U.S. Flowable Hemostat Market Size and Forecast – 2026-2033
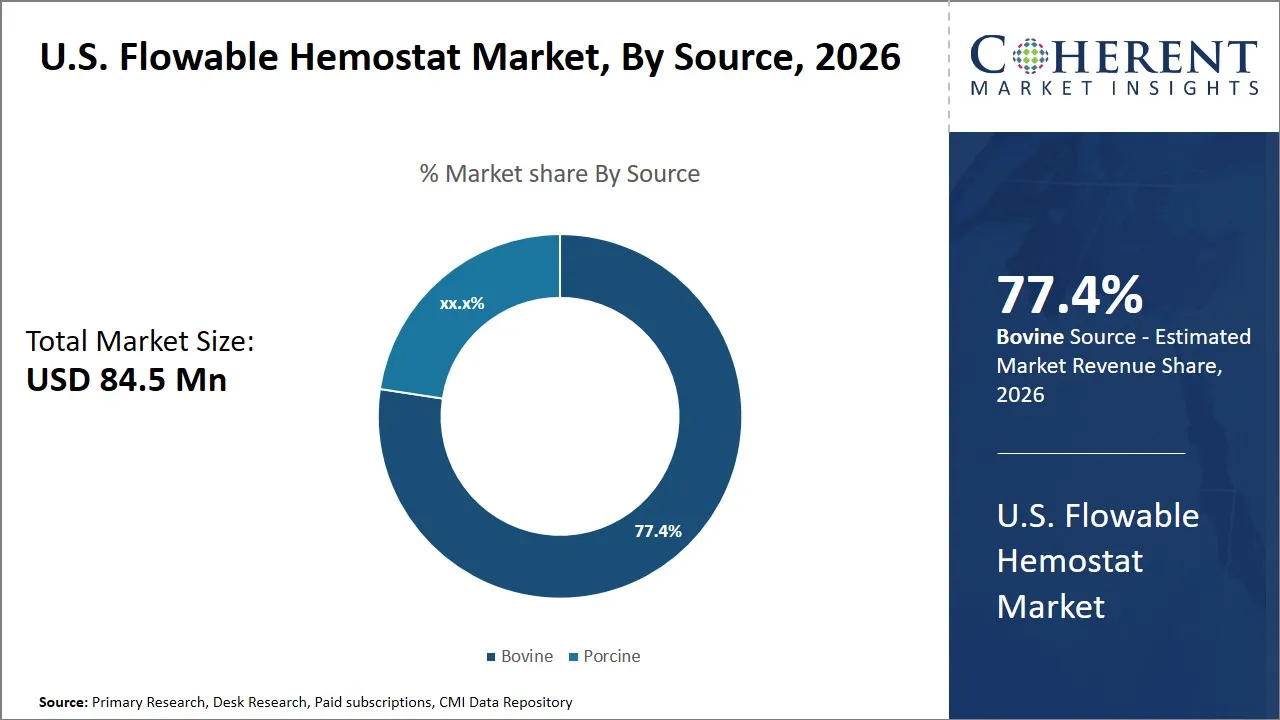
The U.S. flowable hemostat market is estimated to be valued at USD 84.5 Mn in 2026 and is expected to reach USD 114.8 Mn by 2033, exhibiting a compound annual growth rate (CAGR) of 6.2% from 2026 to 2033. This steady growth reflects rising demand for minimally invasive surgical procedures and increased adoption of advanced hemostatic agents in hospitals and outpatient surgical centers across the country.
Key Takeaways of the U.S. Flowable Hemostat Market
- Bovine segment is expected to lead the U.S. flowable hemostat market, capturing 77.4% share in 2026.
- Surgeries segment is estimated to represent 89.0% of the U.S. flowable hemostat market share in 2026.
- Hospital segment is projected to dominate with 60.7% of the U.S. flowable hemostat market share in 2026.
Market Overview
- U.S. flowable hemostat market is witnessing steady growth, driven by the increasing use of advanced bleeding control products in complex surgical procedures across cardiovascular, orthopedic, spine, trauma, and general surgery.
- The market is also benefiting from the broader shift toward minimally invasive and robotic surgeries, where surgeons need hemostatic products that can be applied precisely in narrow and irregular surgical fields.
- The market is further supported by strong opportunities in ambulatory surgery centers and outpatient surgical settings, where speed, convenience, and workflow efficiency are becoming more important in product selection.
- Another important opportunity for the U.S. flowable hemostat market is the expanding surgical burden linked to an aging patient population. Older adults account for a high share of cardiovascular, orthopedic, spine, and other procedures where bleeding management is critical, which supports sustained demand for advanced flowable hemostats in hospital settings.
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
Expansion of minimally invasive and robotic surgeries in the U.S. |
|
|
Continued growth of ambulatory surgery center and outpatient procedure volumes |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Why Does the Bovine Segment Dominate the U.S. Flowable Hemostat Market in 2026?
The bovine segment is expected to hold the largest market share of 77.4% in 2026, as bovine sources offer a well-established safety profile and are recognized for their consistent quality, which assures clinicians of reliability during critical surgical procedures. The widespread application of bovine collagen in the hemostats could be traced back a long time in the medical profession, and as such there is a great deal of clinical confidence and familiarity that contributes to its continued usage.
Furthermore, bovine flowable hemostats are admired due to their high effectiveness of the hemostats. Bovine sources of collagen contain naturally present thrombogenic material that actively stimulates platelet aggregation, followed by the subsequent development of clots, which will help in a faster and effective control of bleeding. This mechanical benefit plays an important role in surgeries where hemostasis is essential and must be prompt and as much as possible to reduce blood loss and shorten the duration of the operation. The biocompatibility of bovine collagen also makes sure that these flowable hemostats blend with human tissue without causing very negative reactions in terms of inflammation.
For instance, in April 2025, Baxter International announced the launch of a room-temperature version of its HEMOPATCH Sealing Hemostat, designed to improve accessibility and readiness in operating rooms. The updated product eliminates refrigeration requirements, making it easier for surgeons to keep the hemostatic patch readily available during open and minimally invasive procedures.
(Source- Baxter International)
Why Does the Surgeries Segment Dominate the U.S. Flowable Hemostat Market in 2026?
The surgeries segment is expected to hold 89.0% of the market share in 2026, predominantly led by general surgeries, which register the highest market share. The broad group of surgeries includes many subtypes such as orthopedic, cardiovascular, gynecological, trauma, and others, with their own unique hemostatic requirements demanding the popularity of flowable hemostats. The overall surge behind the prevailing existence of surgeries in this industry is the growing number and size of surgical procedures being undertaken in this country.
The innovation of technology and an increase in chronic diseases that require surgical intervention have increased the scope of use of flowable hemostats in various specialties. Surgeons are relying on these agents to provide effective bleeding control and minimize intraoperative complications and particularly in minimally invasive or complex surgeries where using traditional techniques such as sutures, and cauterization might be not effective or potentially harmful to adjacent tissues. The flowability of hemostats allows adapting the irregular wound sites and also facilitates quick clotting, which may be invaluable in other operating rooms.
For instance, in March 2025, Medcura announced the formation of a commercial spine advisory board to support market development of LifeGel, a surgical hemostatic agent that has received U.S. Food and Drug Administration Breakthrough Device Designation for spine surgery. This matters because it reflects continued innovation aimed at high-value surgical specialties, reinforcing that surgery remains the core demand center for advanced hemostatic products in the market.
(Source- Medcura)
The Hospital Segment Dominates the U.S. Flowable Hemostat Market
The hospital segment is expected to hold the largest market share of 60.7% in 2026, driven by their complex care capabilities and higher procedural volumes. Hospitals are leading in dealing with various medical and surgical conditions necessitating efficient and effective hemostatic procedures and therefore they are the major purchasers and consumers of flowable hemostats.
Another major growth driver in hospitals in this segment is that they are a full care provider, fully equipped to do high-risk and specialized surgeries which require high quality and advanced hemostatic products. In comparison to smaller clinics or ambulatory surgical centers, hospitals usually deal with trauma cases, heart surgery, cancer surgery and other surgeries demanding that the bleeding is regulated. This expanded scope of services leads to the regular use of flowable hemostats in operating rooms, emergency departments, and the inpatient units.
Adoption Trends in Gelatin-Thrombin Matrix Products Versus Synthetic Alternatives
- Adoption of gelatin-thrombin matrix products continues to be high in the U.S. flowable hemostat market due to the fact that the products are commonly relied on to control moderate to severe bleeding during cardiovascular, orthopedic, spine and general surgeries. Their liquid nature, high compliance to irregular surfaces and rapid clot-promoting effect predisposes them to be of use in operating rooms where quick and dependable hemostasis is essential. These products are usually opted in complicated surgeries by the hospitals and surgeons though they are reported to be more expensive as they may help decrease the amount of blood loss, save time and make the procedure more efficient.
- Synthetic alternatives are finding their way into practice, primarily in operations where convenience of storage, reduced biologic hazard, and price are of greater importance than utmost hemostatic force. The products have become attractive to the facilities who are interested in ready-to-use products that offer longer shelf life and fewer issues associated with the human or animal products. They are being used more selectively in specific low-to-moderate bleeding situations and in outpatient practices, but adoption remains more of a selective use as opposed to gelatin-thrombin matrix products, which are still more clinically favored in more challenging surgical settings.
Market Players, Key Developments, and Competitive Intelligence

To learn more about this report, Request Free Sample
Key Developments
- In May 2025, Merit Medical Systems acquired Biolife Delaware, LLC in a merger transaction. Biolife manufactures hemostatic devices under the StatSeal and WoundSeal brands, making this a relevant 2025 acquisition in the broader U.S. hemostasis products space.
- In April 2025, Teleflex Incorporated received U.S. Food and Drug Administration (FDA) 510(k) clearance for expanded use of its QuikClot Control+ Hemostatic Device, allowing it to be used for all grades of internal and external bleeding across multiple surgical procedures, including orthopedic, gynecologic, and general surgery. The expanded indication broadens its application in hospital operating rooms and trauma care settings.
- In February 2024, Baxter reported the launch of FLOSEAL + RECOTHROM, described as the first active flowable hemostat with recombinant thrombin. The product improves preparation speed and removes reliance on human blood components, supporting broader use in surgical bleeding control.
Growing Use of Flowable Hemostats in Minimally Invasive Surgeries
- Flowable hemostats are also known to be increasingly utilized in minimally invasive surgeries since they may be placed accurately through small openings and shaped appropriately to uneven bleeding. Surgeons have little tactile input and visualization in laparoscopic, robotic and endoscopic procedures and thereby flowable products are useful in managing bleeding in inaccessible regions whereby sutures, clips, or traditional sponges may not work effectively. They are particularly beneficial in surgical operation procedures that require delicate tissues and confined surgical areas because they can be easily dispersed and their location is maintained at the target site.
- Their application is increasing in minimally invasive general surgery, urology, gynecology, cardiovascular and spine surgery as hospitals scramble to shorten operating time, reduce blood loss and speed up recovery. Flowable hemostats facilitate these objectives by aiding the surgeon to attain quick and efficient hemostasis without necessitating the shift to open surgery which is important in preserving the clinical and financial benefits of minimally invasive procedures. With the further growth of robotic-assisted and outpatient surgeries, the requirements of easy-to-use and highly efficient flowable hemostatic products are likely to increase as well.
Top Strategies Followed by U.S. Flowable Hemostat Market Players
|
Player Type |
Strategic Focus |
Examples |
|
Established Market Leaders |
Leading players maintain their position by investing heavily in research and development to improve product safety, biocompatibility, and speed of bleeding control. They also strengthen their market presence through partnerships with hospitals, healthcare systems, and industry stakeholders while expanding distribution in high-growth regions. |
In May 2025, Baxter launched a room-temperature version of Hemopatch Sealing Hemostat, designed for rapid and effective hemostasis with easier storage and wider surgical usability. This reflects how major players use product innovation and commercial expansion to reinforce market leadership. |
|
Mid-Level Players |
Mid-level players compete by offering cost-effective flowable hemostat solutions that deliver acceptable clinical performance at a lower price point. Their products are often more appealing to smaller hospitals, ambulatory surgery centers, and providers that face tighter budget controls. These companies also rely on manufacturing partnerships and targeted collaborations to improve efficiency, expand portfolios, and strengthen market reach without matching the scale of top-tier firms. |
Advanced Medical Solutions has built market relevance by focusing on surgical sealants and hemostatic technologies that support value-driven healthcare settings, helping it competes in segments where affordability and practical performance matter more than premium branding. |
|
Small-Scale Players |
Small-scale players compete by focusing on niche innovation rather than broad market coverage. They often develop specialized hemostatic products for specific surgical needs, such as minimally invasive procedures, targeted bleeding control, or biomaterial-based formulations that stand out from standard offerings. To improve visibility and market access, they usually depend on local partnerships, selective distribution, and surgeon-focused adoption strategies. |
Marine Polymer Technologies has built relevance through specialized hemostatic products designed for surgical and trauma applications, showing how smaller companies can stay competitive by targeting focused clinical use cases instead of competing head-on with larger manufacturers. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Market Report Scope
U.S. Flowable Hemostat Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 84.5 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 6.2% | 2033 Value Projection: | USD 114.8 Mn |
| Segments covered: |
|
||
| Companies covered: |
Baxter International Inc, Teleflex Incorporated, Becton Dickinson and Company, Ethicon Inc, Pfizer Inc, Integra LifeSciences Holdings Corporation, Medtronic plc, B Braun Melsungen AG, CSL Behring LLC, Artivion Inc, Marine Polymer Technologies Inc, Advanced Medical Solutions Group plc, Biom Up SA, Hemostasis LLC, and Zimmer Biomet Holdings Inc |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
U.S. Flowable Hemostat Market Dynamics
To learn more about this report, Request Free Sample
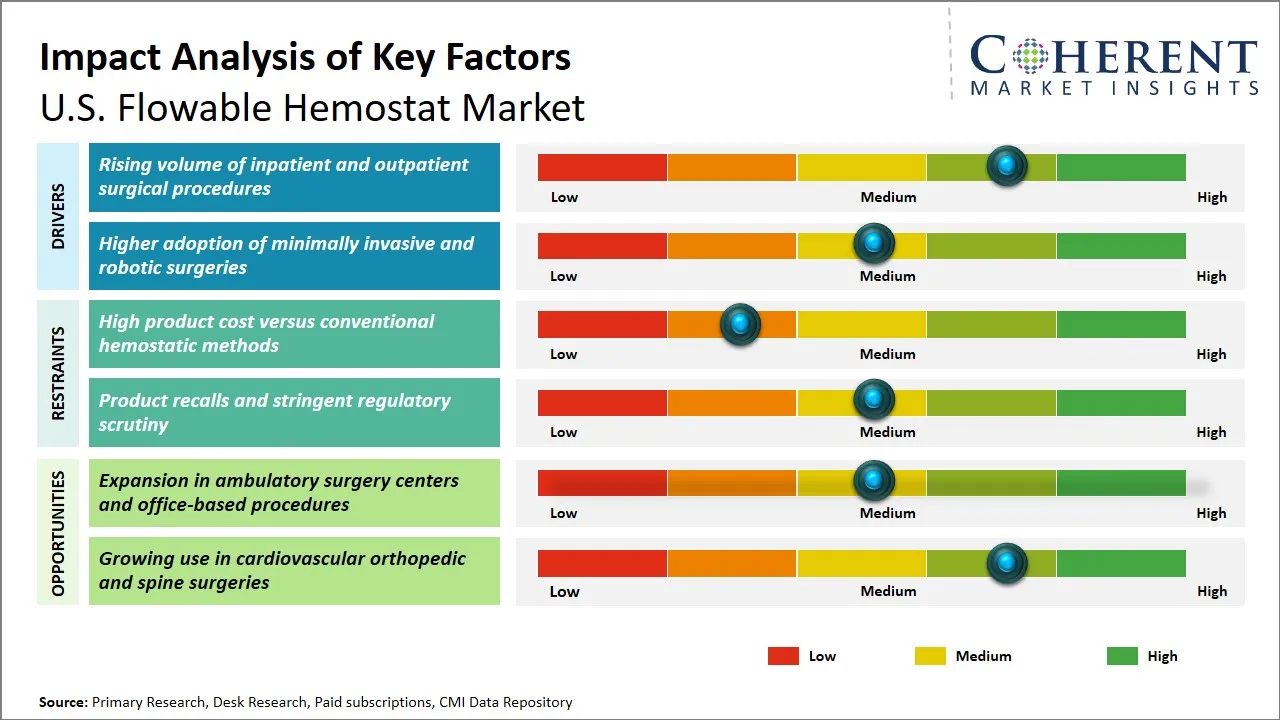
U.S. Flowable Hemostat Market Driver - Increasing Volume of Inpatient and Outpatient Surgical Procedures
The increased number of inpatient and outpatient surgeries conducted in the U.S. is another contributing factor to the demand of flowable hemostats. With the growing size of the healthcare infrastructure and the development of the surgical methodology, the number of surgeries performed in many different fields and types of general surgeries (cardiovascular, orthopedic, and the least invasive method) increases significantly. Flowable hemostats, which are considered to be easy to use and efficient in controlling bleeding in uncommon and challenging wound locations, are turning out to be a necessity in operation rooms to deal with surgical bleeding in an effective way. The trend in moving towards outpatient surgeries, in turn, caused by technological advances and patients being more comfortable with low-invasiveness and shorter recovery period, further increases the necessity of high-quality hemostatic agents that would ensure patient safety and decrease the rate of complications.
For instance, in May 2025, Intuitive received U.S. FDA clearance for expanded da Vinci SP colorectal use, reflecting rising minimally invasive surgical procedure volumes in the U.S., which supports demand for flowable hemostats used for rapid bleeding control during surgery.
(Source- Intuitive)
U.S. Flowable Hemostat Market Opportunity - Expansion in Ambulatory Surgery Centers and Office-Based Procedures
The market for the flowable hemostat in the U.S. can be seen to gain a lot with the fast growth in ambulatory surgery centers (ASCs) and the growing trend in office-based surgeries. ASCs represent a less expensive, yet more efficient, alternative to the conventional hospital environment, addressing a rise in patient demand of less invasive procedures with shorter recovery times. This has resulted in the increase in outpatient procedures with the need to look into demand of convenient, easy to use and effective hemostatic agents such as flowable hemostats increasing. Flowable hemostats deliver specific benefits in these environments because of its versatility to non-uniform wound surfaces, quickness in activity, and simplicity of use, all of which fit into the fast-paced working environment of ASCs and office-based clinics. Also, the lower costs of operation and reimbursement benefits of ASCs are causing providers to use advanced surgical adjuncts to enhance the outcomes of the procedures and patient satisfaction.
For instance, in November 2024, the Centers for Medicare & Medicaid Services finalized the CY 2025 outpatient and ambulatory surgical center payment updates, supporting continued growth in outpatient procedures and indirectly strengthening demand for flowable hemostats in high-efficiency surgical settings.
Analyst Opinion (Expert Opinion)
- The U.S. flowable hemostat market is growing because hospitals and surgeons need faster bleeding control in complex and minimally invasive procedures. Demand is strongest in cardiovascular, orthopedic, spine, trauma, and robotic surgeries where speed and precision directly affect outcomes. Innovation is also supporting growth, with Baxter highlighting FLOSEAL + RECOTHROM in 2024 and Teleflex expanding QuikClot Control+ indications through U.S. Food and Drug Administration clearance in 2025. The main upside is wider use in ambulatory and robotic surgery, while high cost and strict hospital purchasing reviews remain the biggest barriers.
- Industry events continue to shape adoption by giving surgeons and perioperative teams direct exposure to new products and techniques. The AORN Global Surgical Conference & Expo 2024 and the American College of Surgeons Clinical Congress remain important platforms for product visibility, training, and clinical discussion, which matters in a market where evidence and surgeon familiarity drive uptake more than broad policy support.
Market Segmentation
- Source Insights (Revenue, USD Mn, 2021 - 2033)
- Bovine
- Porcine
- Application Insights (Revenue, USD Mn, 2021 - 2033)
- Surgeries
- Orthopedic Surgery
- Gynecological Surgery
- Cardiovascular Surgery
- Others
- Trauma (Accidents, Burns and Others)
- Surgeries
- End User Insights (Revenue, USD Mn, 2021 - 2033)
- Hospital
- Clinics
- Ambulatory Surgical Centers
- Others
- Sales Channel Insights (Revenue, USD Mn, 2021 - 2033)
- Offline
- Online
- Key Players Insights
- Baxter International Inc
- Teleflex Incorporated
- Becton Dickinson and Company
- Ethicon Inc
- Pfizer Inc
- Integra LifeSciences Holdings Corporation
- Medtronic plc
- B Braun Melsungen AG
- CSL Behring LLC
- Artivion Inc
- Marine Polymer Technologies Inc
- Advanced Medical Solutions Group plc
- Biom Up SA
- Hemostasis LLC
- Zimmer Biomet Holdings Inc
Sources
Primary Research Interviews
Industry Stakeholders list
- Trauma surgeons
- General surgeons
- Hospital procurement managers
- Operating room directors
- Medical device regulatory affairs managers
- Hemostasis product development specialists
End-users list
- Ambulatory surgery center administrators
- Surgical nurses
- Perioperative pharmacists
- Hospital materials management teams
- Wound care specialists
- Interventional radiologists
Government and International Databases
- U.S. Food and Drug Administration
- ClinicalTrials.gov
- Centers for Medicare & Medicaid Services
- National Center for Biotechnology Information
- World Health Organization
Trade Publications
- MedTech Dive
- Medical Design & Outsourcing
- Pharmaceutical Technology
- Infection Control Today
- Med Device Online
- Outsourced Pharma
Academic Journals
- Annals of Surgery
- Journal of Trauma and Acute Care Surgery
- The Annals of Thoracic Surgery
- Transfusion
- Journal of Surgical Research
- AORN Journal
Reputable Newspapers
- The Wall Street Journal
- Financial Times
- The New York Times
- The Washington Post
- USA Today
Industry Associations
- Association of periOperative Registered Nurses
- American College of Surgeons
- Society of Thoracic Surgeons
- American Hospital Association
- AABB
- Society for the Advancement of Patient Blood Management
Public Domain Resources
- FDA device databases
- FDA recall database
- ClinicalTrials.gov
- Centers for Disease Control and Prevention
- Agency for Healthcare Research and Quality
Proprietary Elements
- CMI Data Analytics Tool: Proprietary analytics tool to analyze real-time market trends, consumer behavior, and technology adoption in market
- Proprietary CMI Existing Repository of Information for Last 10 Years
Share
Share
About Author
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients