Asia Gout Disease Treatment Market is estimated to be valued at USD 1,180.6 Mn in 2026 and is expected to reach USD 2,214.3 Mn in 2033, exhibiting a compound annual growth rate (CAGR) of 9.4% from 2026 to 2033.
Analysts’ Views on Asia Gout Disease Treatment Market:
The gout therapeutics market is primarily driven by the increasing prevalence of this condition due to changing lifestyle patterns and the growing geriatric population. Globally, the life expectancy of people is increasing, which has led to a rise in the geriatric population. This trend has led to a higher incidence of this condition and per capita demand for its therapeutics, thus positively affecting the market. As a result of the increased incidence of gout and the link between alcohol intake and gout, higher alcohol intake is likely to fuel the expansion of the gout therapeutics market over the projected period. Major pharmaceutical companies are investing in the development of Asia Gout Disease Treatment to sustain its position in the market.
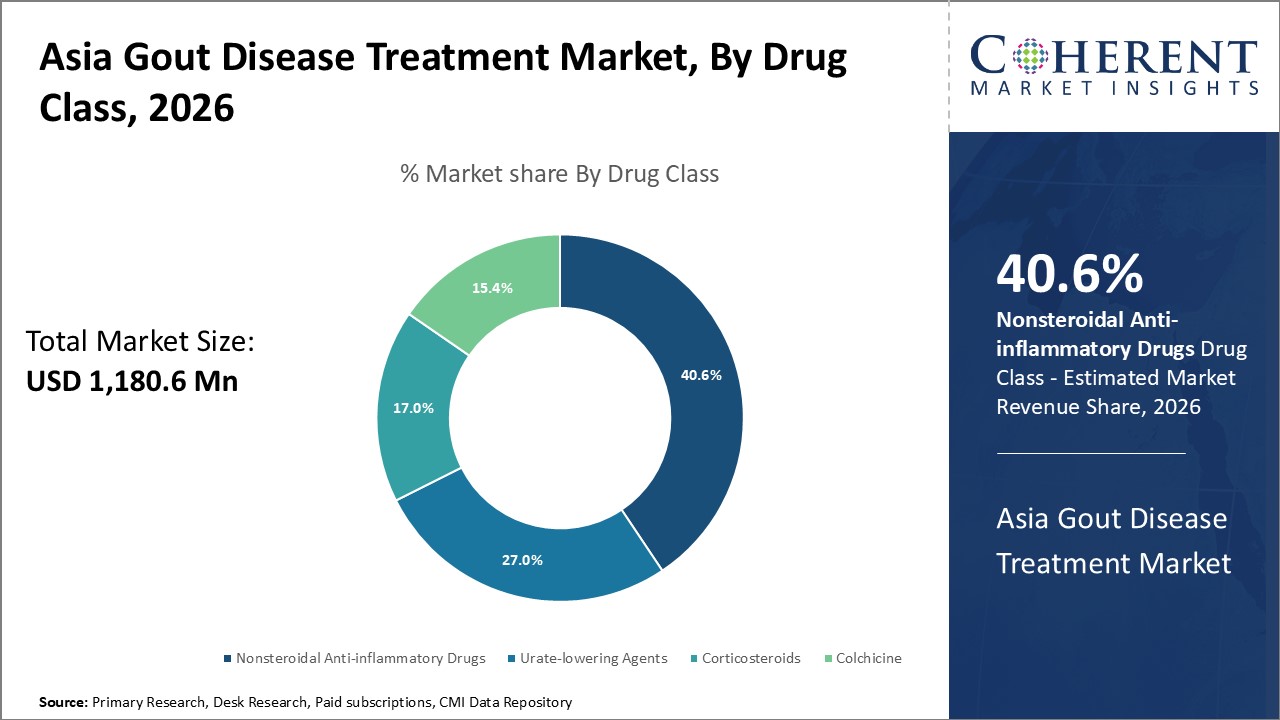
Figure 1. Asia Gout Disease Treatment Market Share (%), By Drug Class, 2026
To learn more about this report, Request Free Sample
Asia Gout Disease Treatment Market– Driver
Rising incidence of gout diseases
Rising incidence of gout diseases is expected to drive the market growth over the forecast period. For instance, according to an article published in the National Library Of Medicine, in June 2020, reported the prevalence and incidence of gout vary widely according to the population studied and methods employed but range from a prevalence of <1% to 6.8% and an incidence of 0.58-2.89 per 1,000 person-years. Gout is more prevalent in men than in women, with increasing age, and in some ethnic groups.
Adoption of Inorganic Strategies by Key Players
Adoption of inorganic strategies by key players can also drive the gout disease treatment market growth. For instance, Market players are more focused on adoption of inorganic growth strategies such as acquisitions, collaborations, and partnerships to strengthen its anti-gout drugs portfolio. For instance, in September 2020, Simcere Pharmaceutical Group, a manufacturer and supplier of generic pharmaceuticals in the China market, and JW Pharmaceutical , a pharmaceutical companies in Korea, entered into a collaboration and exclusive license agreement for the development of anti-gout drug candidate Uniform Resource Characteristics (URC-102) in China. Under this agreement, the responsibilities of clinical trial and registration will be taken by Simcere Pharmaceutical Group. The candidate, URC-102, is a selective inhibitor of uric acid transporter (URAT-1) and it is in Phase IIb clinical trial in patients with gout disease in South Korea.
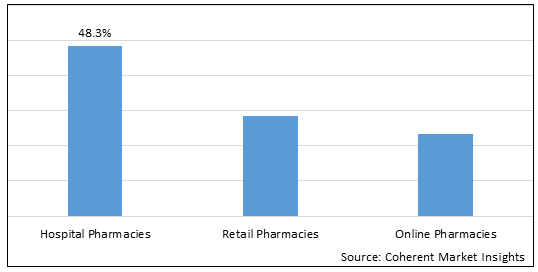
Figure 2. Asia Gout Disease Treatment Market Share (%), By Distribution Channel, 2026
To learn more about this report, Request Free Sample
Asia Gout Disease Treatment Market- Country Analysis
Among Countries, China is estimated to hold a dominant position in the asia gout disease treatment market over the forecast period. China is estimated to hold 38.3% of the market share China holds a significant market share in the gout disease treatment market, mainly due to the key market players are focusing on inorganic stratergies such as Acquisition and increased manufacturing of drugs. For instance, in April 2020, Takeda Pharmaceutical, a patient-focused, R&D-driven global biopharmaceutical company in Japan, acquired the URL Pharma Inc a pharmaceutical company with fully integrated technology development, product development, manufacturing in china a Philadelphia-based company, in order to expand its gout disease treatment portfolio with the acquisition of marketed gout therapy Colcrys (colchicine), developed by URL Pharma Inc.
Asia Gout Disease Treatment Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1,180.6 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 9.4% | 2033 Value Projection: | USD 2,214.3 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
AstraZeneca Plc, Takeda Pharmaceutical Company Limited, Teijin Pharma Limited, Hikma Pharmaceuticals Plc., Zyla Life Sciences, Horizon Therapeutics plc, JW Pharmaceutical CORPORATION, Abbott Diagnostics (Abbott Laboratories), Labcorp ,Antibodies Inc., Networks LLC, Beckman Coulter, Inc., Euro Diagnostica AB, F. Hoffmann-La Roche Ltd., Qiagen NV, Siemens Healthcare GmbH, and Augurex Life Sciences Corp., Thermo Fisher Scientific Inc., Bio Rad Laboratories Inc., Exagen Inc., Genway Biotech, Inc., Microdrop LLC (imaware), Svar Life Science and LG Chem. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Asia Gout Disease Treatment Market Segmentation:
The Asia Gout Disease Treatment market report is segmented into By Drug Class, By Route of Administration,By Distribution Channel, and By Country.
By Drug Class, the Asia gout disease treatment market is segmented into Nonsteroidal anti-inflammatory drugs (NSAIDS), corticosteroids, colchicine, and urate-lowering agents. Out of which, the Nonsteroidal anti-inflammatory drugs (NSAIDS), segment is expected to dominate the gout disease Treatment market during the forecast period and this is due to the high prevelance of gout disease.
By Route of Administration, the Asia gout disease treatment market is segmented into Oral and Injectable. Out of which, the Injectable segment is expected to dominate the Asia gout disease treatment market during the forecast period and this is attributed to the fast onset of action in injectable.
By Distribution Channel, the Asia gout disease treatment market is segmented into hospital pharmacies, retail pharmacies, and online pharmacies. Out of which, the hospital pharmacies segment is expected to dominate the Asia gout disease treatment market during the forecast period and this is attributed to the providing disease information and adequate statistics on medication consumptions.
Among all the segmentations, the Drug class has the potential due to the increasing use of onsteroidal Anti-inflammatory Drugs this is expected to drive the growth of the segment over the forecast period. For instance, in September 2021, Heron Therapeutics, Inc., a commercial-stage biotechnology company focused on improving the lives of patients by developing best-in-class treatments to address some of the most important unmet patient needs, announced that the U.S. Food and Drug Administration (FDA) has approved ZYNRELEF (bupivacaine and meloxicam) extended-release solution for use in adults for soft tissue or periarticular instillation to produce postsurgical analgesia for up to 72 hours after bunionectomy, open inguinal herniorrhaphy and total knee arthroplasty. ZYNRELEF, the first and only extended-release dual-acting local anesthetic (DALA), delivers a fixed-dose combination of the local anesthetic bupivacaine and a low dose of the nonsteroidal anti-inflammatory drug (NSAID) meloxicam. The synergy between bupivacaine and meloxicam in ZYNRELEF has resulted in patients experiencing significantly less pain, including severe pain, and significantly more patients requiring no opioids (opioid-free) after surgery
Asia Gout Disease Treatment Market: Key Developments
On May 31, 2023, Horizon Therapeutics plc., biotechnology company, announced new analyses from the MIRROR randomized controlled trial of KRYSTEXXA (pegloticase) injection with methotrexate show a multi-faceted impact when treating adults living with chronic gout refractory to oral therapy, or uncontrolled gout.
In December 2022, JW Pharmaceutical, a pharmaceutical companies in Korea, announced through its disclosure on Monday that it had applied last Friday for a phase 3 trial for Epaminurad, a gout treatment, with the Taiwan Food and Drug Authority (TFDA).
In August 2020, Teijin Pharma Limited, a developing business in the field of pharmaceuticals and home healthcare equipments, and Astellas Pharma Inc., a multinational pharmaceutical company in Japan, announced that they have entered into an exclusive distribution agreement for febuxostat, a novel drug for the treatment of gout and hyperuricemia, in Southeast Asia and India.
In April 2022, Specialty Networks, LLC, a portfolio company of Linden Capital Partners is an integrated, multi-specialty organization with GPOs, Life Sciences, and Research solutions in U.S., announced the acquisition of United Rheumatology, LLC is the Rheumatology group purchasing organization (GPO) and real-world evidence (RWE) data & analytics company, community Rheumatology clinics will be able to maintain their independence while also improving the standard of treatment for patients.
In May 2021, the Arthritis Foundation, the nonprofit organization dedicated to the prevention control and cure of America's leading cause of rheumatoid arthritis, announced the launch of a application (Vim, the Arthritis Foundation's mobile app) and kicks off Arthritis Awareness with the release of key findings from a patient study.
In May 2022, United Rheumatology, the rheumatology care management organization empowering rheumatologists to advance the standard of care, and CreakyJoints, a support, education, advocacy, and research organization for people living with arthritis and rheumatic disease, announced a partnership to develop the Rheumatoid Arthritis Wellness Center, a digital resource aimed at aiding those living with rheumatoid arthritis.
In September 2021, Labcorp, a life sciences company based in the U.S., announced that it has entered into a definitive agreement to acquire select operating assets and intellectual property (IP) from Myriad Genetics’ autoimmune business unit, including the Vectra rheumatoid arthritis (RA) assay.
Asia Gout Disease Treatment Market: Key Trends
Increasing product approval by regulatory bodies
Increasing product approval by regulatory bodies is expected to offer lucrative growth opportunities for players in the Asia Gout Disease Treatment market. For instance, in November 2020, The U.S. Food and Drug Administration has approved a new treatment for gout. When used in combination with a xanthine oxidase inhibitor (XOI), Zurampic (lesinurad) helps to reduce the high levels of uric acid (hyperuricemia) that is associated with gout.
Asia Gout Disease Treatment Market: Restraint
The adverse effects and risks associated with drugs such as NSAIDS
The adverse effects and risks associated with drugs such as NSAIDS and urate-lowering agents is major factor restraining growth of this market. Most of the gout drugs are associated with some severe side effects, which may lead to decrease in demand for these drugs. Nonsteroidal anti-inflammatory drugs (NSAIDs) may cause some gastric problems such as indigestion. In some cases, these drugs can cause gastric ulcers, bleeding, and rashes in gastrointestinal GI tract. For instance, in February 2020, the U.S. Food and Drug Administration (FDA) announced to add a new boxed warning for febuxostat, as there is increased risk of death associated with Uloric (febuxostat) compared to another gout medicine, allopurinol. Febuxostat belongs to the urate-lowering agents.
Key market players are focusing on safety and efficacy of drugs.
Asia Gout Disease Treatment Market- Key Players
Major players operating in the Asia Gout Disease Treatment market include AstraZeneca Plc, Takeda Pharmaceutical Company Limited, Teijin Pharma Limited, Hikma Pharmaceuticals Plc., Zyla Life Sciences, Horizon Therapeutics plc, JW Pharmaceutical CORPORATION, and LG Chem.
Asia Gout Disease Treatment Market– Definition
Gout is a chronic disease characterized by pain and inflammation in the joints. It is also known as gout arthritis, in which a high level of uric acid in the blood leads to the formation of needle-like crystals in the joints, resulting in inflammation and pain. This condition can be treated or prevented with the use of drugs such as nonsteroidal anti-inflammatory drugs, corticosteroids, colchicine, and urate-lowering agents. Moreover, monoclonal antibodies are also used to treat gout in certain cases in which, nonsteroidal anti-inflammatory drugs, corticosteroids, colchicine, and urate lowering agents are unresponsive to the patient.
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients