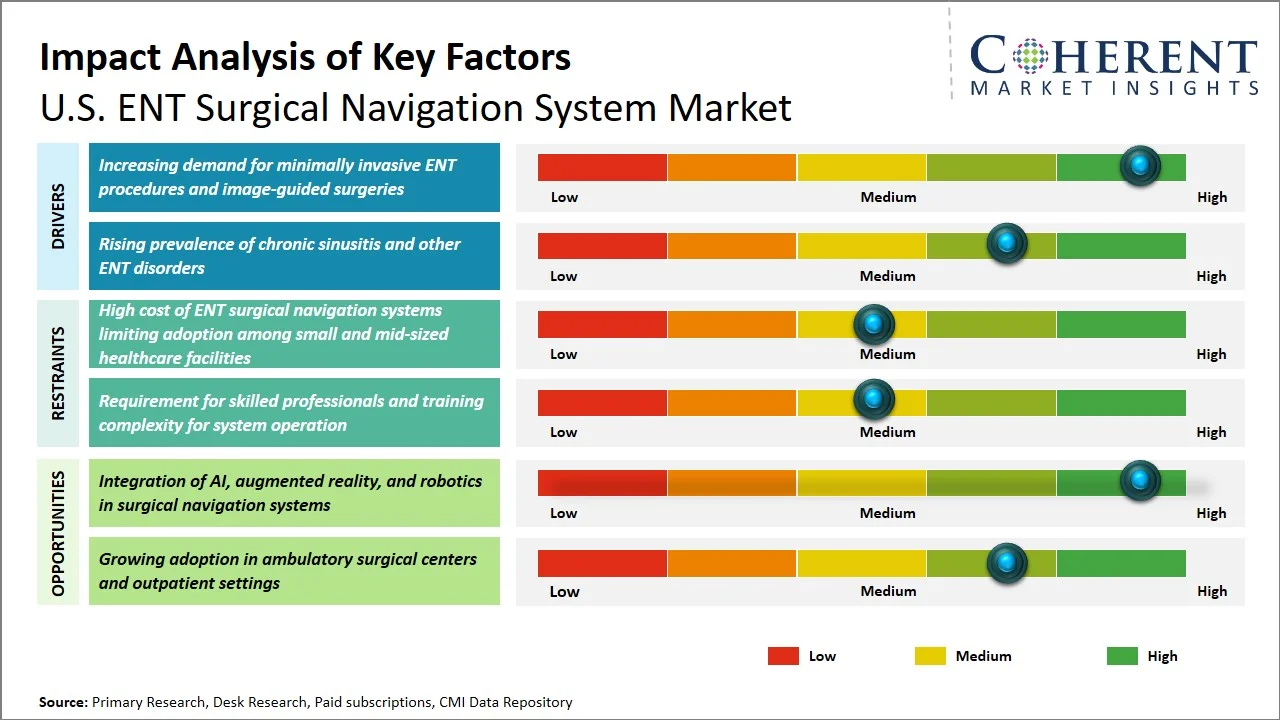
U.S. ENT Surgical Navigation System Market Dynamics
To learn more about this report, Request Free Sample
Market Drivers
- Increasing demand for minimally invasive ENT procedures and image-guided surgeries: The rise in popularity of minimally invasive surgery in ENT including the use of Functional Endoscopic Sinus Surgery (FESS) has been the primary driver behind the increased acceptance of surgical navigation systems in the U.S., given that these procedures have shorter recovery times and good outcomes for patients. In accordance with the American Academy of Otolaryngology–Head and Neck Surgery, endoscopic sinus surgery is usually used in conditions such as chronic rhinosinusitis, hence, there is an elevated demand for these procedures (Source: American Academy of Otolaryngology–Head and Neck Surgery). Moreover, image-guided surgery has been recommended for difficult sinus surgery and skull base surgery in order to improve anatomical accuracy and reduce complications, thereby resulting in an increased adoption of navigation systems. A clinical literature published by National Library of Medicine indicated that there is improvement in accuracy along with good results in rhinology surgery and skull base surgeries using image guidance (Source: National Library of Medicine). This growing number of procedures, along with the importance placed on navigational assistance within ENT surgery to ensure accuracy and safety, is rapidly driving the acceptance of surgical navigation systems across U.S. hospitals and ambulatory surgical centers.
- Rising prevalence of chronic sinusitis and other ENT disorders: The rising incidence rate of chronic sinusitis and other ENT diseases within the U.S. is one of the primary forces behind the utilization of navigated ENT surgeries. According to the Centers for Disease Control and Prevention, there are millions of adult patients who receive the diagnosis of sinusitis per year, making this condition among the most prevalent chronic diseases that require clinical treatment. A large number of them eventually proceed to surgeries, especially functional endoscopic sinus surgery (FESS), which requires a high level of precision on behalf of physicians. Thus, an increasing number of patients alongside with the necessity for precise anatomical navigation during surgeries leads to a high demand for such tools.
Emerging Trends
- Increasing Integration with Robotic-Assisted ENT Procedures: The use of surgical navigation systems along with robotic surgery systems are becoming popular as a way to increase the accuracy of ENT surgeries and skull base surgeries. The combination of the two systems makes it possible for doctors to adopt better control of surgical tools, and thus better surgical outcomes.
- Growing Focus on Data Integration and Interoperability with Hospital Systems: A recent development in the field of ENT surgical navigation systems involves the capability of such navigation systems to seamlessly integrate with hospital IT systems. This is largely due to the increasing need for interoperability in the design of medical devices, especially in the context of the health IT movement in the U.S.
Regulatory & Reimbursement Landscape Shaping the U.S. ENT Surgical Navigation System Market
|
Category |
Authority |
Impact on Market |
Key Insights |
|
Regulatory Approval |
U.S. Food and Drug Administration (FDA) |
High impact |
ENT navigation systems must obtain U.S. FDA clearance before commercialization; recent approvals are expanding use into ENT and cranial procedures, supporting market adoption |
|
Device Classification & Pathway |
FDA (510(k) pathway) |
High impact |
Most surgical navigation systems are cleared via the 510(k) process, requiring demonstration of safety and substantial equivalence to existing devices |
|
Reimbursement Structure (Medicare/ASC) |
Centers for Medicare & Medicaid Services (CMS) |
High impact |
Navigation systems are typically not separately reimbursed; costs are bundled into procedure payments under hospital outpatient and ASC reimbursement models |
|
Coding & Billing Framework |
CMS CPT / HCPCS coding system |
Moderate impact |
Reimbursement depends on accurate coding of ENT procedures (e.g., endoscopic sinus surgery); navigation is included as part of the overall surgical service rather than billed separately |
|
Private Payer Policies |
Commercial insurers (e.g., UHC, EmblemHealth) |
High impact |
Many insurers consider navigation systems as facility equipment, limiting separate reimbursement and influencing hospital purchasing decisions |
|
Clinical Guidelines & Advocacy |
American Academy of Otolaryngology–Head and Neck Surgery (AAO-HNS) |
Moderate impact |
Supports reimbursement for imaging and navigation-assisted procedures when medically necessary, influencing payer policy evolution |
|
Procedure-Based Reimbursement Trends |
CMS OPPS & ASC Payment Rules |
High impact |
Favorable reimbursement for minimally invasive ENT procedures indirectly drives adoption of navigation systems used in these surgeries |
|
Coverage Determination Factors |
Payers & CMS |
Moderate impact |
Coverage is based on medical necessity, clinical evidence, and documentation, not just FDA approval, affecting utilization rates |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
How is the integration of AI, augmented reality, and robotics in surgical navigation systems creating new growth opportunities in the U.S. ENT surgical navigation system market?
Innovation in the application of artificial intelligence (AI), augmented reality (AR), and robotics technology has created tremendous opportunities for the growth of the market in terms of ENT surgical navigation system. The use of AI technology will aid in anatomical mapping and surgical planning, whereas the use of AR will add extra dimensions in terms of visualization of anatomical structures in the surgical field. Robotics can increase accuracy during minimally invasive ENT surgery. For instance, Medtronic's Stealth AXiS combines navigation, imaging, and robotic technology in one package for ENT and cranial surgeries. The integration allows surgeons to position surgical tools accurately and quickly. (Source: Medtronic)
Moreover, Medtronic has collaborated with surgical theater to develop a navigation system that integrates real-time AR visualization software, enabling surgeons to visualize anatomical features that may not be seen inside the surgical field. The technology helps increase surgical accuracy (Source: Medtronic). These advancements are helping to boost the deployment of next-generation systems, allowing higher pricing, more applications in complicated ENT surgeries, and generating new revenue opportunities for the market participants.
Market Players, Key Development, and Competitive Intelligence

To learn more about this report, Request Free Sample
Key Developments
- In March 2026, Medtronic received the U.S. FDA approval for its Stealth AXIS Surgical Navigation System, which is specifically targeted at cranial and ear nose throat (ENT) surgical applications. The Stealth AXIS surgical navigation system improves accuracy and efficiency of image-guided surgery. This event underscores the developments that are constantly taking place in the field of surgical navigation systems. It will further consolidate Medtronic's presence in the ENT surgical navigation system market in the U.S.
- In December 2023, Integra LifeSciences entered into an agreement to buy out Acclarent, one of the largest companies dealing with surgical solutions for ENT. Integra will increase its capacity to provide ENT surgical solutions in minimally invasive processes with the acquisition. This is one of the most recent consolidations in the industry that is geared towards enhancing competition within the U.S. ENT surgical navigation system market.
Competitive Landscape
The U.S. ENT surgical navigation system market is intensely competitive due to constant innovation in the field of image-guided surgeries and the ever-increasing need for precision during ENT surgeries that are less invasive. Major players in the industry like Medtronic, Stryker Corporation, and Johnson & Johnson have been working towards improving the accuracy, real-time image integration, and efficiency of the process for surgeons. There is an increasing focus on the role of artificial intelligence in surgical planning and visualization techniques in intraoperative settings. Key focus areas include:
- Integration of AI and augmented reality for enhanced surgical visualization and planning
- Development of compact and cost-effective navigation systems for ambulatory surgical centers
- Expansion of product portfolios through acquisitions and partnerships in ENT-specific surgical technologies
- Improving system interoperability with CT/MRI platforms and hospital IT infrastructure
Market Report Scope
U.S. ENT Surgical Navigation System Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 379.4 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7.2% | 2033 Value Projection: | USD 617.2 Mn |
| Segments covered: |
|
||
| Companies covered: |
Medtronic, Stryker Corporation, Johnson & Johnson MedTech, Zimmer Biomet, GE HealthCare, Siemens Healthineers, Brainlab, B. Braun, KARL STORZ, and Integra LifeSciences |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Analyst Opinion (Expert Opinion)
- The further trajectory of U.S. ENT surgical navigation system market will be determined by the convergence of navigation systems with artificial intelligence, AR, and robotics used for ENT surgery. Due to the growing digitalization of the entire process of conducting surgery, navigation systems are predicted to be embedded in the entire system of surgery encompassing imaging, planning, and assistance. The growth of the use of minimally invasive surgeries are expected to lead to more compact navigation systems becoming more popular among surgeons.
- Electromagnetic technology for functional endoscopic sinus surgery (FESS) in the U.S. presents the greatest opportunity due to the large number of sinus surgeries performed each year and the requirement for an accurate and flexible tool tracking system. The electromagnetic system provides several benefits, including not being limited by line of sight and flexibility during tool tracking, thus making it ideal for ENT procedures in hospitals with large volumes of cases.
- Strategies related to the implementation of technology and work process improvement can help market participants. The application of surgical planning based on artificial intelligence, analytical tools, and integration with CT/MRI systems are such area that needs consideration. Moreover, the expansion of the company’s product offering to ambulatory surgical centers by using affordable and portable equipment will create additional demand areas.
Market Segmentation
- Product Insights (Revenue, USD Mn, 2021 - 2033)
- System
- Accessories
- Application Insights (Revenue, USD Mn, 2021 - 2033)
- Functional endoscopic sinus surgery
- Balloon Sinuplasty
- Craniomaxillofacial surgery
- Others
- End User Insights (Revenue, USD Mn, 2021 - 2033)
- Hospitals
- Ambulatory Surgery Centers
- ENT Clinics
- Others
- Key Players Insights
- Medtronic
- Stryker Corporation
- Johnson & Johnson MedTech
- Zimmer Biomet
- GE HealthCare
- Siemens Healthineers
- Brainlab
- Braun
- KARL STORZ
- Integra LifeSciences
Sources
Primary Research Interviews
- Chief of Otolaryngology / ENT Surgeons – Image-guided sinus and skull base surgery (U.S. hospitals)
- Director – Surgical Navigation / Operating Room Technologies (U.S. hospital networks)
- Clinical Specialists – ENT navigation systems (medical device companies)
- Ambulatory Surgical Center (ASC) Administrators – ENT procedure adoption and technology usage
Stakeholders
- ENT Surgical Navigation System Providers
- End-use Sectors
- Hospitals (tertiary care & academic medical centers)
- Ambulatory Surgical Centers (ASCs)
- Specialty ENT clinics
- Regulatory & Health Bodies: U.S. Food and Drug Administration (FDA) – medical device approvals & guidance, Centers for Medicare & Medicaid Services (CMS) – reimbursement policies for ENT procedures, American Academy of Otolaryngology–Head and Neck Surgery (AAO-HNS) – clinical guidelines
Databases
- Centers for Medicare & Medicaid Services (CMS) – procedure volumes, reimbursement, ASC utilization
- Healthcare Cost and Utilization Project (HCUP) – ENT surgery hospitalization data
- National Center for Health Statistics (NCHS) – surgical procedure trends
- ClinicalTrials.gov – ENT surgical navigation-related studies
- FDA MAUDE Database – device performance and safety reports
Magazines
- FDA Medical Device Safety Communications – navigation system updates
- NIH Research Matters – surgical innovation and imaging technologies
- AHRQ Patient Safety Network – surgical safety and navigation benefits
- AAO-HNS Bulletin – ENT surgical advancements and best practices
Journals
- Otolaryngology–Head and Neck Surgery – clinical outcomes in ENT navigation
- The Laryngoscope – sinus surgery and image-guided procedures
- International Forum of Allergy & Rhinology – FESS and navigation system usage
- Journal of Neurological Surgery Part B (Skull Base) – skull base surgical navigation
- JAMA Otolaryngology–Head & Neck Surgery – clinical and surgical advancements
Newspapers
- U.S. FDA press announcements – device approvals and clearances
- NIH news releases – surgical innovation and research funding
- HHS press releases – healthcare technology adoption
- Major hospital system announcements on surgical advancements
Associations
- American Academy of Otolaryngology–Head and Neck Surgery (AAO-HNS)
- American College of Surgeons (ACS)
- Healthcare Information and Management Systems Society (HIMSS)
- Society for Minimally Invasive Spine Surgery (relevant for navigation technologies overlap)
Public Domain Sources
- U.S. Food and Drug Administration (FDA) – device approvals and regulatory pathways
- Centers for Medicare & Medicaid Services (CMS) – reimbursement and ASC policies
- Centers for Disease Control and Prevention (CDC) – ENT disease burden and procedure trends
- National Institutes of Health (NIH) – surgical research and imaging technology funding
- Agency for Healthcare Research and Quality (AHRQ) – patient safety and surgical outcomes
Proprietary Elements
- CMI Data Analytics Tool, Proprietary CMI Existing Repository of information for last 10 years.
Share
Share
About Author
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients