Global brain health devices market is estimated to be valued at USD 17.84 Bn in 2026 and is expected to reach USD 32.20 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 8.8% from 2026 to 2033.
Discover market dynamics shaping the industry: Request Free Sample
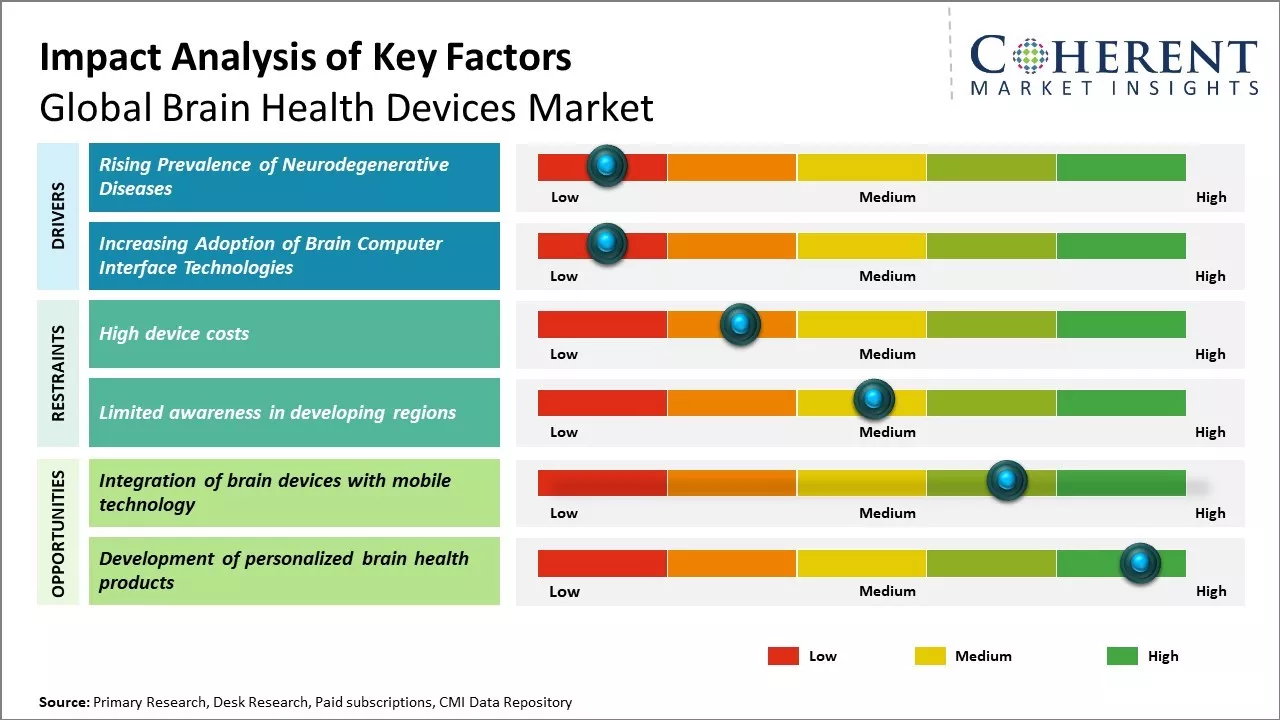
Rising prevalence of neurological diseases globally can drive the market growth. Technological advancements in monitoring devices along with growing aging population can also drive the market growth. Moreover, increasing awareness among people regarding brain fitness can boost adoption of brain monitoring and diagnostics devices. North America is expected to dominate the market due to high awareness levels and developed healthcare infrastructure. However, Asia Pacific is likely to demonstrate highest growth owing to growing healthcare expenditure, rise in income levels and large population base. Key market players are focused on new product launches and geographical expansion to strengthen their market position.
Rising Prevalence of Neurodegenerative Diseases
Rising prevalence of neurological disorders such as Alzheimer's disease, Parkinson's disease, epilepsy, headaches, and traumatic brain injuries can drive the brain health devices market growth. According to the World Health Organization, there are around 50 million dementia patients worldwide with nearly 10 million new cases reported every year. As people age, their risk of developing these life-altering conditions increases exponentially. Alzheimer's disease, the most common form of dementia, has no cure and is largely underdiagnosed. As global population continues to grey at an unprecedented rate, one can expect to see an enormous rise in the number of people living with cognitive impairment and neurological issues. Brain health devices aim to improve lives by assisting with disorders, aiding in diagnoses, and monitoring conditions. Devices that can assist people suffering from memory loss, poor motor control, seizures, or other debilitating symptoms are deeply meaningful for patients and caregivers alike. For physicians, such tools have the potential to support earlier and more accurate detection. While more research and development are still needed, the rapid pace of technological advancement combined with growing demand from an aging population can offer market growth opportunity. Rising neurodegenerative illnesses that impose heavy human and economic burden worldwide can boost demand for devices that can enhance lives or slow decline.
Get actionable strategies to beat competition: Request Free Sample
Increasing Adoption of Brain- Computer Interface Technologies
Rising developments in brain-computer interface (BCI) technologies can drive the brain health devices market growth. Brain-computer interface facilitates a direct communication pathway between the brain and an external device, paving the way for innovative applications that can interpret neural signals and translate them into action. For example, BCIs can help restore lost functions through assistive technologies like thought-controlled prosthetics for paralysis patients. These also show promise for conditions like epilepsy, depression, and other mental health issues that are difficult to treat via conventional methods alone. As the precision and usability of BCI systems advance alongside growing knowledge of neuroscience, the potential for transformative solutions grows exponentially.
Key Takeaways from Analyst:
The global brain health devices market is poised to experience strong growth over the coming years driven by the rising geriatric population who are more prone to neurological disorders. The increasing prevalence of conditions like Alzheimer's, dementia, Parkinson's disease, and traumatic brain injuries is expected to propel the demand for brain health devices for both diagnosis and treatment purposes. Additionally, growing awareness among people about the importance of brain health and early detection of cognitive issues through screening will boost the market expansion.
North America currently dominates the market owing to advanced healthcare infrastructure and frequent introduction of novel technologies. However, Asia Pacific is likely to emerge as the fastest growing region supported by rising healthcare spending, large patient population and increasing research activities in countries like China and India. The high costs associated with brain health devices can negatively impact their adoption in price-sensitive developing nations. Similarly, the lack of skilled professionals required to operate complex brain diagnostic devices poses a challenge.
On the opportunities side, rising R&D investments by leading players for developing innovative products, growing acceptance of brain monitoring devices due to tiny size and high comfort levels, and increasing applications of brain computer interface technology are expected to open new avenues of growth.
Market Challenges: High device costs
High device costs can hamper the global brain health devices market growth. The cost of brain health devices like electroencephalogram (EEG) machines, intracranial pressure monitoring devices, oximeters, and transcranial dopplers devices are quite expensive, ranging from thousands to tens of thousands of dollars. This makes these devices unaffordable for many people globally especially in developing nations. The high cost is attributed to various factors like R&D involved in developing sophisticated brain monitoring technology is extensive that increases cost of device. Manufacturing complex devices requires high-precision engineering and expensive components like sensors and processors, which inflate prices. Commercial producers aim to recover their investments and earn profits, which further increases costs. Thus, many people facing financial constraints simply cannot afford these life-saving and crucial devices even if prescribed by doctors. When devices are unaffordable, it limits the number of potential customers significantly.
Market Opportunities: Integration of brain devices with mobile technology
The integration of brain devices with mobile technology can offer opportunities in the global brain health devices market. As more people adopt smartphones and other mobile gadgets, combining these with wearable brain devices allows for convenient real-time monitoring of brain activity. This move towards increased mobility and flexibility affords wider access to brain health solutions. By linking brain monitoring devices to mobile apps, users get a round-the-clock view of their brain function from the convenience of their phones. Data collected on brain signals, emotions, attention span, and others over extended periods while people go about their daily lives offers deeper insights compared to isolated clinical sessions. The constant feedback helps users track patterns and progress of conditions like Alzheimer's, depression, epilepsy. It also enables prompt medical interventions if any risks are detected.
This type of continuous monitoring ensures better management of brain health. Powerful yet affordable computing in mobile devices allows for sophisticated data analysis of brain activities. Advances in miniaturization enables development of small, discreet and wireless brain-monitoring wearables compatible with mobile phones. As 5G networks expand globally, there will be exponentially more data that can be gathered and shared with doctors and care teams in real-time for evaluation. In 2022, according to a report published by The World Health Organization, dementia cases are estimated to triple by 2050, leading to huge demand for such mobile-enabled solutions to facilitate remote care and early detection.
Discover high revenue pocket segments and roadmap to it: Request Free Sample
By Device Type- Precision Imaging for Improved Diagnosis and Treatment Planning
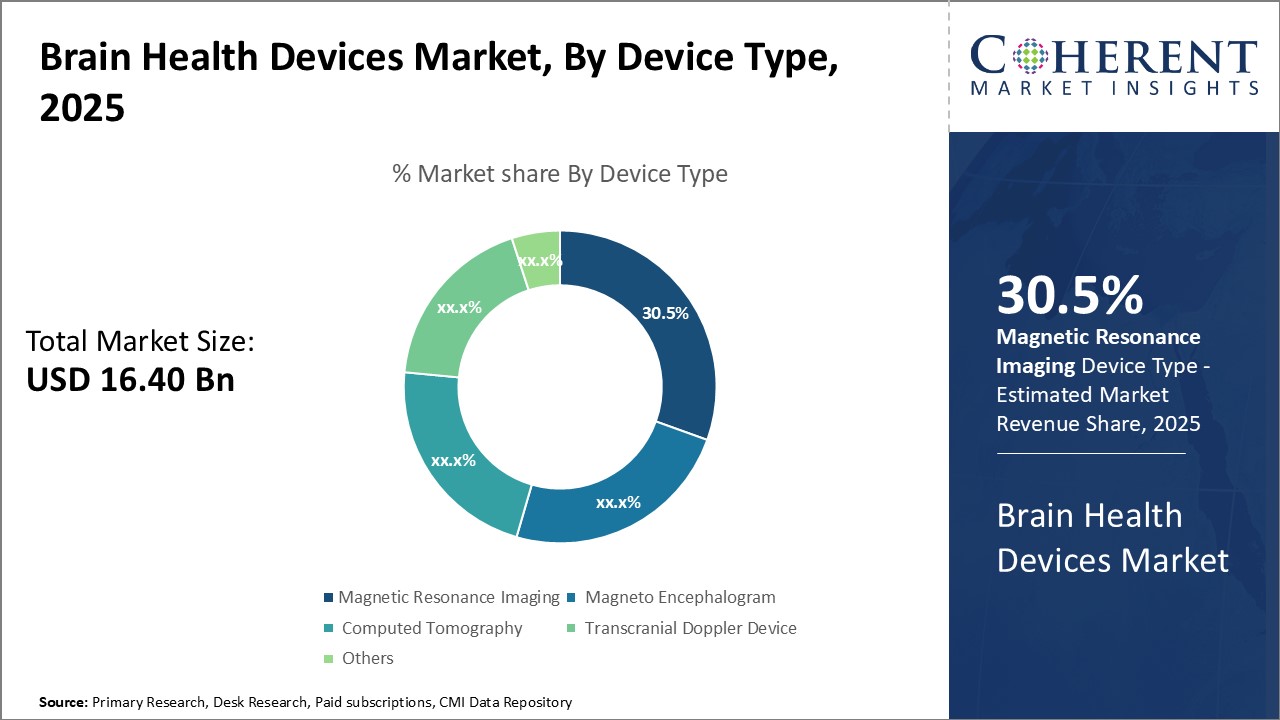
In terms of device type, magnetic resonance imaging segment is estimated to contribute the highest market share of 31.5% in 2026, owing to its precision in detecting even the subtlest changes in the brain. MRI utilizes powerful magnetic fields and radiofrequency pulses to generate detailed images of the brain and detects abnormalities that may indicate conditions like tumors, stroke, infections, demyelinating diseases and other issues. Its non-invasive nature makes it the preferred diagnostic test for evaluating the brain. MRI provides superior soft tissue contrast as compared to other modalities like CT or ultrasound. This allows physicians to differentiate between grey matter, white matter and cerebrospinal fluid with great clarity. Detection of lesions and abnormalities is more reliable on MRI images. Moreover, advanced MRI techniques like functional MRI, diffusion tensor imaging and perfusion imaging provide physiological and molecular details that aid improved diagnosis, prognosis evaluation and treatment planning. For example, MRI helps map brain activity patterns which is useful for pre-surgical planning in epilepsy patients. The ability of MRI to detect even subtle changes serially allows close monitoring of disease progression or response to therapy over time. This makes it valuable for detecting diseases like multiple sclerosis, tumors, and others. Advanced applications like diffusion tensor imaging and functional MRI are used in diagnostic evaluation of conditions like Alzheimer's, Parkinson's, traumatic brain injuries and strokes. MRI's high sensitivity and specificity along with continual technological advances cement its position as the most widely used and preferred imaging modality for evaluation of the brain.
To learn more about this report, Request Free Sample
By Application - Stroke Management Remains Crucial
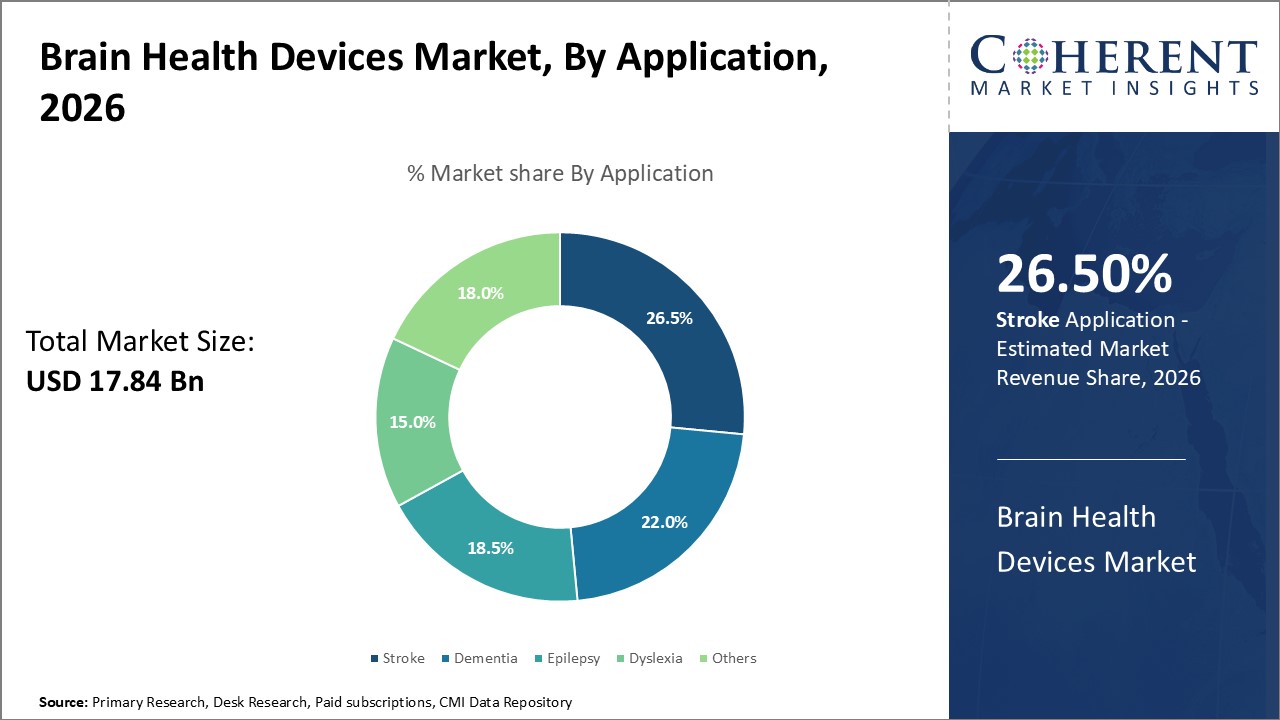
In terms of application, stroke segment is estimated to contribute the highest market share of 26.5% in 2026, due to its high global incidence and disease burden. Stroke occurs when blood supply to part of the brain is disrupted, depriving brain cells of oxygen and nutrients. This can lead to cell death and impaired functioning. Globally, stroke is among the top five causes of death and a leading cause of long-term disability. Timely diagnosis and management of stroke is critical to mitigate disability and save lives. Brain health devices play a key role by enabling early detection of stroke, determining severity, evaluating risks, and guiding acute treatment decisions. Computed tomography (CT) and magnetic resonance imaging (MRI) provide precise evaluation of the location and size of injury which determines the appropriate course of management - whether thrombolytic drugs can be administered or neurointerventional procedures need to be employed. Follow up scans track the extent of recovery or identify complications which help optimize post-stroke rehabilitation management. Monitoring blood flow and vascular functionality through transcranial doppler devices gives important information to guide long term secondary stroke prevention as well. With rising burden of lifestyle disease, there has been increase in numbers of strokes and associated disabilities. This boosts demand for advanced brain health technologies that can help streamline stroke care across the continuum from emergency response to long term care.
By End User - Expanding Role in Hospitals and Specialty Clinics
In terms of end user, hospitals segment is estimated to contribute the highest market share of 41.5% in 2026, owing to their expanding role in providing advanced brain health services. Hospitals are typically the primary contact point and preferred locations for emergency and inpatient brain care. These are equipped with specialized stroke units, epilepsy monitoring units, neurology/neurosurgery departments and intensive care facilities essential for managing critical neurological conditions. Availability of high-end brain diagnostics and dedicated neuroscience staff enables comprehensive evaluation and multidisciplinary management. Growing demand for specialized brain care has also led many hospitals to establish dedicated neuroscience institutes offering advanced surgical/non-surgical therapies, research and global patient referrals. Integration of advanced brain health devices within digital hospital ecosystems further improves the efficiency, continuity and quality of care. Their expertise and personalized approach enhance clinical outcomes and quality of life.
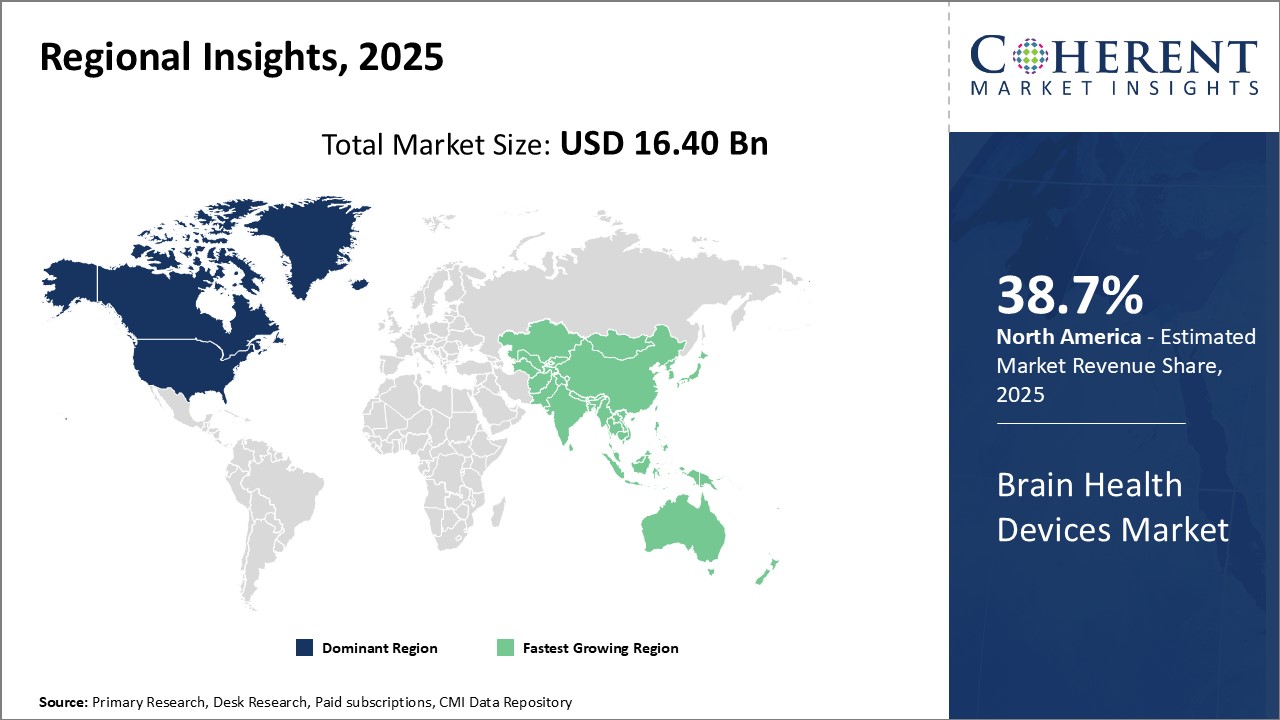
Need a Different Region or Segment? Request Free Sample
North America dominates the global brain health devices market with an estimated market share of 40.2 % in 2026. The U.S. accounts for the major share primarily due to strong healthcare infrastructure, high awareness about brain health issues, and rapid adoption of advanced technologies. The region is home to several leading global companies that are constantly investing in R&D to develop new and innovative products. Supportive regulatory guidelines have ensured speedy clearance of devices, allowing patients faster access.
Asia Pacific region is poised to witness the fastest growth in the near future. Factors such as rising geriatric population, improving healthcare infrastructure and increasing per capita spending on healthcare can drive the market growth. Countries like China, Japan and India represent major opportunities due to their huge population base and growing medical tourism industries. Local players are also entering the market and offering cost-effective products. This has made brain health devices more accessible to customers.
Governments of various APAC countries have initiated several favorable regulations to encourage domestic manufacturing and promote adoption of new technologies. For instance, the 'Make in India' program focuses on making India a global hub for medical devices. It offers several fiscal and non-fiscal incentives for companies to set up manufacturing units. China has removed regulatory barriers and made the approval process more streamlined to attract foreign investments. These initiatives are positively impacting the import and export of brain health devices within the region.
Brain Health Devices Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 17.84 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 8.8% | 2033 Value Projection: | USD 32.20 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
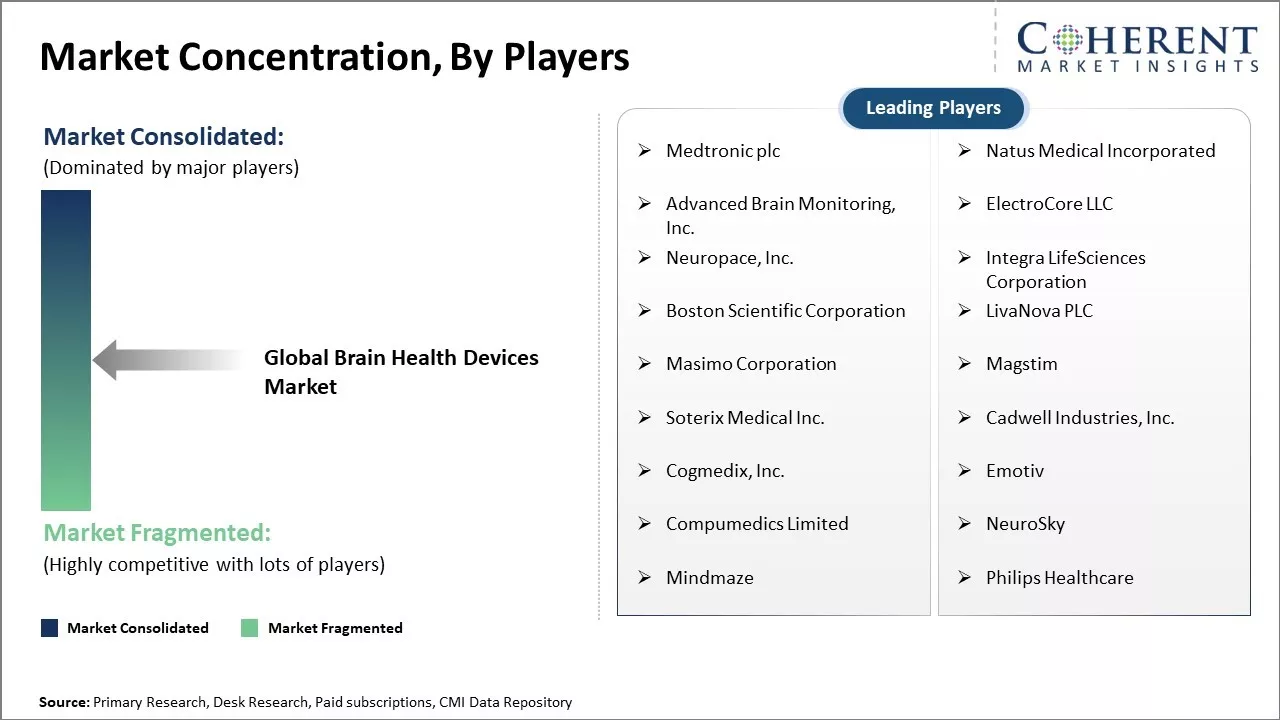
Medtronic plc, Natus Medical Incorporated, Advanced Brain Monitoring, Inc., ElectroCore LLC, Neuropace, Inc., Integra LifeSciences Corporation, Boston Scientific Corporation, LivaNova PLC, Masimo Corporation, Magstim, Soterix Medical Inc., Cadwell Industries, Inc., Cogmedix, Inc., Emotiv, Compumedics Limited, NeuroSky, Mindmaze, Philips Healthcare |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
*Definition: Global Brain Health Devices Market refers to the market for medical devices and technologies that help diagnose, monitor and treat various brain health conditions and disorders. This includes devices such as EEG machines, brain stimulators, neuroimaging devices and other wearable brain monitoring systems that help detect and manage neurological diseases like Alzheimer's, dementia, epilepsy, migraines and other brain related illnesses. The market driven by the rising aging population and neurological disease burden worldwide.
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients