Apheresis Equipment Market size is estimated to be valued at USD 5.30 Bn in 2026 and is expected to reach USD 11.29 Bn in 2033, exhibiting a compound annual growth rate (CAGR) of 11.4% from 2026 to 2033.
Apheresis is a medical technology that involves separating different components of blood ex vivo for different therapeutic purposes. The main products in the apheresis equipment industry are apheresis machines and disposable apheresis kits. Apheresis machines are advanced automated blood component separators that separate desired components from whole blood based on centrifugal force or membrane filtration. The two major types of apheresis machines are centrifugation equipment and membrane separation equipment.
Centrifugation equipment uses centrifugal force to separate components based on their density, while membrane separation uses a semi permeable filter to separate components based on molecular weight. Recent data show that strong investments in therapeutic plasma exchange services across the U.S. with all new Adia Med clinics now including TPE are boosting regional device procurement, reflecting rising apheresis equipment market demand.
|
Current Event |
Description and its Impact |
|
Economic Trends |
|
|
Public Health and Epidemiological Events |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
In the United States, reimbursement for apheresis (such as therapeutic plasma exchange or red-cell exchange) is complex and evolving, with several key policy dynamics shaping access:
AABB, America’s Blood Centers, and the AABB have urged the Centers for Medicare & Medicaid Services (CMS) to establish a dedicated labor code for therapeutic apheresis nurses, stating current codes under-reimburse for the clinical complexity and cost involved in procedures like TPE, photopheresis, and immunoadsorption.
In September 2024, AABB expressed support for CMS updates that better reflect the resource intensity of apheresis used in CAR‑T and other cell therapies, indicating potential coverage improvements from 2025 onward.
In terms of equipment type, the disposable apheresis kits segment estimated to contribute 64.0% share of the market in 2026, due to their critical role in ensuring safety, efficiency, and hygiene in blood component separation procedures. Single-use kits eliminate the risk of cross-contamination and bloodborne infections, which complies with strict hospital hygiene protocols and regulatory standards. Blood banks and donation centers prefer disposable kits for plasma, platelets, and RBC separation due to their convenience and safety. This is particularly important in developing countries where infrastructure for sterilization is limited.
In January 2024, Inspira Technologies announced its plan to introduce a single-use, disposable blood oxygenation kit designed for cardiopulmonary bypass and ECMO procedures. The kit, tailored for the company’s FDA-cleared INSPIRA ART100 system, includes integrated performance monitoring to alert users of functional changes during use.
To learn more about this report, Request Free Sample
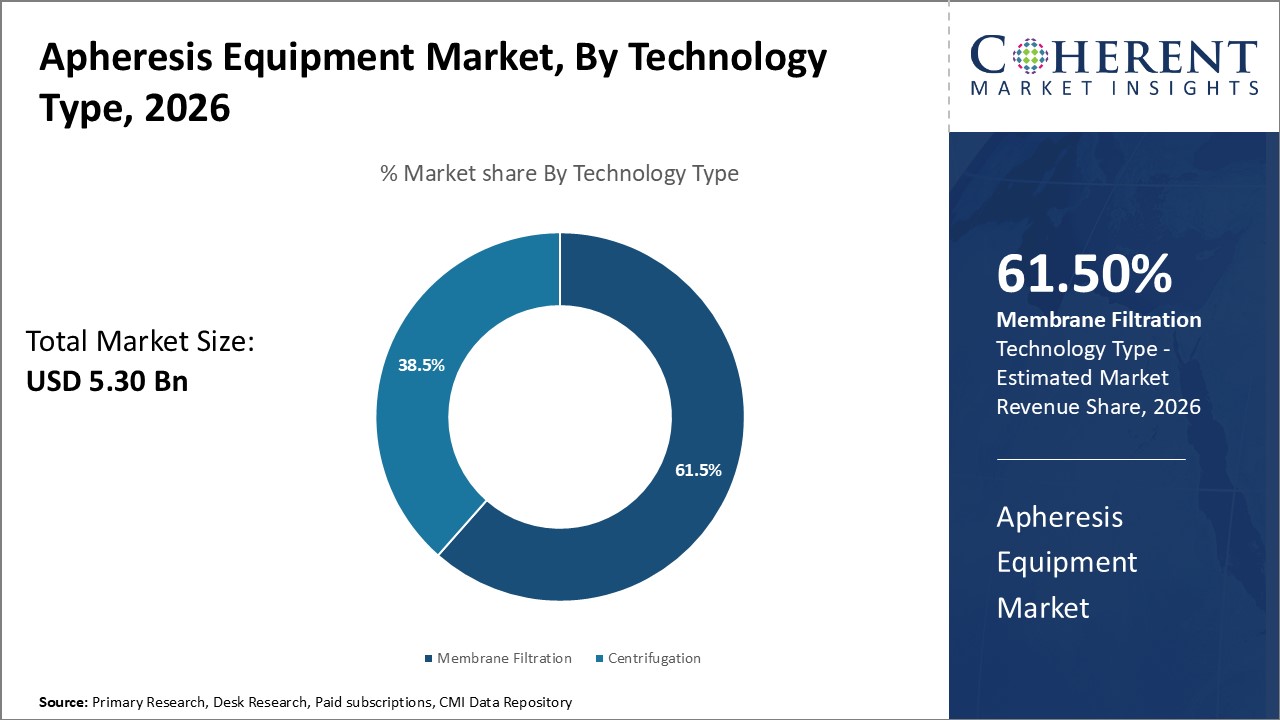
In terms of technology type, the membrane filtration segment is expected to hold the highest marker share in 2026. The membrane filtration segment is driven by its ability to offer superior performance in complex separations. Membrane filtration excels at reducing cell trauma, complement activation, and membrane fouling, providing safer and more precise separations during procedures like plasmapheresis and leukapheresis. The adoption of membrane filtration system has favored healthcare systems including Western Europe, Japan, and North America because of their scalability, safety, and compatibility with regenerative medicine applications.
For instance, Scientists at the Indian Institute of Information Technology, Allahabad (IIIT‑A) have developed an innovative, membrane‑based micro‑filtration device capable of separating plasma from whole blood within minutes. Utilizing hydrodynamic micro‑channels rather than conventional replaceable filters, the patented system swiftly isolates red and white blood cells, platelets, and plasma at approximately one drop per second.
In terms of procedure type, the plasmapheresis segment is expected to hold the highest share of the market in 2026, largely driven by wide clinical applications, and innovative device performance. Plasmapheresis, also known as therapeutic plasma exchange (TPE), which is extensively used to treat various conditions, including autoimmune and neurological disorders such as Guillain–Barré Syndrome, myasthenia gravis, and thrombotic thrombocytopenic purpura, making it the most performed apheresis procedure. According to Red Cross Blood Donation, every 2 seconds someone in the U.S. needs blood and or platelets. Approximately 29,000 units of red blood cells are needed every day in the U. S. Nearly 5,000 units of platelets and 6,500 units of plasma are needed daily in the U.S.
For instance, Mandaya Puri Hospital in Indonesia has become the country’s first private hospital to offer plasma exchange (plasmapheresis) for patients with advanced multiple myeloma and autoimmune diseases such as myasthenia gravis. The procedure separates and replaces patients’ blood plasma to remove harmful antibodies and proteins, relieving severe symptoms, including kidney complications and respiratory challenges. This is further propelling the apheresis market share.
In terms of application, the hematological disorders segment is set to dominate the market with the highest share of the market in 2026, driven by rising prevalence of blood disease. Apheresis devices are extensively used for treating blood group disorders such as sickle cell disease, thalassemia, leukemia, lymphoma, and platelet disorders, where frequent blood processing or component removal is required. Apheresis equipment is used in the treatment of blood disease such as hemophilia A, sickle cell, and various hematological malignancies are on the rise globally, fuelling increased demand for leukapheresis and plateletpheresis in both clinical and transfusion settings.
In August 2024, a dedicated haematology lab with an apheresis machine launched at the district hospital of Pandit Ram Sumer Shukla Government Medical College, Rudrapur, US Nagar.
In terms of end user, the hospitals and clinics segment are expected to contribute the largest shares of the market in 2026, due to their comprehensive procedural capabilities, high patient throughput, and integrated infrastructure. Hospitals and clinics are equipped for a wide range of apheresis procedures including plasma exchange, platelet collection, leukapheresis for autoimmune, hematological, neurological, and other disorders. With escalating rates of chronic diseases such as sickle cell, leukemia, Guillain-Barré syndrome, hospitals register high volume of apheresis procedures, driving strong demand for equipment.
In June 2025, UK hospitals expanded access to automated red blood cell exchange (RCX) machines to tackle long-standing inequalities in sickle cell disease treatment. Now, several NHS hospitals including Manchester Royal Infirmary and Homerton Hospital, no longer rely solely on manual emergency exchanges, enabling rapid, 24/7 exchange therapy close to patients' homes. This investment reduces critical care admissions, eases hospital pressure, and delivers lifesaving care more equitably.
To learn more about this report, Request Free Sample
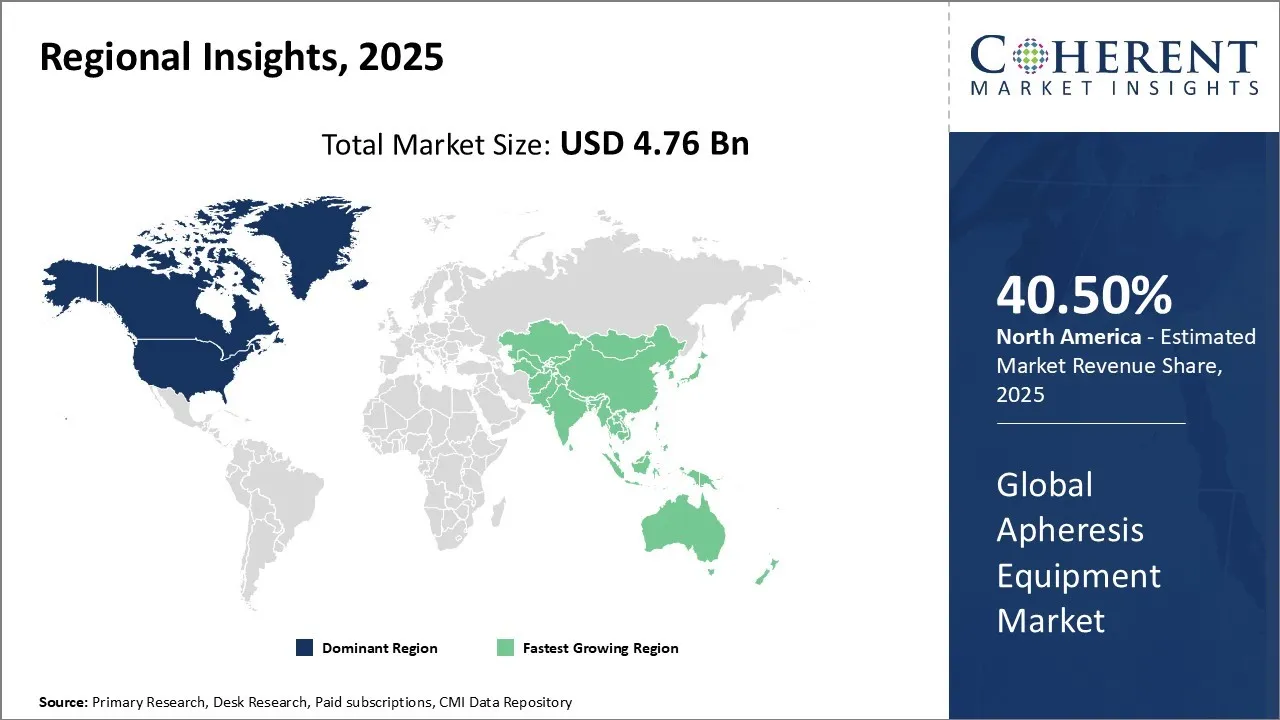
North America is expected to be the largest market for apheresis equipment during the forecast period, accounting for over 40.50% of the market share in 2026. North America has dominated the global apheresis equipment market for many years due to a strong presence of leading players in the region. The U.S. is home to many large multinational companies, such as Haemonetics Corporation and Terumo BCT, which have a significant market share in blood component collection and therapeutic apheresis products globally. Additionally, North America has a higher healthcare spend per capita as compared to other regions, enabling faster adoption of new and advanced apheresis technologies.
Asia Pacific is expected to be the fastest-growing market for apheresis equipment, which is expected to grow at a CAGR of over 10.2% during the forecast period. The growth of the market in Asia Pacific is attributed to the large patient pool, increasing healthcare expenditure, and improving healthcare infrastructure in the region. Asia Pacific region has rising rate of blood-related disease in countries like China and India face growing incidences of leukemia, thalassemia, and other hematologic disorders, fueling needs for therapeutic apheresis and component collection. Asia pacific region is expanding its healthcare infrastructure and government support by improving hospital networks, especially in tier‑2/3 cities, and proactive government investment such as state-sponsored plasma donation drives in China resulting in enhancing access to apheresis.
For instance, in January 2023, Australian Red Cross Lifeblood issued a nationwide appeal for plasma donors, responding to record-high demand and ongoing shortages in blood and plasma supplies. Plasma donations now exceed whole blood in usage, doubling as the primary life-saving treatment for conditions like cancer, immune disorders, and trauma. The organization aims to recruit 100,000 new donors within a year and deploys awareness campaigns—including partnerships with sports teams—to raise visibility. This is further propelling the apheresis market share.
The United States continues to acquire the prominent share in global demand of the apheresis equipment, fueled by rising cases of leukemia, autoimmune disorders, and therapeutic plasma exchange (TPE). According to Leukemia & Lymphoma Society, an estimated combined total of 187,740 people in the US are expected to be diagnosed with leukemia, lymphoma or myeloma in 2024. Hospitals and research institutions are rapidly adopting automated and AI-powered apheresis systems, particularly the popular Spectra Optia platform, which supports CAR-T cell collections and plasma therapy trials. For instance, Adia Med recently announced that all upcoming clinics across the U.S. will offer TPE services, focusing on advanced conditions such as Alzheimer’s and autoimmune diseases. This reflects strong infrastructure for blood donation, cell therapy, and cutting-edge clinical research making the U.S. the largest and fastest adopter of next-generation apheresis technology.
China demonstrates remarkable momentum through increased focus on plasma-derived therapies, prompting expansion of automated apheresis systems to supply immunoglobulins, albumin, coagulation factors, and more. With the support of Chinese government and improved collection standards, the market is growing under the Healthy China 2030 strategy, China has strengthened regulations and funded infrastructure for plasma collection, boosting domestic production capacity and reducing imports of plasma-derived therapies.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 5.30 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 11.4% | 2033 Value Projection: | USD 11.29 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Asahi Kasei Medical Co., Ltd., Terumo Corporation, Inc., Fenwal, Inc., Haemonetics Corp., Therakos, Inc., B. Braun Melsungen AG, HemaCare Corporation, Haemonetics Corporation, Cerus Corporation, Kawasumi Laboratories, Miltenyi Biotec, NxStage Medical, and Kaneka Medix Corporation |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The apheresis equipment market is undergoing a pivotal transformation is one that reflects a broader shift toward precision-driven, resource-efficient, and patient-centric blood component management. The momentum is not only sustained by clinical demand, but increasingly by the convergence of reimbursement reforms, digital integration, and biologics pipeline expansion, particularly in high-yield geographies such as the Asia-Pacific and Eastern Europe.
Moreover, the traditional use of apheresis in therapeutic plasma exchange is being rapidly outpaced by its integration into the collection of cell-based therapies and plasma-derived biologics. Consider this: in the U.S. alone, plasma collection centers have more than doubled over the last five years, reaching over 1,000 sites as of 2024. These centers are heavily reliant on automated plasmapheresis systems. Manufacturers that fail to integrate features like customizable protocols and donor-centric interfaces are at risk of being bypassed in procurement processes dominated by vertically integrated biologics producers.
Apart from this, regulatory alignment in markets such as China and India has sharply reduced adoption friction. For example, India’s National Blood Policy has incentivized public-private partnerships to install apheresis devices in Tier-II hospitals for the treatment of conditions like sickle cell anemia and dengue hemorrhagic fever. These diseases are not episodic in nature; they represent a recurring demand pool for plateletpheresis and leukapheresis systems. The implications are clear: manufacturers must pivot product portfolios toward mobile, modular, and scalable equipment suitable for decentralized settings.
Besides this, the North American and Western European markets are no longer solely driven by hardware innovation. Instead, the value is shifting toward interoperability with hospital information systems (HIS) and real-time patient monitoring. Haemonetics’ NexSys PCS system, for example, now integrates biometric screening and AI-based donor health assessments, setting a new standard for what is expected in both fixed and mobile setups. Competitive differentiation is now less about centrifuge speed and more about machine learning models that predict adverse donor events or optimize anticoagulant ratios on-the-fly.
Share
Share
Manisha Vibhute is a consultant with over 5 years of experience in market research and consulting. With a strong understanding of market dynamics, Manisha assists clients in developing effective market access strategies. She helps medical device companies navigate pricing, reimbursement, and regulatory pathways to ensure successful product launches.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients