The androgenetic alopecia treatment market size is anticipated to grow at a CAGR of 8.4% with USD 2.62 Bn in 2026 and is expected to reach USD 4.5 Bn in 2033. The market is growing with the rising diagnosis of pattern hair loss, topical minoxidil use, oral prescription therapies, hair transplant procedures, and dermatology clinic visits. As per NIH MedlinePlus, androgenetic , thereby contributing to the treatment uptake.
On the basis of drug type, the topical agents segment accounts for the largest androgenetic alopecia treatment market share of 45.20% in 2026. The segment’s growth is because topical therapies, especially minoxidil-based foams and solutions, remain the most widely accessible, non-invasive, and label-supported treatment option for male and female pattern hair loss. DailyMed, maintained by the U.S. National Library of Medicine, lists 5% minoxidil foam as a hair regrowth treatment for men and women, while 2% minoxidil topical solution is also shown to regrow hair in women with thinning on the top of the scalp.
The segment is also supported by repeat-use requirements and ongoing topical innovation. According to the data from DailyMed, minoxidil results may be visible after 2 months with twice-daily use, while some men may need at least 4 months. The continual use is necessary to maintain the regrowth. In April 2026, ClinicalTrials.gov listed new topical androgenetic alopecia studies comparing peptide-based hair serum and topical 2-deoxy-D-ribose hydrogel against topical minoxidil, thereby showing that minoxidil is still the key benchmark for next-generation topical therapies.
To learn more about this report, Request Free Sample
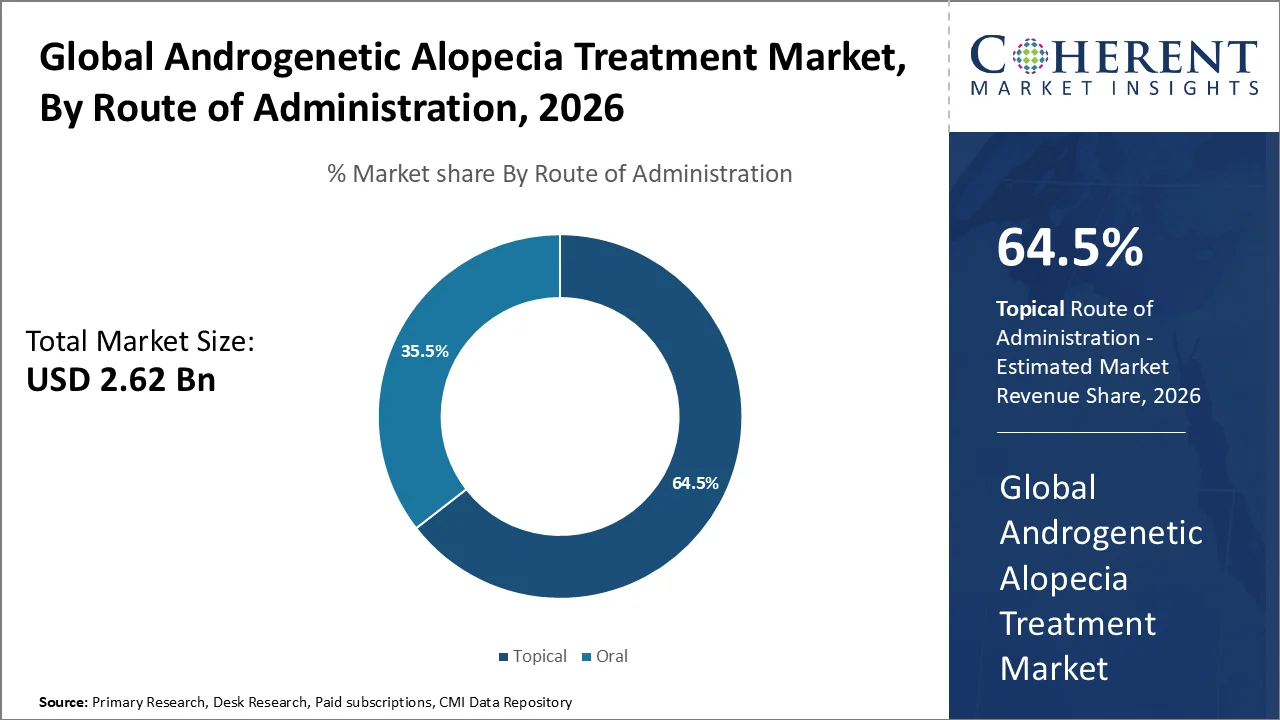
The topical segment accounts for the largest androgenetic alopecia treatment market share of 64.5% in 2026. The segment’s growth is driven by increasing awareness of early intervention benefits, higher dermatology consultations, and a growing preference for non‑invasive regimens. Topical therapies are the most accessible and frequently adopted treatment for androgenetic alopecia mainly owing to ease of application, minimal systemic effects compared with oral drugs, as well as the widespread availability of FDA‑approved minoxidil solutions and foams. The topical administration allows direct delivery to affected scalp regions, supports long‑term compliance, and reduces risks associated with systemic treatment modalities.
The male pattern hair loss prevalence increases with age, with 16% of men aged 18, thereby emphasizing the demand for topical treatments in age groups. The research also indicates that 67.1% of men as well as 23.9% of women are affected by androgenetic alopecia in population, thus underlining the widespread need for effective treatments. The female pattern hair loss, thereby constituting to the demand for topical products.
In December 2025, Cosmo Pharmaceuticals N.V. announced strong topline results from its two pivotal Phase III trials of clascoterone 5% topical solution for male androgenetic alopecia (AGA), also known as male-pattern hair loss. These results represent a potential first major therapeutic breakthrough in hair-loss treatment in over three decades.
U.S. demand is rising as androgenetic alopecia remains a common, progressive, and long-term condition, encouraging wider use of topical therapies, oral DHT-blocking drugs, hair restoration procedures, and dermatologist-led treatment plans.
MedlinePlus Genetics, from NIH/NLM, states that more than 50% of men above age 50 have some degree of hair loss. This shift is also backed by stronger late-stage clinical activity. ClinicalTrials.gov lists a Phase 3 clascoterone solution study for male pattern hair loss, with 703 participants, U.S. trial locations, FDA-regulated drug oversight, along with March 2026 status verification.
Drug developers are therefore moving toward scalp-targeted, topical, and anti-androgen approaches designed to improve local delivery, adherence, and cosmetic acceptability. This pipeline activity is making innovation central to U.S. market growth with patients and dermatologists opting for alternatives beyond older minoxidil- and finasteride-based treatment routes.
In April 2026, Veradermics announced positive Phase 2/3 results for VDPHL01, an extended-release oral minoxidil formula particularly for male pattern hair loss. The study showed important hair-count improvement versus placebo at Month 6, thereby suggesting the product as a potential first FDA-approved oral non-hormonal treatment for pattern hair loss if approved.
A substantial shift is emerging in androgenetic alopecia treatment with the development of stem-cell reactivation therapies which aim to stimulate new hair growth rather than only preserving existing follicles. PP405, a topical regenerative therapy developed by Pelage Pharmaceuticals, is one of the most promising candidates. It is designed to reactivate dormant hair follicle stem cells and is advancing through the FDA clinical development pathway.
In July 2025, Pelage Pharmaceuticals announced positive Phase 2a trial results for PP405 in 78 men as well as women with androgenetic alopecia. In the Phase 2a trial, PP405 was tested in 78 men as well as women with androgenetic alopecia, thus showing groundbreaking results. The early findings revealed that 31% of treated men with higher hair loss severity achieved more than 20% improvement in hair density by week 8, compared to 0% in the placebo group.
This breakthrough could change how AGA is treated by focusing on reactivating hair follicles instead of just managing the symptoms. Currently, no treatments on the market reawaken dormant follicles. This makes PP405 a promising advancement that could provide hope for those struggling with hair loss.
|
Current Event |
Description and its Impact |
|
FDA Safety Alert on Compounded Topical Finasteride Products |
|
|
FDA Warning Letter on Unapproved Minoxidil Hair Regrowth Products |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
To learn more about this report, Request Free Sample
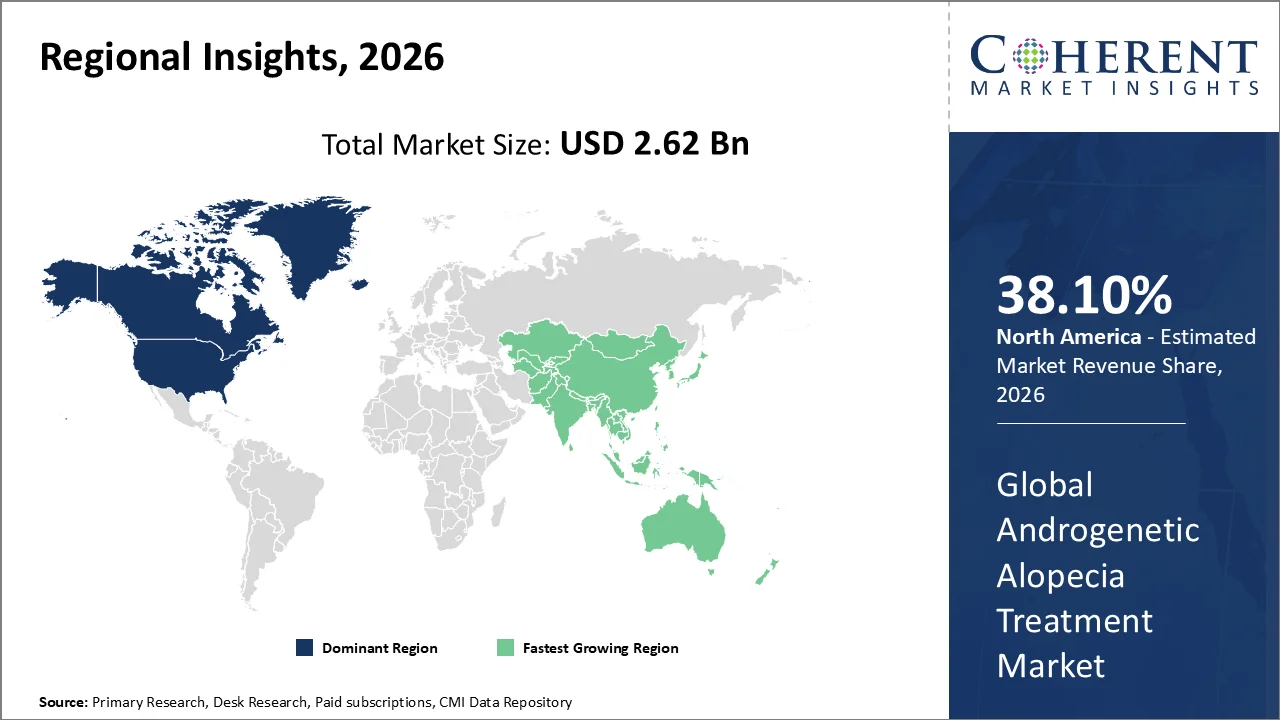
The North America region accounts for 38.10% of the market in 2026. The region’s growth is owing to a high prevalence of hair loss, widespread access to dermatology services, alongside the availability of FDA and Health Canada-regulated treatments. According to the NIH, AGA is one of the most common forms of hair loss in men as well as women, thereby contributing to a large patient base which seeks medical treatment.
In addition, access to dermatology consultations plays a crucial role in treatment uptake. The CDC's National Ambulatory Medical Care Survey reported that 44 million annual visits were made to office-based dermatologists. This focuses the need for professional guidance in androgenetic alopecia management.
Moreover, the regulatory oversight has a considerable impact on treatment choices. The FDA has cautioned against the use of compounded topical finasteride, which is not FDA-approved for treating AGA, thus urging the consumers as well as healthcare providers to prefer medically supervised therapies. This regulatory stance strengthens the demand for FDA-approved and reliable treatments.
Canada’s contribution to the market is also substantial, with Health Canada regulating finasteride 1 mg as the standard for male hair loss treatments. In addition to this, the growing presence of clinical trials like the PP405 trial for regenerative treatments, signals the potential for new therapies in the market. The combination of regulatory backing coupled with cutting-edge research keeps North America at the forefront of AGA treatment.
In March 2026, Xtresse announced that the U.S. Food and Drug Administration (FDA) has accepted its Investigational New Drug (IND) application for Xvie, a regenerative therapy for androgenetic alopecia.
The Asia Pacific region is poised to be as the fastest-growing region through 2026-2033, driven by the increasing prevalence of androgenetic alopecia (AGA) and the growing demand for effective treatments. A large communityThe prevalence increases substantially with age, from 2.8% in men aged 18. This highlights a clear age‑related disease escalation.
AGA prevalence in Asia is generally lower than in Caucasian populations, but it rises steadily with age and family history. This regional epidemiological trend underscores the growing burden of AGA, particularly among younger adults, thereby contributing to the increased healthcare consultations as well as a higher demand for dermatological services.
In terms of clinical management, minoxidil and finasteride is still the dominant treatment options in the region, with both topical as well as oral therapies widely adopted in dermatology practices. The emerging preferences for more advanced treatments like low‑level laser therapy (LLLT) and follicular unit extraction (FUE), also reflect the rising patient interest in combination pharmacologic and device-based strategies for managing AGA.
The U.S. Androgenetic Alopecia Treatment Market is gaining demand as patients shift toward clinically validated, FDA-regulated options rather than informal compounded hair-loss formulations. The data from NIH’s Endotext notes that male androgenetic alopecia affects 30–50% of men by age 50, while topical minoxidil and oral finasteride remain the only FDA-approved treatments for male AGA, requiring continual use and often 6–12 months to show response. This creates sustained demand for repeat-use topical as well as oral therapies in dermatology clinics, online care models, and pharmacy channels. The market is also being shaped by stronger safety scrutiny. For example, in April 2025, the FDA issued a warning stating that there is no FDA-approved topical finasteride and also highlighted 32 reports of adverse events associated to compounded topical finasteride from 2019 to 2024. Meanwhile, ClinicalTrials.gov-listed VDPHL01 studies indicate the rising innovation in oral AGA treatments for both male and female patients.
In January 2025, Eirion Therapeutics Inc., a biopharmaceutical company dedicated to developing innovative aesthetic prescription products, announced the successful results of its first-in-man clinical trial for the treatment of androgenic alopecia (age-related hair loss) using its topical pharmaceutical, ET-02.
China’s Androgenetic Alopecia Treatment Market is experiencing growth, driven by an aging population and an increasing prevalence of AGA. A population-based study found that AGA prevalence in men rose from 2.8% in those aged 18–29 to 41.4% in those aged 70+. It also notes that women show a similar trend, increasing from 1.3% to 11.8% as age progresses. According to the National Bureau of Statistics, China’s elderly population reached 323.38 million in 2025, thus representing 23% of the total population. These demographic trends, coupled with rising consultations for hair loss treatments, are fueling demand for both topical as well as oral AGA therapies. Additionally, recent clinical trials have shown increasing use of advanced treatments, such as minoxidil, finasteride, and innovative laser therapies, further propelling market growth.
Some of the major key players in Androgenetic Alopecia Treatment are Histogen Inc., Cipla Limited, Aclaris Therapeutics, Inc., Merck & Co., Inc., Daiichi-Sankyo Co., Ltd., Johnson and Johnson Services, Inc., Lexington International LLC, Vita-Cos-Med Klett-Loch GmbH, PureTech, Vitabiotics, Dr. Reddy’s Laboratories, HCell Inc., Follica, Inc. and Ranbaxy Laboratories Ltd.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 2.62 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 8.4% | 2033 Value Projection: | USD 4.5 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Histogen Inc., Cipla Limited, Aclaris Therapeutics, Inc., Merck & Co., Inc., Daiichi-Sankyo Co., Ltd., Johnson and Johnson Services, Inc., Lexington International LLC, Vita-Cos-Med Klett-Loch GmbH, PureTech, Vitabiotics, Dr. Reddy’s Laboratories, HCell Inc., Follica, Inc. and Ranbaxy Laboratories Ltd. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients