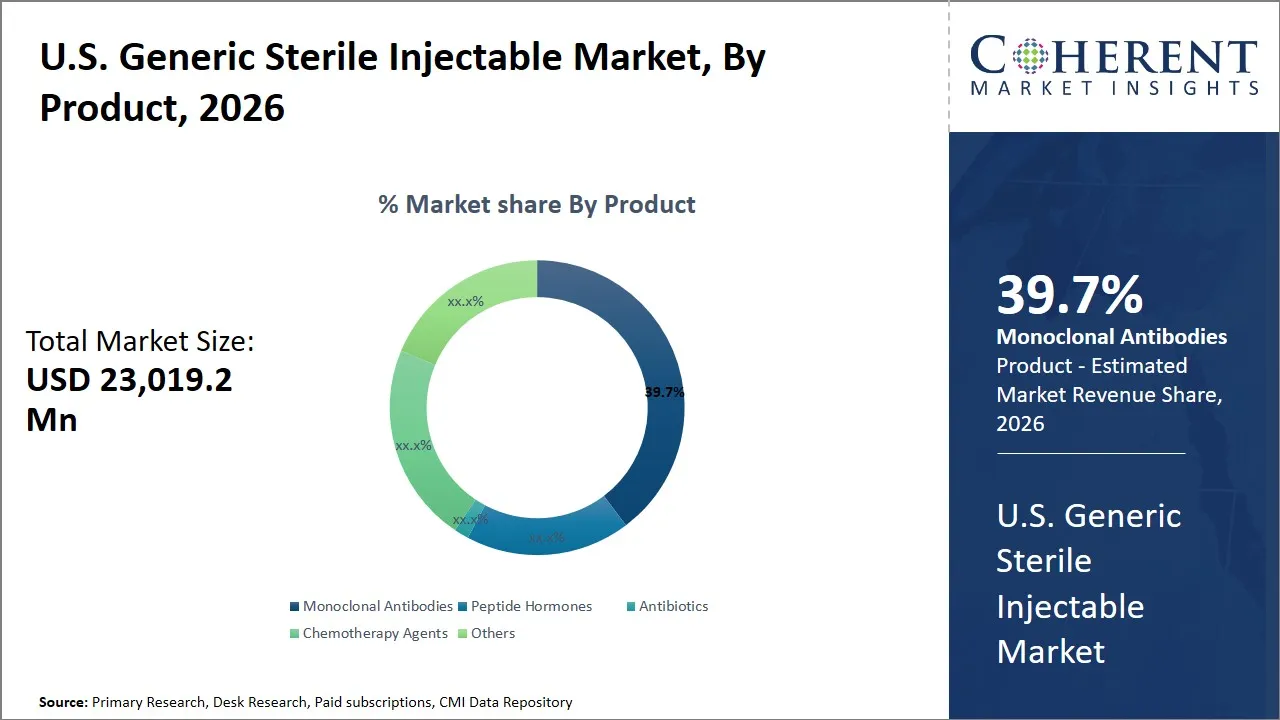
U.S. Generic Sterile Injectable Market Size and Forecast – 2026 to 2033
The U.S. generic sterile injectable market is estimated to be valued at USD 23,019.2 Mn in 2026 and is expected to reach USD 38,797.8 Mn by 2033, exhibiting a compound annual growth rate (CAGR) of 11% from 2026 to 2033. This steady growth reflects sustained demand for cost-effective injectable therapies within healthcare, driven by increasing prevalence of chronic diseases and a shift towards outpatient care settings.
Key Takeaways of the U.S. Generic Sterile Injectable Market
- Monoclonal antibodies segment is expected to lead the U.S. generic sterile injectable market, capturing 39.7% share in 2026.
- Oncology segment is estimated to represent 25.9% of the U.S. generic sterile injectable market share in 2026.
- Intravenous segment is projected to dominate with 48.2% of the U.S. generic sterile injectable market share in 2026.
Market Overview
- The U.S. generic sterile injectable market is a hospital-driven market supported by steady demand for cost-effective drugs used in oncology, anesthesia, anti-infectives, critical care, and emergency treatment.
- Market growth is being driven by rising pressure on healthcare systems to reduce treatment costs, along with continued demand for high-volume injectable medicines in inpatient and outpatient settings.
- Recurrent shortages of essential sterile injectables have made supply reliability a major competitive factor, pushing buyers to prefer manufacturers with strong production continuity and quality track records.
- The market is technically demanding because sterile manufacturing requires strict contamination control, regulatory compliance, and high capital investment, which creates barriers for new entrants.
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
Ongoing drug shortages across essential sterile injectables |
|
|
Rising focus on domestic manufacturing capacity |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Why Does the Monoclonal Antibodies Segment Dominate the U.S. Generic Sterile Injectable Market in 2026?
The monoclonal antibodies segment is expected to hold the largest market share of 39.7% in 2026, driven by their expanding role in precision medicine and biologic therapies. The monoclonal antibodies are engineered proteins that are created to target a given set of antigens and thus very useful in the treatment of complex diseases particularly cancer and autoimmune diseases. The increase in chronic diseases like cancer and auto immune diseases has made these targeted therapies important in demand because they have better efficacy and safety profiles as compared to the conventional small molecule drugs.
The other important reason that has led to the success of monoclonal antibodies is the current trend of biosimilar development. As many patents of a variety of monoclonal antibodies have expired, generic companies have spent millions of dollars to develop biosimilar products, which offer more affordable treatments but have clinical effectiveness comparable to their original. The U.S. has also modified its regulatory environment to facilitate approval pathways of biosimilars to increase the availability and uptake of biosimilars in hospital formularies and specialty clinics.
For instance, in April 2025, Teva and Samsung Bioepis launched EPYSQLI (eculizumab-aagh) injection in the U.S., a biosimilar to Soliris for rare diseases including PNH, aHUS, and gMG. The launch expanded access to monoclonal antibody injectables in the U.S. generic sterile injectable market at a lower cost than the reference product.
(Source - Teva)
Why Does the Oncology Segment Dominate the U.S. Generic Sterile Injectable Market in 2026?
The oncology segment is expected to hold 25.9% of the market share in 2026, fueled by increasing cancer incidence and the continual introduction of novel therapeutic agents. Cancer has been one of the major causes of morbidity and mortality and there has been a general trend of using injectable pharmaceuticals that provide more specific, systemic treatment with greater effectiveness. The modern oncological regimens implemented in different solid tumors and hematological malignancies are based on injectable chemotherapy agents and biosimilar monoclonal antibodies.
This segment growth is closely associated with the development of immuno-oncology, precision medicine, and combination therapy strategies, which need injectable biologics to realize significant clinical outcomes. This is because sterile injectable formulations have a rapid onset of action and bioavailability that is significant in aggressive disease conditions and the accurate administration of the drug.
For instance, in August 2024, Lupin launched Doxorubicin Hydrochloride Liposome Injection in the U.S. in 20 mg/10 mL and 50 mg/25 mL single-dose vials after U.S. FDA approval received by ForDoz Pharma. It is a generic equivalent of Doxil indicated for ovarian cancer, AIDS-related Kaposi’s sarcoma, and multiple myeloma.
(Source - Lupin)
The Intravenous Segment Dominates the U.S. Generic Sterile Injectable Market
The intravenous segment is expected to hold the largest market share of 48.2% in 2026, because it enables immediate and controlled delivery of medications directly into the bloodstream. This quick systemic delivery is critical to acute care as well as oncology, and acute therapeutic cases where rapid response of the drug is required to treat life threatening conditions or intensified infections.
Intravenous (IV) route is preferred with a broad scope of generic sterile injectables such as monoclonal antibodies, chemotherapy, antibiotics, and support treatment. IV administration gives the opportunity to accurately control the dosage and infusion rates that improves treatment effectiveness and minimizes adverse events, particularly during complex treatment regimens that demand close attention.
Regulatory Scenario Analysis for Sterile Injectable Approvals in the U.S.
|
Regulatory Scenario Metric |
What it Measures |
Why it Matters for Generic Sterile Injectable Market |
Interpretation |
|
Number of ANDA Approvals for Sterile Injectables (FDA) |
Total FDA approvals granted for Abbreviated New Drug Applications related to sterile injectable products |
Reflects the pace of generic entry, regulatory activity, and approval momentum in the sterile injectables space |
A higher number indicates a more active approval environment, rising competition, and broader product availability |
|
Number of First Generic Injectable Approvals |
Number of FDA approvals granted to the first generic version of a branded sterile injectable drug |
Shows how quickly branded monopoly products are opening up to generic competition and highlights major market-entry opportunities |
A higher number suggests stronger pipeline conversion, more competitive disruption, and greater pricing pressure on branded products |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
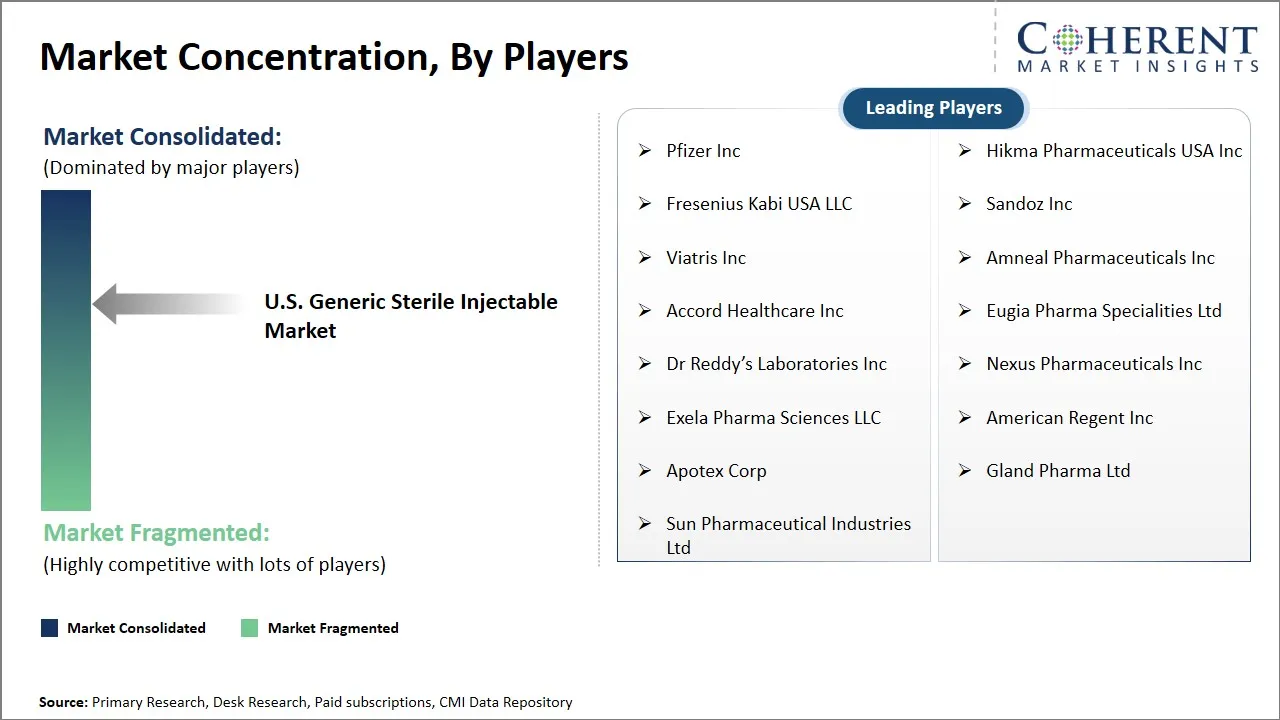
Market Players, Key Developments, and Competitive Intelligence
To learn more about this report, Request Free Sample
Key Developments
- In November 2025, Zydus Lifesciences announced that it had entered into an exclusive licensing and commercialization agreement with U.S.-based, RK Pharma Inc. for a novel sterile injectable oncology supportive care product targeting the U.S. market. Under the agreement, RK Pharma will manufacture and supply the finished injectable, while Zydus will handle New Drug Application (NDA) submission and commercialization in the U.S.
- In March 2025, Mallinckrodt plc announced that it had entered into a definitive agreement to merge with Endo, Inc. in a transaction valued at approximately US$ 6.7 billion, creating a scaled and diversified pharmaceutical company with a strong U.S. footprint. Under the terms of the deal, Endo shareholders will receive around US$ 80 million in cash and hold a 49.9% stake in the combined entity, while existing Mallinckrodt shareholders will retain 50.1%.
- In April 2024, Eli Lilly and Company announced that it will acquire a sterile injectable manufacturing facility from Nexus Pharmaceuticals located in Pleasant Prairie, Wisconsin, as part of its strategy to expand production capacity for injectable medicines in the U.S.
Patent Expiry Pipeline for Injectable Drugs in the U.S.
- The U.S. injectable drug patent expiry pipeline represents the number of branded injectable products expiring exclusivity over a particular time. This is one of the most important future pointers in the generic sterile injectables business because any single patent expiry may provide the opportunity of a window to file the abbreviated new drug application, first-generic products, and wider-scale competition penetration. An increased off-patent pipeline is typically an indicator of greater future potential of manufacturers who have the ability to develop injectables and regulatory capacity.
- The more injectable drugs are going off-patent in the market, the larger the pool of opportunity that can be addressed by generic firms and contract manufacturers. It has the ability to facilitate portfolio growth in the oncology, anti-infective, anesthetics and hospital-based treatment and also to further pressure the originator brands pricing. This pipeline is particularly relevant in sterile injectables since technical complexity, barriers in manufacturing and reliability of supply remain a factor influencing which companies can effectively transform expiry of patents into commercial market entry.
Top Strategies Followed by U.S. Generic Sterile Injectable Market Players
|
Player Type |
Strategic Focus |
Examples |
|
Established Market Leaders |
Established players lead the U.S. generic sterile injectable market by using strong R&D, regulatory expertise, and large-scale sterile manufacturing to keep their portfolios competitive. They also strengthen their position through partnerships with hospitals, distributors, and healthcare networks while expanding distribution to capture demand across both mature and emerging markets. |
Fresenius Kabi strengthened its position in the U.S. sterile injectables space through continued portfolio expansion and hospital-focused distribution, including the December 17, 2024, U.S. launch of Epinephrine Injection, USP, 1 mg/1 mL in single-dose vials, which added a first-generic injectable product to its hospital medicines portfolio. |
|
Mid-Level Players |
Mid-level players compete by offering affordable sterile injectables without sacrificing core quality and compliance. They usually strengthen their position through partnerships, shared manufacturing capabilities, and tighter supply-chain execution, which helps them control costs, improve delivery reliability, and stay relevant against larger firms. |
On May 8, 2025, Amneal partnered with Apiject to expand sterile and blow-fill-seal manufacturing capabilities in the U.S., a move aimed at increasing production efficiency and strengthening its position in advanced injectable delivery formats. |
|
Small-Scale Players |
Small-scale players compete by targeting niche sterile injectable segments and offering specialized products where larger companies may not move quickly. Their advantage is agility: they can focus on limited-competition therapies, adopt specialized manufacturing approaches, and build local partnerships to improve visibility and market access. |
Nexus Pharmaceuticals is a good fit for this strategy. On October 24, 2024, the U.S. FDA approved Minocycline for Injection from Nexus Pharmaceuticals, LLC as a first generic, showing how a smaller player can build position through targeted injectable launches in narrower hospital-use categories. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Market Report Scope
U.S. Generic Sterile Injectable Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 23,019.2 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 11% | 2033 Value Projection: | USD 38,797.8 Mn |
| Segments covered: |
|
||
| Companies covered: |
Pfizer Inc, Hikma Pharmaceuticals USA Inc, Fresenius Kabi USA LLC, Sandoz Inc, Viatris Inc, Amneal Pharmaceuticals Inc, Accord Healthcare Inc, Eugia Pharma Specialities Ltd, Dr Reddy’s Laboratories Inc, Nexus Pharmaceuticals Inc, Exela Pharma Sciences LLC, American Regent Inc, Apotex Corp, Gland Pharma Ltd, and Sun Pharmaceutical Industries Ltd |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
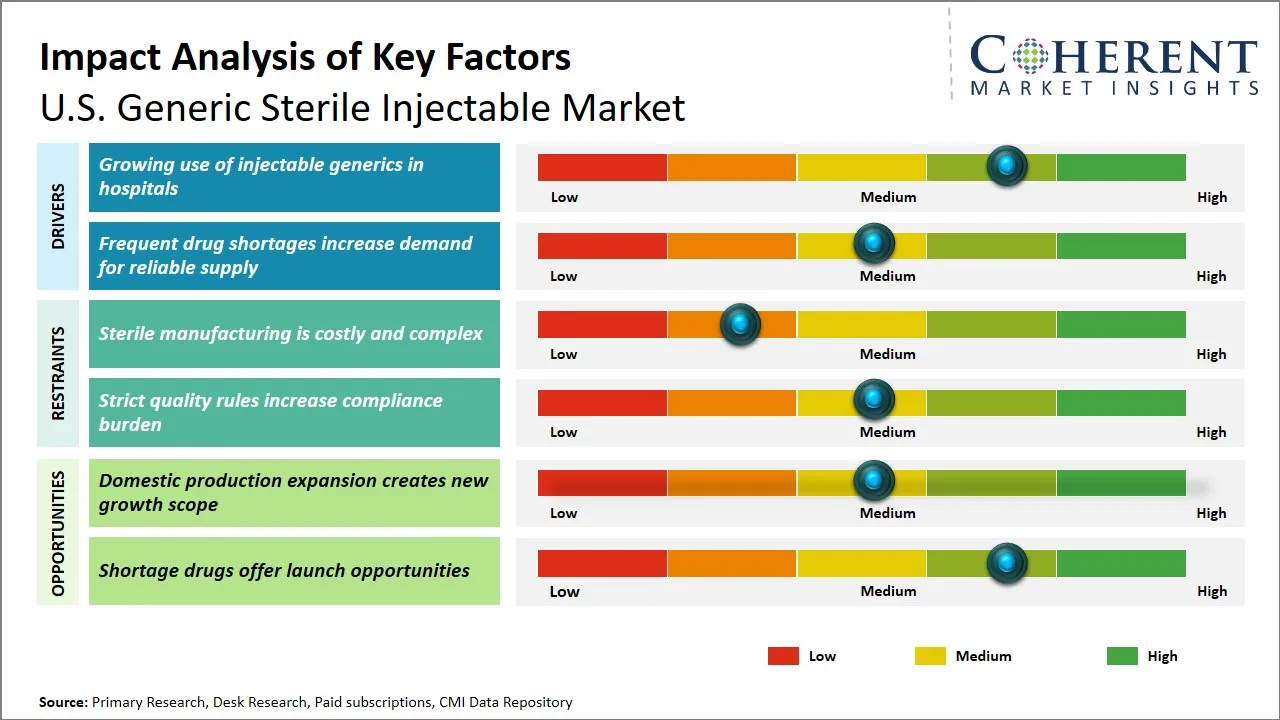
U.S. Generic Sterile Injectable Market Dynamics
To learn more about this report, Request Free Sample
U.S. Generic Sterile Injectable Market Driver - Growing Use of Injectable Generics in Hospitals
The use of generic sterile injectables in hospitals is another major force that is influencing the U.S. generic sterile injectable market. With the increasing costs of healthcare, hospitals always seek cheaper substitutes of branded injectable medications so that they can address the increasing costs without affecting the care of the patients. Generic sterile injectables have the same therapeutic value as their branded counterparts, but at a significantly reduced price, thus appealing to the hospital formulary. Also, new breakthroughs in manufacturing technologies have enhanced the quality and supply of generic sterile injectables, and this further stimulates its use in critical care, anesthesia, chemotherapy, and infection suppression. The regulatory processes have also been simplified meaning that generic injectable products can be approved and accepted in the market faster.
For instance, in December 2024, Endo, Inc. announced the U.S. launch of Doxycycline for Injection, USP, expanding its sterile injectable portfolio for hospital and institutional use. The product is indicated for the treatment of various bacterial infections when oral therapy is not appropriate, providing healthcare providers with an additional intravenous antibiotic option. The launch supports increasing hospital demand for reliable, cost-effective generic injectable therapies and strengthens Endo’s position in the U.S. generic sterile injectable market.
(Source- Endo, Inc.)
U.S. Generic Sterile Injectable Market Opportunity - Domestic Production Expansion Creates New Growth Scope
The development of the local manufacturing potential offers a serious growth prospective to the U.S. generic sterile injectable market. The demand of sterile injectables has been on the increase due to the growing number of chronic illnesses, hospitalizations, and outpatient procedures, which has led to shortcomings in the supply chain, particularly those that rely on the manufacture of injectables abroad. In order to reduce risks related to global disruptions, i.e. pandemics, trade limitations and quality control issues, investors and manufacturers are focusing on reshoring production plants to the U.S. Not only does this increase the resilience of the supply chain, but also leads to innovation of the manufacturing process with the use of more sophisticated technologies such as continuous manufacturing and automation.
For instance, in June 28, 2025, Hikma Pharmaceuticals USA announced a USD 1 billion investment plan through 2030 to expand its U.S. manufacturing and research and development capabilities for essential generic medicines, including sterile injectables. The investment focuses on increasing domestic production capacity, strengthening supply chain resilience, and supporting the availability of critical hospital medicines across the United States, reinforcing Hikma’s long-term commitment to the U.S. pharmaceutical manufacturing ecosystem.
(Source- Hikma Pharmaceuticals USA)
Analyst Opinion (Expert Opinion)
- The U.S. generic sterile injectable market is expanding due to the demand of the hospitals to have cheaper drugs and at the same time the demand of the hospitals to have trustworthy supply after frequent shortages in oncology and emergency care and other vital hospital drugs. U.S. FDA encouragement of generic competition and quality of manufacturing is assisting but the greatest opportunities lie in complex injectables, ready-to-use products and shortage-prone products where the technical obstacles are high. The biggest issue is that sterile production is uncompromising, and breaches of compliance, closure of the plant, and pressure on the margins are continuing to restrain growth. The 2023 AAM Access! and 2025 PDA conferences have strengthened the emphasis of central quality, resilience, and regulatory execution on this market.
- Recent trends indicate that the market is shifting towards the expansion of domestic capacity and supply security. The Virginia manufacturing facility run by Civica is one of the largest, which was constructed to facilitate large-scale manufacturing of the necessary sterile medicines, and the EQUIP-A-Pharma initiative at ASPR is also supporting the advanced manufacturing plans to enhance the U.S. drug supply chains. The introduction of products like the generic epinephrine injection Fresenius Kabi is also an indication that the companies are focusing on the hospital markets that deal with such short-age sensitive markets where demand is predictable and entry into the market can be commercially significant.
Market Segmentation
- Product Insights (Revenue, USD Mn, 2021 - 2033)
- Monoclonal Antibodies
- Peptide Hormones
- Antibiotics
- Chemotherapy Agents
- Others
- Therapeutic Application Insights (Revenue, USD Mn, 2021 - 2033)
- Oncology
- Metabolic Disorders
- Cardiovascular Diseases
- Musculoskeletal Disorders
- Autoimmune Disorders
- Neurological Disorders
- Respiratory Disorders
- Bacterial Disorders
- Others (Viral Disorders, etc.)
- Route of Administration Insights (Revenue, USD Mn, 2021 - 2033)
- Intravenous
- Intramuscular
- Subcutaneous
- Others (Intradermal, etc.)
- Packaging Type Insights (Revenue, USD Mn, 2021 - 2033)
- Vials
- Ampoules
- Prefilled Syringes
- Cartridges
- IV Bags
- Age Group Insights (Revenue, USD Mn, 2021 - 2033)
- Pediatric
- Adult
- Geriatric
- Distribution Channel Insights (Revenue, USD Mn, 2021 - 2033)
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
- Key Players Insights
- Pfizer Inc
- Hikma Pharmaceuticals USA Inc
- Fresenius Kabi USA LLC
- Sandoz Inc
- Viatris Inc
- Amneal Pharmaceuticals Inc
- Accord Healthcare Inc
- Eugia Pharma Specialities Ltd
- Dr Reddy’s Laboratories Inc
- Nexus Pharmaceuticals Inc
- Exela Pharma Sciences LLC
- American Regent Inc
- Apotex Corp
- Gland Pharma Ltd
- Sun Pharmaceutical Industries Ltd
Sources
Primary Research Interviews
Industry Stakeholders list
- FDA Office of Generic Drugs
- U.S. Pharmacopeia
- Association for Accessible Medicines
- Parenteral Drug Association
- International Society for Pharmaceutical Engineering
- Contract Development and Manufacturing Organization executives
End Users
- Hospital pharmacists
- Procurement heads at health systems
- Oncology infusion center directors
- Group purchasing organization sourcing managers
- Sterile compounding pharmacy leaders
- Anesthesiology department buyers
Government and International Databases
- U.S. FDA Drug Shortages Database
- Drugs@FDA
- FDA Orange Book
- FDA National Drug Code Directory
- Centers for Medicare & Medicaid Services Data
Trade Publications
- Drug Topics
- Pharmaceutical Commerce
- BioPharma Dive
- Fierce Pharma
- Pharmaceutical Technology
- American Journal of Health-System Pharmacy
Academic Journals
- Clinical Pharmacology & Therapeutics
- Health Affairs
- Journal of Pharmaceutical Sciences
- American Journal of Health-System Pharmacy
- Value in Health
- Nature Reviews Drug Discovery
Reputable Newspapers
- The Wall Street Journal
- The New York Times
- Financial Times
- The Washington Post
Industry Associations
- Association for Accessible Medicines
- American Society of Health-System Pharmacists
- Parenteral Drug Association
- International Society for Pharmaceutical Engineering
- Healthcare Distribution Alliance
- American Hospital Association
Public Domain Resources
- U.S. Pharmacopeia Drug Shortages reports
- ASHP Current Drug Shortages
- U.S. Department of Health and Human Services
- Office of the Assistant Secretary for Preparedness and Response
- National Library of Medicine
- Congressional Research Service
Proprietary Elements
- CMI Data Analytics Tool: Proprietary analytics tool to analyze real-time market trends, consumer behavior, and technology adoption in market
- Proprietary CMI Existing Repository of Information for Last 8 Years
Share
Share
About Author
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients