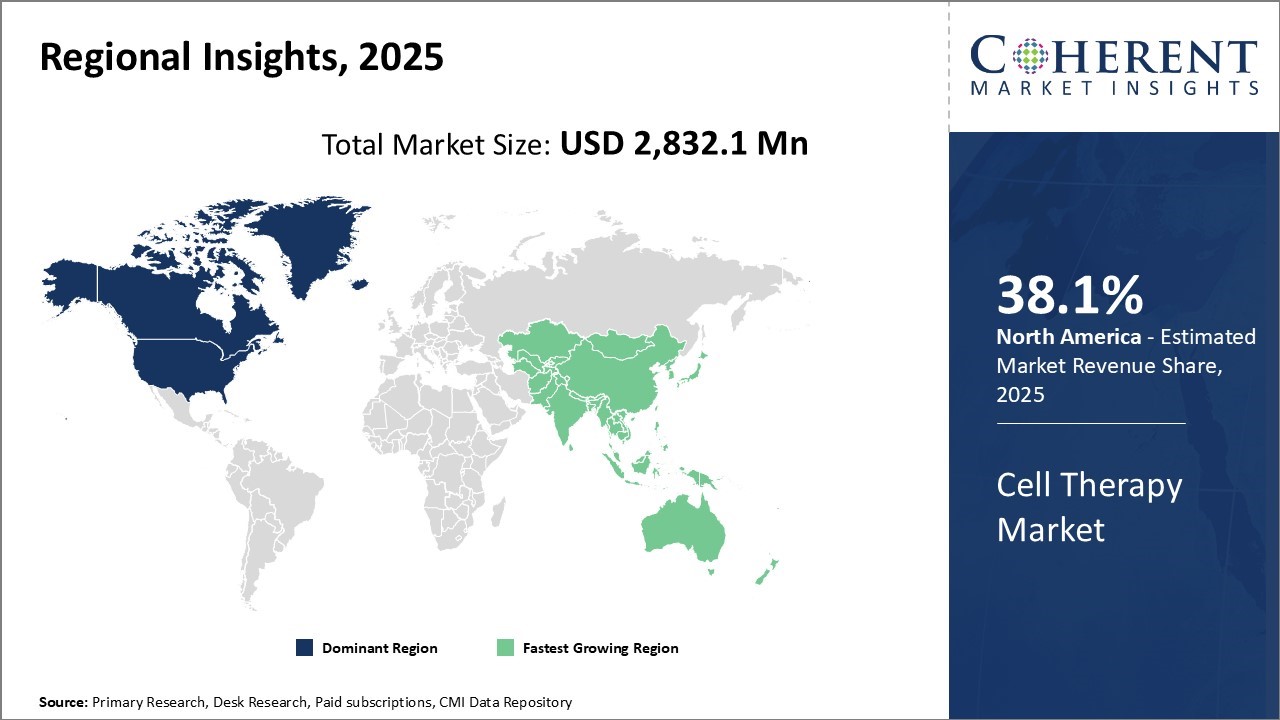
Global cell therapy market is estimated to be valued at USD 2,832.1 Mn in 2025 and is expected to reach USD 5,383.4 Mn by 2032, exhibiting a compound annual growth rate (CAGR) of 9.6% from 2025 to 2032.
The global cell therapy market is rapidly expanding as advancements in regenerative medicine, immunotherapy, and genetic engineering drive innovation. Researchers and companies are developing therapies such as CAR-T cells, stem cells, and TCR therapies to treat oncology, rare diseases, and autoimmune disorders. Rising cases of chronic and genetic diseases, along with supportive regulatory frameworks and strong clinical pipelines, are boosting demand. Hospitals and specialized treatment centers are building infrastructure to administer these therapies, while technological innovations and strategic collaborations are improving accessibility and commercialization worldwide.
|
Current Events |
Description and its impact |
|
Regulatory Developments in Key Regions |
|
|
Technological Advancements and Innovations |
|
|
Investment and Funding Landscape |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
T-cells hold the largest market share of 73.9% in 2025. T-cells or T lymphocytes form an essential part of the adaptive immune system, and are capable of recognizing infected or cancerous cells. Research in recent years has shown the potential of engineered T-cells, known as CAR T-cell therapy, to identify and destroy cancer cells that traditional drugs are unable to eliminate. Pioneering drugs such as Kymriah and Yescarta achieved impressive complete remission rates in late-stage clinical trials for blood cancers. Their subsequent approval by regulatory bodies has validated CAR T-cell therapy as a mainstream treatment option. Key factors powering the T-cell segment include improved understanding of T-cell biology and immunology, development of novel technologies for T-cell engineering and expansion, refinement of lymphodepletion regimens to improve T-cell engraftment, and identification of novel tumor-associated antigens. For instance, in November 2025, India expanded access to cancer immunotherapy by launching its first indigenous CAR-T cell therapy, NexCAR19, developed by ImmunoACT, a spin-off from IIT Bombay.
Oncology expected to hold largest market share of 70.2% in 2025. The oncology segment strongly drives the cell therapy market as the high prevalence of blood and solid cancers and the limitations of conventional treatments create urgent demand. Researchers and companies are advancing CAR-T, TCR, and other immune cell therapies to improve precision and effectiveness in cancer care. Robust clinical pipelines, successful trial results, and supportive regulatory measures are boosting adoption. For instance, in May 2025, HCG Manavata Cancer Centre and SunAct – Advanced Cancer Therapies opened SunAct’s fourth national Centre of Excellence in Cell and Gene Therapy at HCGMCC, Nashik, inaugurated by Dr. Shripad Banavali, Chair of Paediatric Haemato-Oncology at the Indian Academy of Paediatrics and Academic Director at Tata Memorial Hospital, Mumbai.
Hospitals acquired the prominent market share of 41.7% in 2025. Hospitals actively drive the cell therapy market by offering the infrastructure and clinical expertise needed to deliver complex treatments. They operate ICUs, infusion centers, and specialized laboratories to ensure safe administration and manage side effects. Hospitals conduct and support clinical trials, providing early patient access to innovative therapies. Their strong connections with payers and experience in reimbursement processes promote therapy adoption. Moreover, hospitals invest in manufacturing capabilities, integrate advanced technologies, and form strategic partnerships to expand cell therapy services and enhance patient access. For instance, in October 2025, CTMC, a joint venture between Resilience and MD Anderson Cancer Center, collaborated with Einstein Hospital Israelita in São Paulo, Brazil, to accelerate the adoption of cell therapy across Brazil and Latin America.
Artificial Intelligence (AI) is actively transforming the cell therapy market by advancing research, development, and manufacturing processes. AI algorithms analyze complex biological data to identify optimal cell targets, predict therapy outcomes, and accelerate drug discovery. In manufacturing, AI enhances quality control, optimizes cell expansion, and ensures consistent results. It also guides clinical decision-making by personalizing treatments and monitoring patient responses. By streamlining development, increasing precision, and minimizing errors, AI enables companies and healthcare providers to deliver safer, more effective, and accessible cell therapies. For instance, in February 2025, Cellino partnered with Mass General Brigham’s Gene and Cell Therapy Institute to launch the world’s first hospital-based autologous iPSC Foundry. Using Cellino’s AI-driven Nebula technology, the initiative aims to create a nationwide network of decentralized hubs delivering personalized cell and tissue therapies directly at the point of care.
To learn more about this report, Download Free Sample
North America dominates the overall market with an estimated share of 38.1% in 2025. North America’s cell therapy market is rapidly evolving as companies develop allogeneic “off‑the‑shelf” platforms, leveraging advanced manufacturing infrastructure and strong R&D capabilities. Leading biotech hubs are expanding modular, automated production models to increase efficiency. Accelerated regulatory pathways, including U.S. FDA designations, enable faster clinical advancement, while robust payer-provider networks and hospital-based cell therapy centers improve patient access. Biopharma firms and CMOs form strategic partnerships to streamline commercialization and scale delivery of therapies across the region. For instance, in October 2025, Visionary Holdings Inc. has signed a strategic agreement with Jiangsu Yike Regenerative Medical Technology to create Visionary Yike Stemcell Technologies Inc., a Canada-based joint venture focused on advancing stem cell research, clinical applications, and global commercialization.
The Asia‑Pacific cell therapy market is rapidly advancing as a global innovation hub, with Japan, China, and South Korea leading development through strong government support and extensive R&D investments. Companies in the region are conducting a surge of clinical trials, with over 650 ongoing CGT studies. Developers are expanding advanced regenerative platforms, including cell‑sheet therapies, while local CDMOs are scaling up capacity to meet growing demand and accelerate commercialization across the region. For instance, in November 2025, Science and Technology Minister Dr. Jitendra Singh launched India’s first indigenous CRISPR-based gene therapy for Sickle Cell Disease, primarily targeting the tribal population.
In the United States, companies are rapidly advancing the cell therapy market through allogeneic “off‑the‑shelf” CAR‑T platforms, leveraging a thriving CDMO ecosystem and expanding GMP manufacturing capacity. Researchers are applying cutting-edge gene editing and cell engineering technologies to develop new therapies. Regulatory agencies are actively streamlining approval pathways for innovative treatments, while clinical trial activity continues to increase. Biotech firms, contract developers, and hospital networks are forming strong collaborations to accelerate commercialization and enhance patient access to these therapies. For instance, in August 2025, ADIA Nutrition Inc. launched its second nationwide TV campaign, showcasing Adia Med and its advanced regenerative stem cell treatments at the flagship clinic in Winter Park, Florida.
China’s cell therapy market is rapidly advancing as the government provides strong support, regulatory agencies streamline approvals, and reimbursement frameworks expand. Domestic companies are developing both autologous and allogeneic CAR‑T and TCR therapies while reducing costs through process innovations. Biotech hubs in Shanghai, Shenzhen, and Beijing are scaling up GMP manufacturing and CDMO capabilities. At the same time, companies are conducting more clinical trials, and growing demand for cancer treatments is establishing China as a key player in the global cell therapy market. For instance, in January 2024, Fosun Kite Biotechnology launched China’s first outcome-based CAR-T cell therapy plan, offering cancer patients up to 600,000 yuan ($84,300) refund if the treatment fails.
The market is witnessing a shift from autologous to allogeneic cell therapies, allowing multiple patients to be treated from a single donor source. This trend enhances scalability, reduces manufacturing time, and lowers costs. Biotech companies are investing in universal donor cells and gene editing technologies to create off-the-shelf CAR-T and TCR therapies. Hospitals and CDMOs are adapting infrastructure to support these therapies, enabling faster patient access while maintaining quality and regulatory compliance across regions.
AI and automation are transforming cell therapy development, production, and clinical management. AI models analyze patient data to optimize cell selection, predict therapy responses, and streamline clinical trial design. Automated manufacturing systems enhance reproducibility, reduce human error, and accelerate production cycles. Together, these technologies improve efficiency, precision, and scalability. Stakeholders are increasingly leveraging AI-driven decision support tools to monitor patient outcomes and optimize treatment regimens, driving better clinical results and faster commercialization.
The shift from patient-specific autologous therapies to universal allogeneic platforms presents a major market opportunity. Off-the-shelf therapies reduce manufacturing complexity, shorten treatment timelines, and expand scalability. Biotech companies can invest in gene-editing and immunomodulation to create universally compatible cells. Hospitals and CDMOs can adopt modular production and automated systems to handle larger volumes, improving access and reducing cost barriers while enabling faster treatment delivery to multiple patients simultaneously.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2024 | Market Size in 2025: | USD 2,832.1 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2025 To 2032 |
| Forecast Period 2025 to 2032 CAGR: | 9.6% | 2032 Value Projection: | USD 5,383.4 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Bristol Myers Squibb Company, Amgen Inc., Novartis AG, Gilead Sciences, Inc., Johnson & Johnson Services, Inc., Allogene Therapeutics, Athersys, Inc., Orchard Therapeutics, JW Therapeutics Co., Ltd., Dendreon Pharmaceuticals LLC., Autolus Therapeutics, APAC Biotech, CORESTEM, Inc., BioCardia, Inc., JCR Pharmaceuticals Co., Ltd., Oxford Biomedica, CASI Pharmaceuticals Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
About Author
Abhijeet Kale is a results-driven management consultant with five years of specialized experience in the biotech and clinical diagnostics sectors. With a strong background in scientific research and business strategy, Abhijeet helps organizations identify potential revenue pockets, and in turn helping clients with market entry strategies. He assists clients in developing robust strategies for navigating FDA and EMA requirements.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients