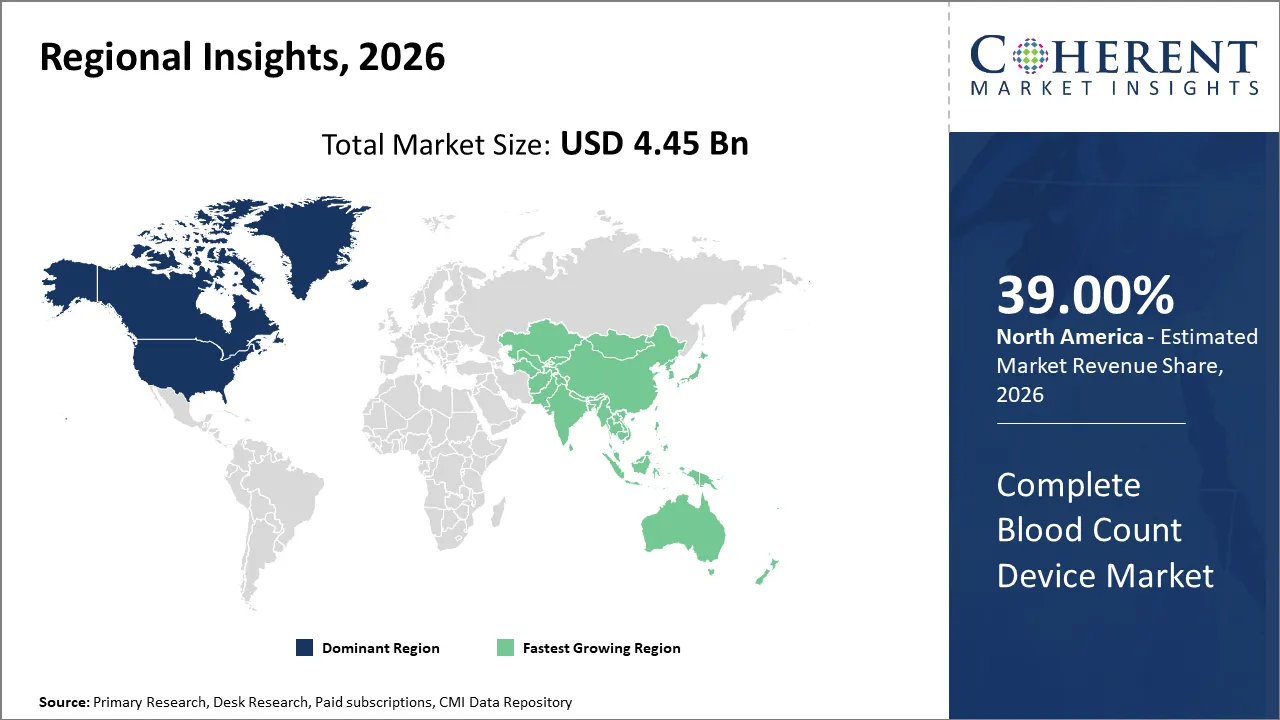
The Complete Blood Count Device Market is estimated to be valued at USD 4.45 Bn in 2026 and expected to reach USD 7.85 Bn by 2033, witnessing a CAGR of 8.1% over the forecast period (2026-2033). This market is primarily driven by the increasing prevalence of blood disorders and rising demand for rapid diagnostic testing across hospitals and laboratories.
The market for complete blood count device represents a key segment in the in-vitro diagnostics (IVD) industry. This important segment consists of devices used to analyze the various components of blood including red blood cells (RBCs), white blood cells (WBCs), hemoglobin level, hematocrit percentage, and platelet count. Hospitals, diagnostic laboratories, research Institutions, and blood banks commonly utilize complete blood count devices to perform routine health screenings and to diagnose diseases.
Complete blood count devices typically employ various technology such as electrical impedance, laser-based detection, and flow cytometry technology, to provide a rapid and precise evaluation of blood samples. Because these devices are designed to aid clinicians in diagnosing conditions such as anemia, infection, leukemia, inflammatory disease, and immune disorders, they are invaluable tools for clinicians.
|
Current Event |
Description and its Impact |
|
AI-Enabled Hematology Analyzer Launch (2025) |
|
|
Expansion of Preventive Health Screening Programs |
|
|
Growth of Point-of-Care Diagnostics |
|
|
Laboratory Automation Initiatives |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
A growing presence of AI in the market for complete blood count devices is contributing to the diagnostic accuracy and efficiency of laboratory operations. AI algorithms allow for the automatic identification of abnormal cell morphology and will alleviate the need for manual review by the laboratory techs. Predictive analytics that utilize AI have also introduced the possibility for hematology analyzers to assist providers in identifying potential disease states prior to clinical diagnosis, thereby enhancing the quality of care delivered to patients.
In April 2025, Sysmex launched an AI-enabled hematology analyzer capable of detecting abnormal cell populations using machine learning algorithms.
The 5-part Analyzer segment accounted for approximately 46% of the global complete blood count device market share in 2026. Due to its advanced ability to differentiate five types of white blood cells, enabling more accurate diagnosis of infections, leukemia, and inflammatory conditions. Hospitals and diagnostics laboratories prefer these analyzers for their high precision and comprehensive hematology testing capabilities. For instance, large hospital laboratories commonly deploy 5-part hematology analyzers for high-volume routine CBC testing and advanced blood cell differentiation.
In 2026, the stationery device segment dominated the complete blood count device market with about 62% of the total market share due to their higher throughput and detailed analysis of blood compared to portable devices. Stationery devices can be found in most centralized hospital laboratories and diagnostic centers because they routinely process large volumes of blood samples on a daily basis. For instance, high-capacity stationery hematology analyzers installed in tertiary hospitals can process hundreds of blood samples per hour.
The white blood cell count segment accounted for approximately 34% of the market in 2026. White blood cells testing, which includes the count of neutrophils, eosinophils and basophils, was a large share of the global market because WBC testing is important for the detection of infection, immune disorder and inflammatory disease. The intensity and routine frequency of WBC testing as part of CBC testing support physicians in diagnosing and obtaining pertinent information regarding the status of a patient’s total health. For instance, doctors frequently order WBC count tests to detect bacterial infections or immune system abnormalities in patients.
In January 2024, a Rutgers researcher, through his spinoff company, led a team that developed and tested a device capable of quickly counting white blood cells from a single drop of blood, similar to how glucometers measure blood sugar levels.
The hospital segment holds around 55% of the global market share in 2026, as hospital laboratories are performing the most diagnostic tests. Hospital laboratories routinely performed CBC tests to assist with determining the diagnosis for a patient, continued evaluations of a patient’s medical condition and for determining pre-surgical evaluations. For instance, emergency departments routinely conduct CBC tests to assess infections, anemia or internal bleeding in critical patients.
To learn more about this report, Request Free Sample
The North America leads the total blood count device market because of its sophisticated healthcare system, large number of diagnostics performed and wide acceptance of automated hematology analyzers. The presence of prominent diagnostic companies, favorable reimbursement practices and established clinical laboratories continues to aid to the widespread use of total blood count testing devices within hospitals and diagnostic labs.
The Complete Blood Count Device Market is expected to grow rapidly in Asia Pacific with continued growth in healthcare infrastructure, increased awareness of diagnostic services and increased investments in modernizing hospitals and laboratories. As the number of cases of Anemia and infectious diseases increase, the demand for blood tests also continue to grow within the region.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 4.45 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 8.1% | 2033 Value Projection: | USD 7.85 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Abott Laboratories, bioMérieux SA, Danaher Corporation, Siemens Healthineers, Bio-Rad Laboratories, Inc., PerkinElmer, Inc., Sysmex Asia Pacific Pte Ltd, Becton, Dickinson and Company, Sight Diagnostics, Horiba, and Nihon Kohden Corporation |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
As hematological disorders become more common, from anemia to leukemia to thrombocytopenia, there will be much greater need for testing equipment. CBC testing devices are relied upon by medical professionals for both initial diagnosis and ongoing monitoring, so they are an essential piece of equipment in both hospitals and medical laboratories.
The use of automated diagnostic equipment is becoming prevalent in many years of healthcare, due to a desire to increase efficiency and decrease risk of error from human operators. Automated CBC analyzers allow laboratories to process large quantities of blood samples with high levels of diagnostic accuracy, which helps improve testing volume.
The increasing need for the ability for fast diagnostics at hospitals and remote healthcare facilities has resulted in a growing demand for portable CBC devices that are able to deliver results quickly. Point-of-Care devices allow healthcare professionals to test patients and get their results in less time than if they had sent samples to a laboratory.
Emerging market providers like India, China and Brazil are spending considerable resources to develop their healthcare infrastructure. The growth of hospitals, diagnostic centers and laboratory networks continues to provide new avenues for growth for the manufacturers of CBC devices.
The complete blood count device market is projected to continue to grow steadily as more diagnostic tests are being done, and technology for hematology analyzers continues to advance. According to experts, the landscape for hematology diagnostics will change significantly in the next decade due to automation of devices, integration with artificial intelligence and portable, point-of-care testing devices. Businesses that produce AI-enabled hematology analyzers and develop affordable point-of-care testing solutions will be positioned to be more competitive in the future.
Share
Share
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients