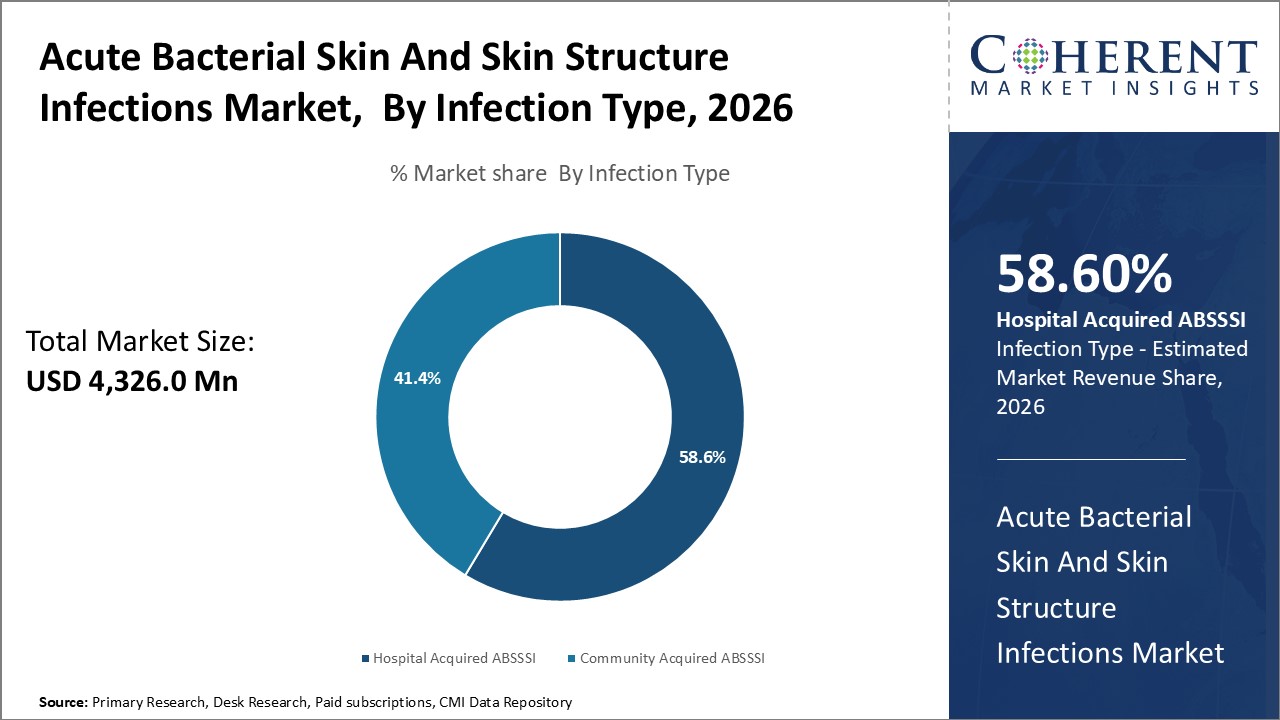
Global acute bacterial skin and skin structure infections market is estimated to be valued at USD 4,326.0 Mn in 2026 and is expected to reach USD 6,591.0 Mn by 2033, exhibiting a compound annual growth rate (CAGR) of 6.2% from 2026 to 2033.
Discover market dynamics shaping the industry: Request Free Sample
There has been increase in prevalence of acute bacterial skin and skin structure infections worldwide due to changing lifestyles and environmental conditions. The market growth is driven by increasing number of surgical procedures that require post-operative infection management. Rising geriatric population prone to such infections further drive the market growth.
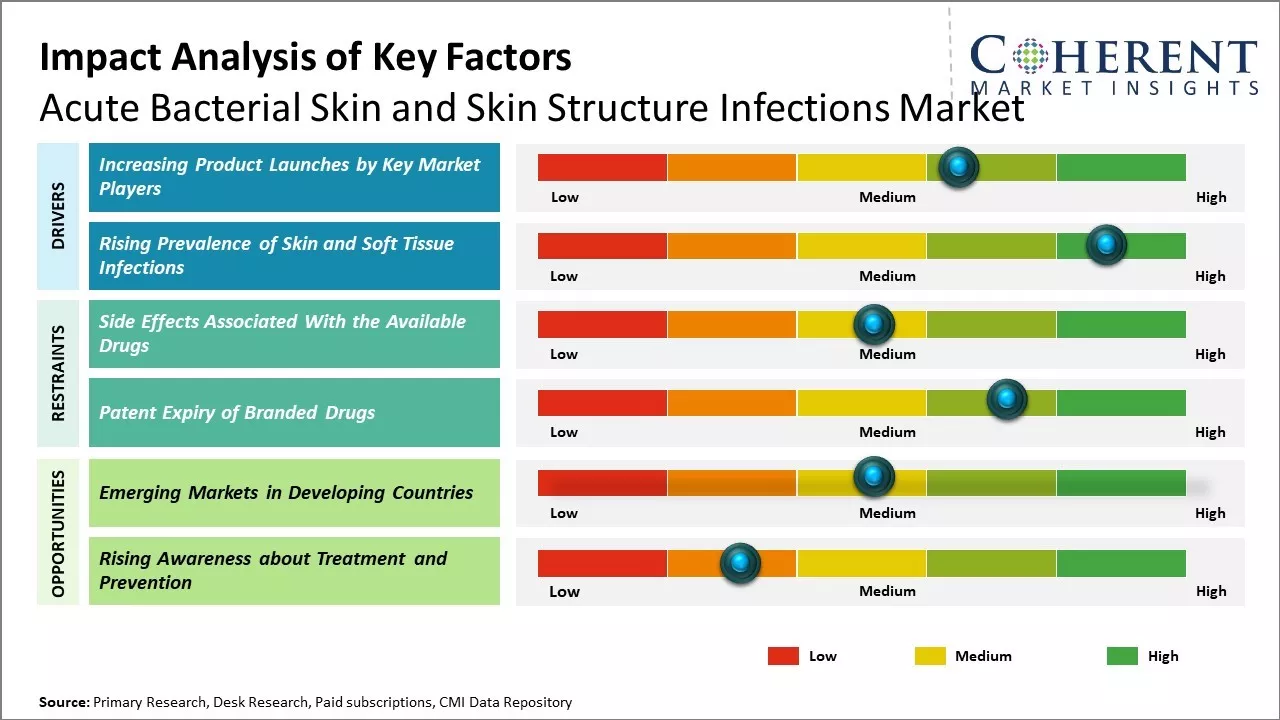
Increasing Product Launches by Key Market Players
Market players are involved in launching new drugs, and this is expected to drive growth of the global acute bacterial skin and skin structure infections market over the forecast period. For instance, on July 7, 2021, Melinta Therapeutics, a pharmaceutical company, launched its product- KIMYRSA. KIMYRSA is a lipoglycopeptide antibiotic that provide a complete course of treatment for acute bacterial skin and skin structure infections. KIMYRSA is effective in a single dose of 1200mg injection.
Get actionable strategies to beat competition: Request Free Sample
Rising Prevalence of Skin and Soft Tissue InfectionsOne of the key drivers fueling growth of global acute bacterial skin and skin structure infections market is rising prevalence of various types of skin infections across both developed and developing regions. Skin and soft tissue infections include a wide range of conditions that affect the epidermis and soft tissues just below the skin. These infections can range from relatively minor and superficial infections to serious and even life-threatening deep infections. Cellulitis is one of the most common acute bacterial skin infections characterized by diffuse swelling and inflammation of the deep layers of the skin and subcutaneous tissues. According to various studies, the global incidence of cellulitis is estimated to be between 33 to 126 cases per 100,000 population per year. The rate of hospitalization due to cellulitis has also increased significantly over the last decade. Increasing prevalence of cellulitis can primarily be attributed to factors such as aging population, rising incidence of diabetes - a major risk factor, and increased microbial resistance. Similarly, wound and surgical site infections are also becoming more common globally due to growing number of surgeries and invasive procedures performed every year.

To learn more about this report, Request Free Sample
Market Challenge – Side Effects Associated With the Available DrugsIncreasing side effects associated with the available drugs such as tigecycline for the treatment of acute bacterial skin and skin structure infections can hamper growth of the global acute bacterial skin and skin structure infections market over the forecast period. For instance, according to the National Institute of Health, U.S., Tigecycline injection have many side effects such as dizziness, vomiting, diarrhea, nausea, headache, pain, itching of the vagina, and others. These side effects may turn into severe symptoms such as heart attack or some forms of cancer.
Market Opportunity – Rising Awareness about Treatment and Prevention
Rising awareness about treatment and prevention could provide significant opportunities for growth in the global acute bacterial skin and skin structure infections market in the near future. Various awareness campaigns by non-profit public health organizations have highlighted the importance of completing the prescribed course of antibiotics and seeking medical advice for recurring or severe symptoms. Rising awareness directly impacts patients' healthcare seeking behavior and compliance with medications. More individuals are now willing to visit a doctor or clinic at the first signs of infection like redness, swelling or pus, rather than adopting an avoidant approach. As a result, infections are caught at an early stage allowing for quicker recovery through appropriate antibiotic therapy. Early detection and management can prevent infections from spreading deeper into tissues or the bloodstream, thus, reducing the need for interventions like surgical drainage or hospitalization.
Discover high revenue pocket segments and roadmap to it: Request Free Sample
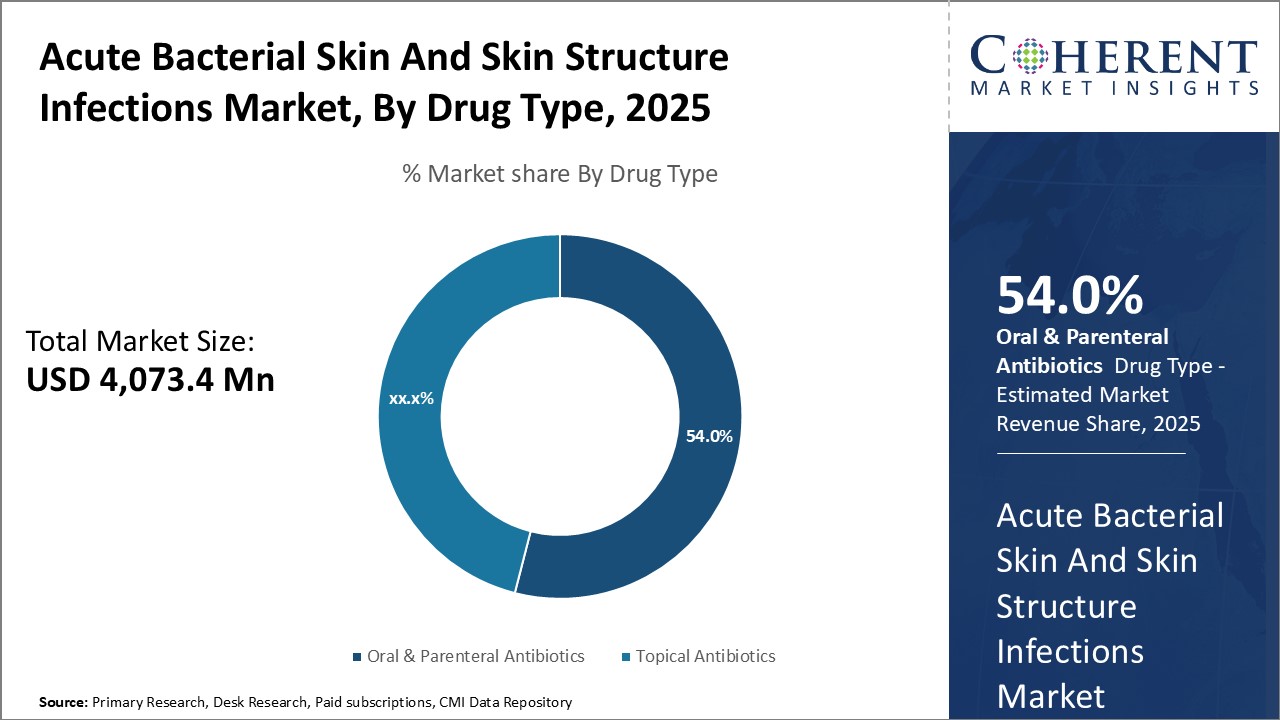
Insights, By Drug Type: Efficacy and broad-spectrum coverage drives the oral & parenteral antibiotics segment growthDrug Type segment is sub-segmented into oral & parenteral antibiotics and topical antibiotics. Oral & parenteral antibiotics sub-segment is estimated to hold 55.0% of the market share in 2026, owing to their high efficacy against a wide range of bacterial infections. Oral antibiotics are the most preferred first line of treatment due to their ease of administration through the oral route. Parenteral antibiotics are used when oral administration is not possible due to nausea, vomiting or other reasons. These provide a higher degree of bioavailability as the drug directly enters the bloodstream after intravenous injection or infusion. This ensures greater tissue penetration and achievement of therapeutic drug levels quickly. Parenteral antibiotics play a crucial role in treating severe cases of ABSSSI that have progressed to bacteremia or sepsis. Several new generation injectable antibiotics with activity against drug-resistant strains are expanding the use of parenteral drugs.
To learn more about this report, Request Free Sample
Insights, By Infection Type: High occurrence in hospitals drives Hospital Acquired ABSSSI segment
Infection Type segment is sub-segmented into hospital acquired ABSSSI and community acquired ABSSSI. Hospital acquired ABSSSI sub-segment is estimated to hold 58.6% of the market share in 2026, due to its high occurrence in hospital settings. Healthcare associated ABSSSI constitutes a major burden in hospitals due to increasing antimicrobial resistance and complex patient profiles seen in tertiary care centres. Prolonged hospital stay, surgery/trauma, insertion of medical devices like catheters, joint replacements and others compromises the skin integrity, thus, making patients vulnerable to exogenous nosocomial infections.
The hospital environment serves as a reservoir for multi-drug resistant organisms. Pathogens like MRSA, VRE, ESBL-producing bacteria and Pseudomonas aeruginosa that cause virulent ABSSSI are commonly acquired through contact with colonized or infected patients, health workers or contaminated surfaces/equipment in hospitals. Immune-suppressed patients undergoing cancer therapy or organ transplantation are at high risk of severe hospital acquired infections. Without timely treatment, these infections can lead to life threatening sepsis.
Insights, By Distribution Channel: Logistic advantages drive Hospital Pharmacies distribution channel
Distribution Channel segment is sub-segmented into hospital pharmacies, retail pharmacies, online pharmacies. Hospital pharmacies sub-segment is estimated to hold 45.3% of the market share in 2026 owing to logistic advantages over other channels. ABSSSI patients are commonly treated within a hospital setting by specialists like dermatologists or infectious disease physicians. Hospital pharmacies are equipped to provide the extensive formulary that includes both conventional and specialized parenteral anti-infectives needed for managing severe cases. The pharmacovigilance activities like therapeutic drug monitoring are also more feasible when the patient is hospitalized. Retail pharmacies and online channels have limitations in providing these specialized services. Home healthcare gained prominence during the pandemic but hospital admissions are still preferred for resistant/complicated ABSSSI.
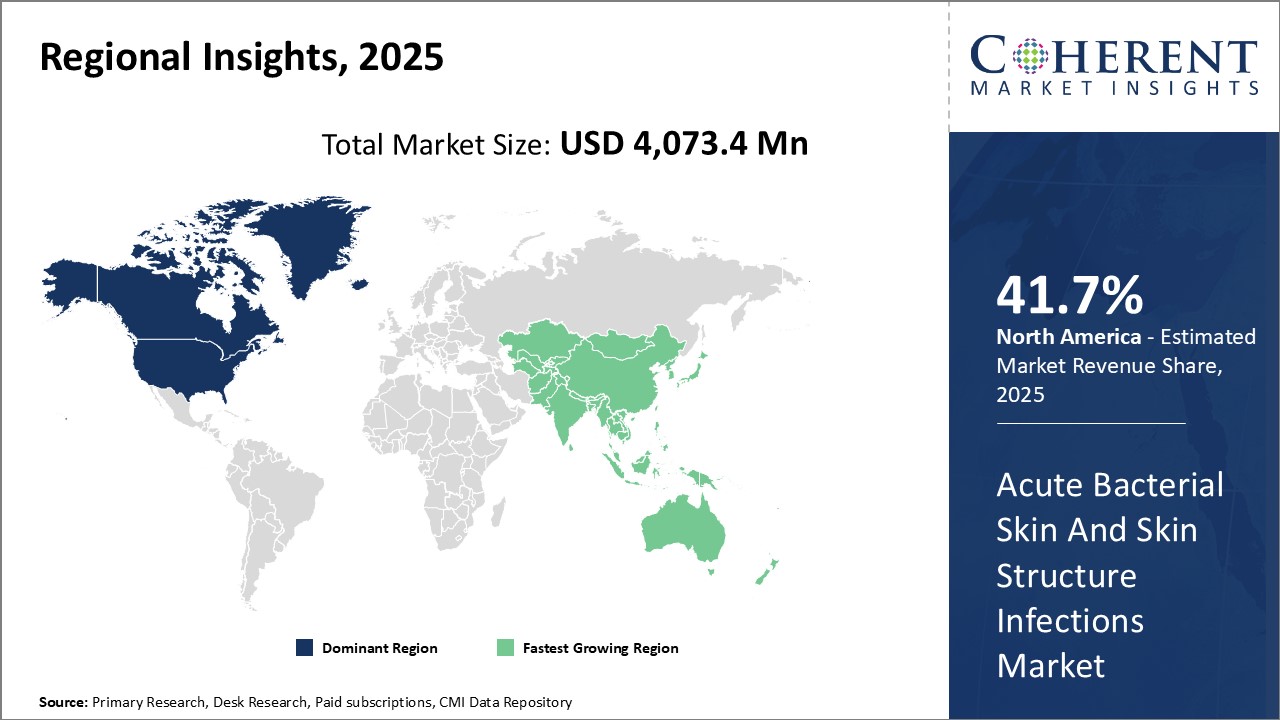
Need a Different Region or Segment? Request Free Sample
North America remains the dominant region in the global acute bacterial skin and skin structure infections market and is estimated to hold 41.7% of the market share in 2026 due to factors such as high per capita healthcare expenditure and presence of many leading industry players in the region. The U.S. accounts for the largest share in the North American market due to wide availability of advanced treatment options and growing awareness about skin infections and their management. Moreover, the reimbursement.
Asia Pacific has emerged as the fastest growing regional market for acute bacterial skin and skin structure infections treatment. Countries like China, India, Japan and South Korea are witnessing increasing prevalence of various skin infections due to growing urbanization, expanding industrial sectors and changing lifestyles. Rising incidence of hospital-acquired and surgical-site skin infections have also boosted demand for effective antibacterial drugs in Asia Pacific nations. Growing medical tourism and rising per capita incomes have made advanced treatment more affordable in the region. Furthermore, the presence of many global pharmaceutical manufacturers in Asia Pacific through their regional production facilities ensures adequate supply and lower prices of drugs.
Acute Bacterial Skin And Skin Structure Infections Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 4,326.0 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 6.2% | 2033 Value Projection: | USD 6,591.0 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
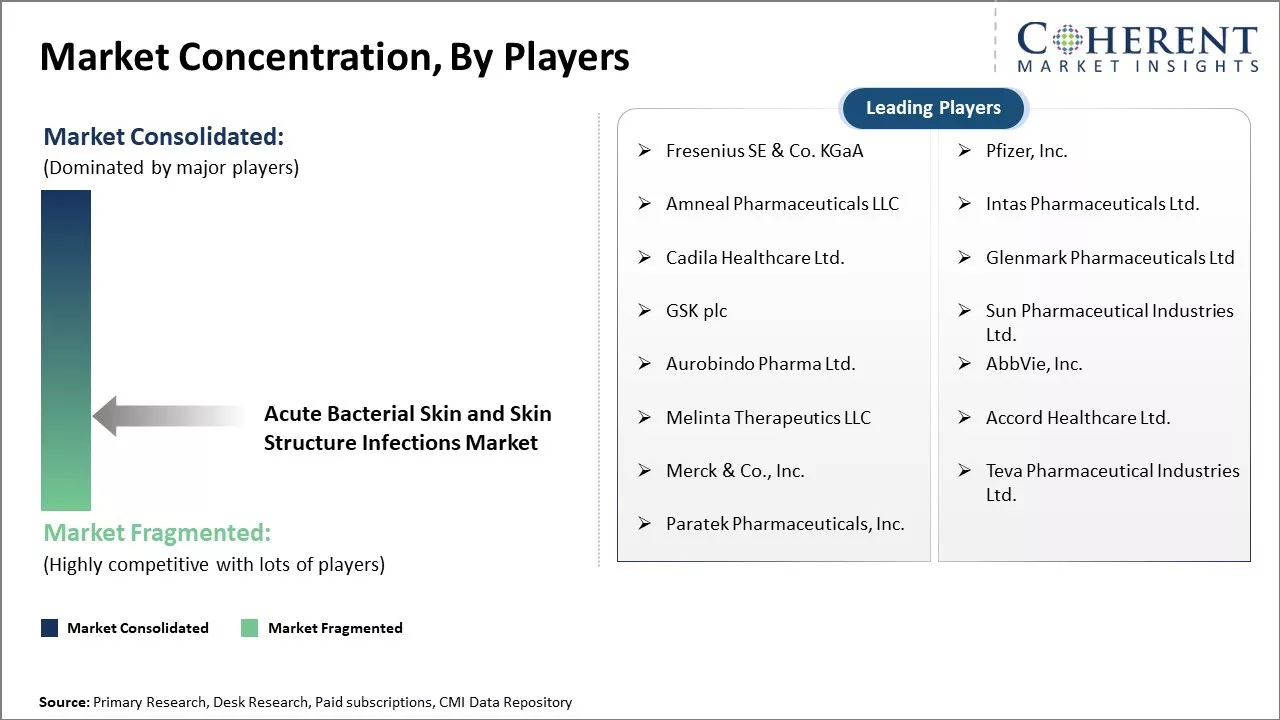
Fresenius SE & Co. KGaA, Pfizer, Inc., Amneal Pharmaceuticals LLC, Intas Pharmaceuticals Ltd., Cadila Healthcare Ltd., Glenmark Pharmaceuticals Ltd, GSK plc, Sun Pharmaceutical Industries Ltd., Aurobindo Pharma Ltd., AbbVie, Inc., Melinta Therapeutics LLC, Accord Healthcare Ltd., Merck & Co., Inc., Teva Pharmaceutical Industries Ltd., Paratek Pharmaceuticals, Inc |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients