Chagas Disease Treatment Market - Insights
Chagas disease is an endemic in 21 Latin American countries caused by kinetoplastid protozoan parasite, Trypanosoma cruzi, primarily transmitted by large, blood-sucking reduviid insects widely known as 'the kissing bugs'. Moreover, the disease can be transferred through other means such as blood transfusion, organ transplantation, as well as congenital and oral transmissions. The recent past has witnessed spread of Chagas disease to developed economies such as Europe and North America, due to migration of people from Latin America to others countries. For instance, according a study published in National Center for Biotechnology Information (NCBI), in 2015, 4.2% of Latin American living in European countries were chronically affected with Chagas disease. Benznidazole is the only FDA approved treatment indicated for the treatment of Chagas disease. However, the drug, Nifurtimox manufactured by Bayer AG, is in the World Health Organization's (WHO) list of 'Essential Medicines' and used in the treatment of Chagas disease.
Increasing approval to Chagas disease treatment drugs is expected to drive growth of the global Chagas disease treatment market
Benznidazole is an essential antiparasitic drug used for the treatment of Chagas disease. In April 2018, Argentinian National Administration for Drugs, Food, and Medical Technologies (ANMAT) approved Laboratorio Elea Phoenix SA’s drug benznidazole for pediatric indication used for the treatment of Chagas disease. Currently, Argentina has the highest number of people suffering from Chagas disease. For instance, according to data published by Mundo Sano (Nongovernmental foundation in Argentina), in 2017, around 7,300,000 people were exposed to Chagas disease and about 1,600,000 were infected, in Argentina. Hence, approval of novel drugs is expected to fuel the market growth in the near future. Such approval of Chagas disease treatment drugs is expected to boost growth of Chagas disease treatment market in the near future.
The global Chagas treatment disease market was valued at US$ 5.67 Mn in 2016 and is expected to witness a CAGR of 7.3% over the forecast period (2017–2025).
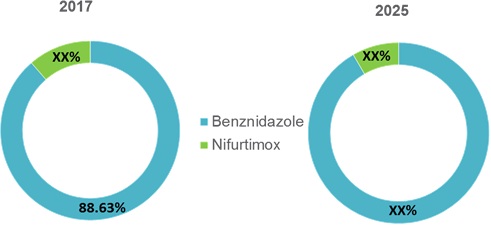
Figure 1. Global Chagas Disease Treatment Market Share (%), By Product Type, 2017 & 2025
To learn more about this report, Request Free Sample
Rising focus on initiatives by private and government organizations for increasing awareness of Chagas disease
In April 2018, Médecins Sans Frontières/Doctors Without Borders (MSF), an international humanitarian organization, launched a campaign. The campaign is aimed at increasing public awareness about Chagas disease as the disease is still prevalent in Latin American countries, and affects about 6 million people, globally. In 2007, the World Health Organization (WHO) launched a program to increase treatment rate and eliminate Chagas disease. This WHO program was supported by Bayer HealthCare, which manufactures nifurtimox, used in the treatment of Chagas disease. In 2007, Bayer entered into agreement with WHO to support the program by providing 2.5 million tablets free of charge to WHO, for the treatment of an estimated 30,000 patients over a period of five years (from 2007 to 2011). In 2011, WHO and Bayer extended its agreement. According to the agreement, Bayer is expected to provide 5 million tablets, free of charge, from 2012 to 2017. Such initiatives is expected to drive growth of market in the near future
Diagnostic Methods
Diagnosis of Chagas disease is done by serological methods, parasitological methods, and molecular method. Parasitological methods includes identification of trypomastigotes in blood by microscopy. Polymerase chain reaction (PCR) is a widely used molecular diagnosis method, as it is a good alternative tool for detection and shows high range of efficiency. Serological method is mostly used in the detection of Trypanosoma cruzi antibodies in blood donors, diagnosis of infected people and for epidemic studies. Enzyme-linked Immunosorbent Assay (ELISA), Indirect Immunofluorescence (IIF) are the widely used tests in serological method. The availability of rapid diagnostic test and point of care methods has made diagnosis of the disease more simple and quick. In 2016, InBios International, Inc. received 510K clearance from the U.S. Food and Drug Administration (FDA) for its Chagas Detect Plus Rapid Test Kit (CDP).
Key players operating in the Chagas disease treatment market include Nortec Quimica SA, Bayer AG, Laboratorio Elea Phoenix SA, Maprimed S.A., and Laboratório Farmacêutico de Pernambuco S/A.
Share
Share
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients